Q: 1. An ester (MW=136) give a Mass spectrum (E.I.) as following. Please deduce the structure of this…
A: Let's analyze the mass spectrum of the ester with a molecular weight of 136.Mass Spectrum Graph:The…
Q: A. Molar Volume of Hydrogen Gas mass of magnesium 0.150 g 0.139 g volume of hydrogen gas 158 ml 141…
A: References: General Chemistry Textbooks:Tro, Nivaldo J. Chemistry: A Molecular Approach. Provides a…
Q: H3C EtO₂C N CH3 Nefidipine from benzaldehyde and alkene derivative CO₂Et
A: Nefidipine is a pharmaceutical drug used to treat chest pain, It is synthesized from benzaldehyde…
Q: This question concerns part 1 of this lab. You need to make a buffer solution by mixing both a weak…
A: Given,molarity of sodium acetate = 0.100 Mvolume of sodium acetate = 5.00 mLpH of the solution =…
Q: i Provide the missing curved arrow notation. ס12 + од H C N O S F P C1 Br
A:
Q: ← Draw the products of the reaction shown below. Ignore inorganic byproducts. CH3(O)CI AICI 3…
A: This is a Friedel-Crafts reaction. In this reaction, a coupling reaction which involves an…
Q: The pK of butyric acid (HBut) is 4.82. Calculate the K₁ for butyrate ion (But). Be sure your answer…
A: Step 1: Step 2: Step 3: Step 4:
Q: A Please predict the products for each of the following reactions: Hint: Treat D (deuterium atom)…
A:
Q: In the reaction A → B + C the concentration of A decreases exponentially with time. Why can't the…
A: The objective of the question is to understand why the rate equation for the reaction A → B + C…
Q: 7. What are the units of k in a second order reaction? A. Ms¹ B. Ms C. M¹¹ D. M²-1 E. S¹
A: Answer:Here, n is the order of reaction
Q: 20 Draw the correct product for the reaction. (If there is no reaction, draw the starting material.)…
A: Na2Cr2O7, H2SO4 oxidises primary alcohol to Carboxylic acid and secondary alcohol to ketone but it…
Q: C. Constitutional isomers D. Two deawing if the sambe comoound
A: Step 1:Cis isomers are the isomers that have two similar atoms lying on the same side of the double…
Q: Give IUPAC names for the following compounds.
A: IUPAC nomenclature is used for naming the organic compound as recommended by the International Union…
Q: One way the U.S. Environmental Protection Agency (EPA) tests for chloride contaminants in water is…
A: Balanced equation for the reaction between CuCl2 and AgNO3 is given as:CuCl2 (aq) + 2AgNO3 (aq) →…
Q: The electron transport chain (ETC), or respiratory chain, is linked to proton movement and ATP…
A: The statements that accurately describe the electron transport chain (ETC) are:Electron carriers are…
Q: The mole fraction of neon in dry air near sea level is 0.00001818, where the molar mass of neon is…
A: Step 1: Use the Raoult's Law to solve the problem. Refer to the solution below.
Q: What mass of sodium lactate (MM = 112.06 g/mol) must be added to 500 mL of 0.100 M lacticacid,…
A: The objective of this question is to find out the mass of sodium lactate that needs to be added to a…
Q: Question 3 What mass of NiCO, will dissolve in 1.4 L of 0.502 M NaCO₂ solution? K of NICO, is…
A: Given: [CO32−]=0.502M;V=1.4L;Ksp=6.6x10−9;massNiCO3=???g;MNiCO3=118.702g/molStep 1: Write the…
Q: When naphthalene undergoes an irreversible electrophilic aromatic substitution, the major product is…
A:
Q: Student Discipline Problems The data for a random sample of 32 months for the number of discipline…
A:
Q: What are the products A and B? 1. LDA 1. NaOEt A 2. CH3CH2Br 2. Ph Br 1. NaOEt 1. LDA 2. CH3CH2Br 2.…
A:
Q: dentify the best reagent(s) for the reaction shown below. ཙ
A: The objective of this question is to identify the appropriate reagents and understand the mechanisms…
Q: triangular pyramid,
A: For an equilateral triangle with side length s, the altitude (h) is given by:h = √(s^2 - (s/2)^2)In…
Q: For each system listed in the first column of the table below, decide (if possible) whether the…
A: Step 1:Entropy is the degree of disorderness or degree of randomness. As Temperature increases,…
Q: Draw the major products of this reaction. Ignore inorganic byproducts. H3O+ Draw Carbonyl Product
A: It is acidic hydrolysis of imine.An imine is functional group characterized by a carbon-nitrogen…
Q: Here are sketches of four electron orbitals: O A B C D O yes Are any of them s orbitals? 0,0,... no…
A: Answer:Orbital is the region around the nucleus where probability of finding an electron is maximum.…
Q: A 1.211 g of benzoic acid (CH§ COOH) was ignited in pure oxygen gas and the heat released from the…
A: Here's a step-by-step explanation:1. **Understanding the System**: We have benzoic acid (C6H5COOH)…
Q: A patient visits her primary care because of the development of symptoms including excessive upper…
A: The objective of the question is to identify the possible medical condition based on the given…
Q: The standard enthalpy of atomization of an element is the energy required to convert one mole of an…
A: Number of moles = 2.77Enthalpy of atomization = 107.5 kj/molIonization energy = 495.9 kj/mol
Q: Which of the following compounds undergoes the most rapid nucleophilic substitution with hydroxide…
A:
Q: Draw the products of the reactions. Include hydrogen a HC=CH H* CH + H2O “་ཡ་ཡང་ཡ་རི། - H3C-C CH3…
A: The problem considers two esters.The first reaction requires the products formed when ester is…
Q: For a compound below tell how many types of nonequivalent protons there are. C1 04 06 05 07
A: We have to find the number of non-equivalent protons,i.e.- The unique protons that are present in…
Q: d) (2 pts) BHCI CH3 + H₂C 1. Mix 2. NaOH, H₂O₂ D
A:
Q: Please show reaekson and don't use hend and correct answer
A: Let's analyze the IUPAC name for the compound 3-ethyl-2,2-dimethyl-1-pentanol and understand the…
Q: Draw the major elimination and substitution products formed in this reaction. Use a dash or wedge…
A: Step 1: Step 2: Step 3: Step 4:
Q: Find the pH during the titration of 20.00 mL of 0.1000 M triethylamine, (CH3CH2)3N, with 0.1000 M…
A: Answer:Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: Question 10 A photon is quantum of electromagnetic radiation. A True B False
A:
Q: (please type answer fast).
A: The objective of this question is to calculate the pH of the solution after a certain volume of…
Q: Suppose a 0.018M aqueous solution of phosphoric acid (H3PO4) is prepared. Calculate the equilibrium…
A: Step 1:[H3PO4] = 0.018 M[HPO42-] = ?Ka1 = 7.5 * 10-3 Ka2 = 6.2 * 10-8 Step 2: First…
Q: Draw the major product of the following reaction. CH3 C(CH3)3 HNO3 H2SO4
A: The given reaction is an example of the nitration of benzene. It is an electrophilic aromatic…
Q: Please don't provide handwriting solution
A: The objective of the question is to determine the signs of the enthalpy change (ΔH) and the entropy…
Q: A chemist dissolves 293. mg of pure potassium hydroxide in enough water to make up 160. mL of…
A: Given that,Mass of pure potassium hydroxide,Molar mass of Volume of the solution,pH of the solution…
Q: Question 24 Identify the product (or multiple products) for each of the following reactions: A BCDEF…
A: Step 1:Step 2: Step 3: Step 4:
Q: What is the pH of 55 g of Mg(OH )2 (Ksp = 5.61 * 10^-12) dissolved in 1.2L of water?
A: The objective of this question is to calculate the pH of a solution of Mg(OH)2 in water. The pH of a…
Q: What is the product? Br CN 1. NaOEt 2. DIBAL 3. H3O+
A: NaOEt is a strong base,DIBAL-H is a reducing agent.H3O+ is acidic water workup.
Q: Draw the major elimination and substitution products formed in this reaction. Use a dash or wedge…
A: Given that, the reaction is:
Q: Lepadiformine is a cytotoxic marine alkaloid that has cardiovascular biological activity. Draw the…
A: SN2 reaction occurs in a single step. Here, the nucleophile is S^- species while electrophile is Br…
Q: Help solve 6.
A: Given: [Ca2+]=2.0x10−3M;massF−=1.0g;V=1.0x103L;MF−=19.00g/mol;Ksp=3.45x10−11The Ksp value is…
Q: w structural f
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: None
A: Step 1:- Most soluble amine (i) because of shorter carbon chain length and hydrogen bond formation…
Step by step
Solved in 2 steps with 1 images

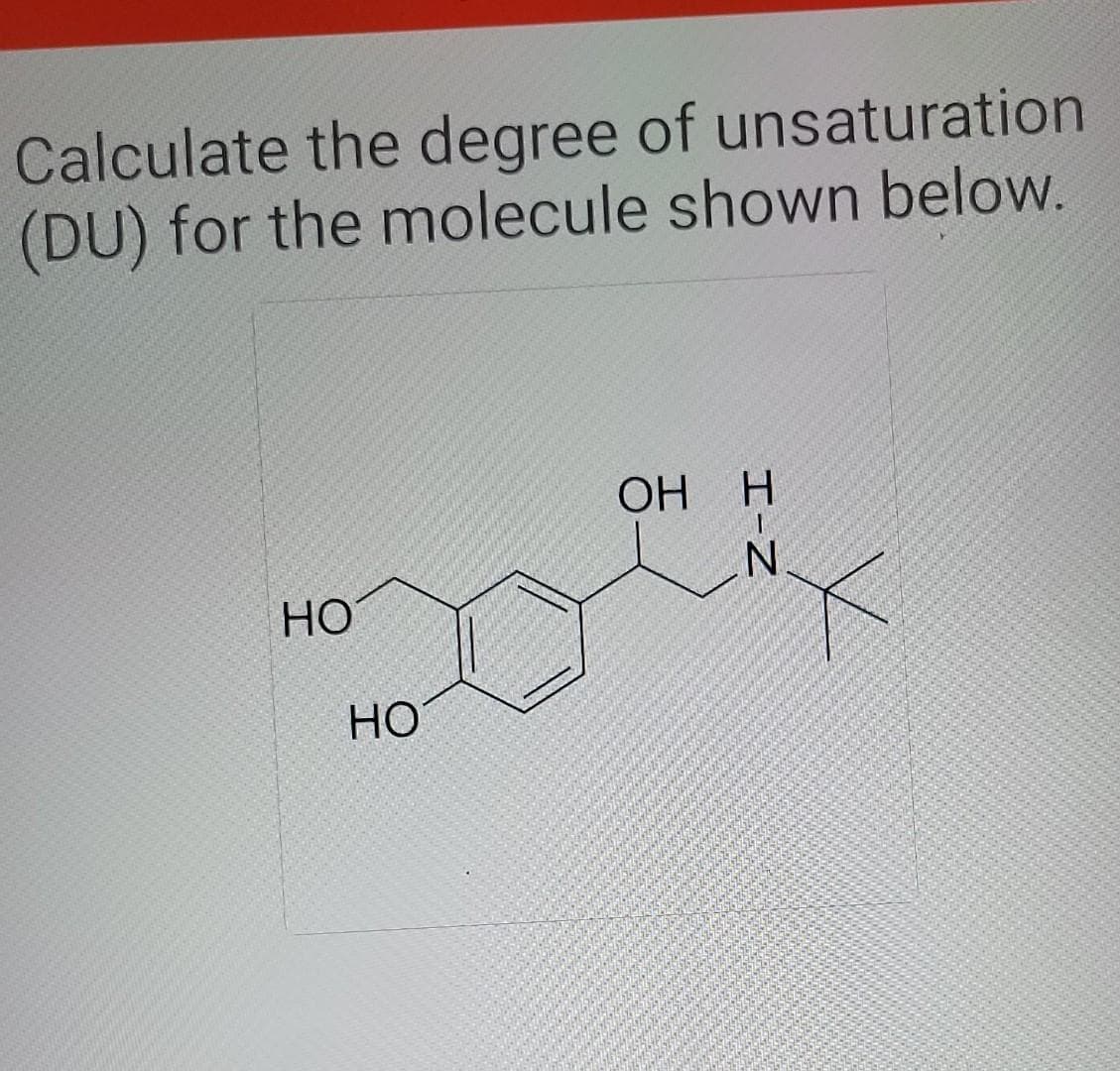
- Determine the degree of unsaturation for each of the following compounds. d. C8H8O4 e. C21H30O2 f. C17H21NO4how do i determine degree of unsaturation for an equation if it includes elements such as N, O, F. when each of these are present in the formula do i subtract, add, or leave it the same from the amount of carbons present. example includes C6H7N or C4H7BrDetermine the degree of unsaturation for each of the following compounds. a. C8H7NO b. C4H4BrNO2 c. C5H3ClN4 d. C8H8O4 e. C21H30O2 f. C17H21NO4
- Consider 1-bromo-2-methylpropane and draw the following. (a) The staggered conformation(s) of lowest energy (b) The staggered conformation(s) of highest energyCalculate the number of degrees of unsaturation for each molecular formula:(a) C5H8O; (b) C6H11Cl; (c) C8H9N. Propose one possible structure for eachcompound.Calculate the number of degrees of unsaturation for each molecular formula, and propose two possible structures: (a) C8H12; (b) C10H10.



