Calculate the energy in Joules required to convert 72 g of ice originally at -15°C into steam at 105°C. Specific Heats of Common Materials Specific °C Specific latent heat of Substance °C latent heat MATERIAL SPECIFIC HEAT vaporization J.g1 of fusion (Joules/gram • °C) J.g1 Liquid water Solid water (ice) Water vapor 4.18 Water 334 2258 100 2.11 Ethanol 109 838 78 -114 2.00 Ethanoic 192 17 395 118 acid Dry air Basalt 1.01 Chloroform 74 -64 254 62 0.84 Mercury 11 -39 294 357 Granite 0.79 Sulphur 54 115 1406 445 Iron Соpper 0.45 Hydrogen Охудеn Nitrogen 60 -259 449 -253 0.38 -219 -210 14 213 -183 Lead 0.13 199 -196
Calculate the energy in Joules required to convert 72 g of ice originally at -15°C into steam at 105°C. Specific Heats of Common Materials Specific °C Specific latent heat of Substance °C latent heat MATERIAL SPECIFIC HEAT vaporization J.g1 of fusion (Joules/gram • °C) J.g1 Liquid water Solid water (ice) Water vapor 4.18 Water 334 2258 100 2.11 Ethanol 109 838 78 -114 2.00 Ethanoic 192 17 395 118 acid Dry air Basalt 1.01 Chloroform 74 -64 254 62 0.84 Mercury 11 -39 294 357 Granite 0.79 Sulphur 54 115 1406 445 Iron Соpper 0.45 Hydrogen Охудеn Nitrogen 60 -259 449 -253 0.38 -219 -210 14 213 -183 Lead 0.13 199 -196
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter4: Energy And Chemical Reactions
Section: Chapter Questions
Problem 40QRT: Calculate the quantity of heating required to convert the water in four ice cubes (60.1 g each) from...
Related questions
Question
Please answer fast
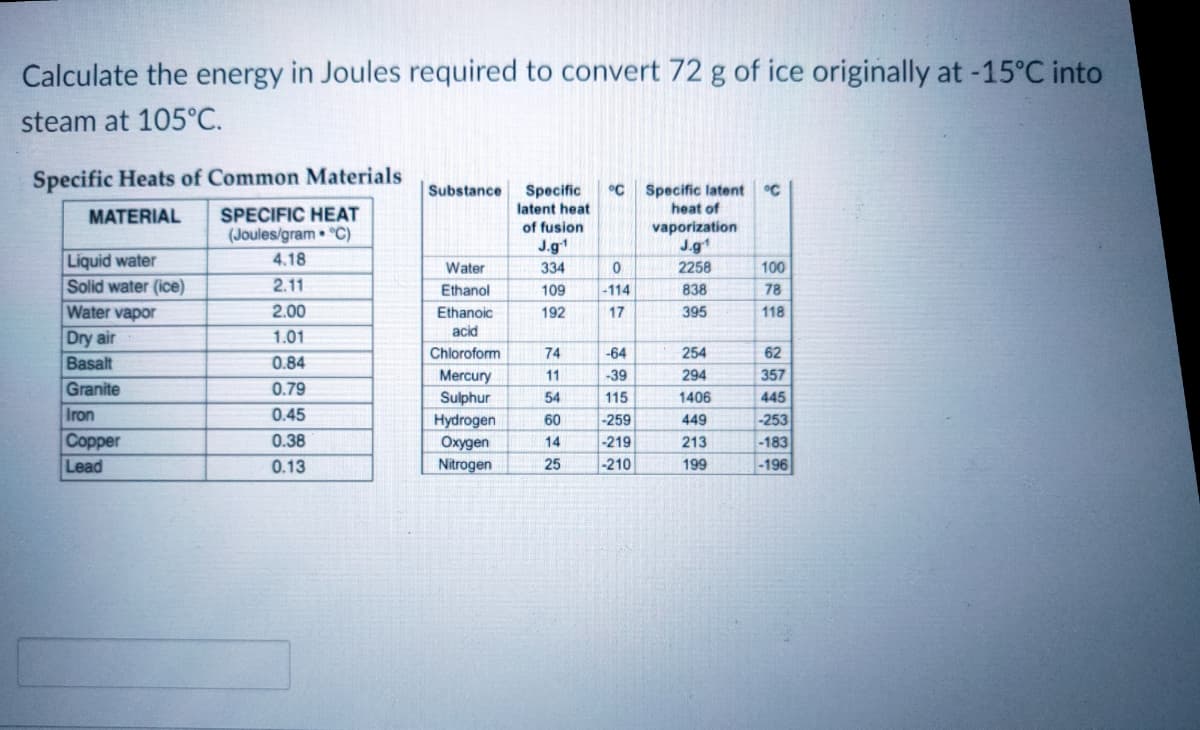
Transcribed Image Text:Calculate the energy in Joules required to convert 72 g of ice originally at -15°C into
steam at 105°C.
Specific Heats of Common Materials
°C
Specific latent
heat of
Substance
°C
Specific
latent heat
MATERIAL
SPECIFIC HEAT
of fusion
(Joules/gram • °C)
vaporization
J.g1
J.g1
Liquid water
Solid water (ice)
Water vapor
4.18
Water
334
2258
100
2.11
Ethanol
109
-114
838
78
2.00
Ethanoic
192
17
395
118
acid
Dry air
Basalt
Granite
Iron
Соpper
1.01
Chloroform
74
-64
254
62
0.84
Mercury
Sulphur
Hydrogen
Охудen
Nitrogen
11
-39
294
357
0.79
54
115
1406
445
0.45
60
-259
449
-253
0.38
14
-219
213
-183
Lead
0.13
25
-210
199
-196
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning