Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question

Transcribed Image Text:Derive an equation of the type pe =
? - ?pH which describes the line on
a Pourbaix diagram which divides
the MnO2 (s) and Mn2+ stability
regions. Assume a solubility limit of
10-6M for any soluble species.
Select one:
а. 25.8- 2 pH
b. 18.8 + 2 pH
с. 18.8-2 рH
d. 23.8 - 2 pH
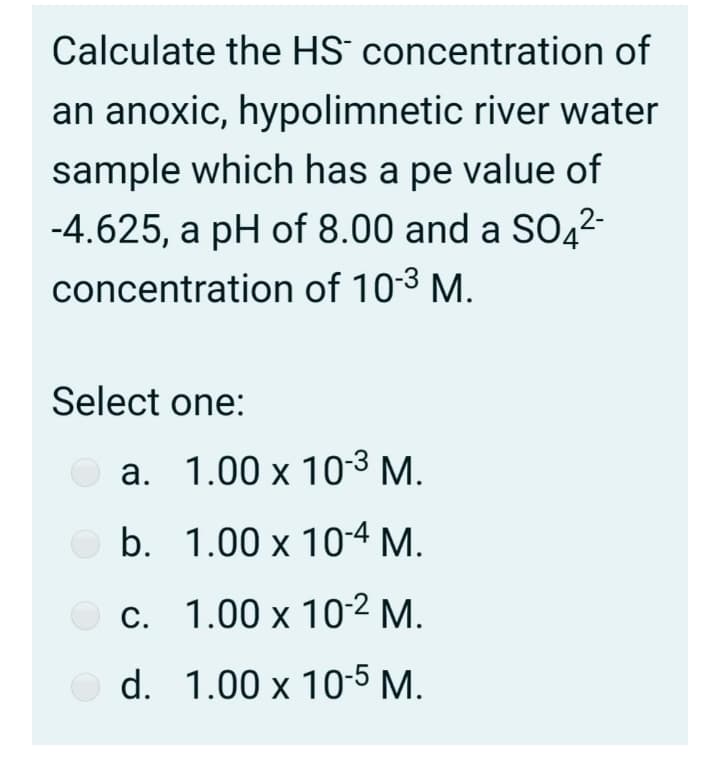
Transcribed Image Text:Calculate the HS concentration of
an anoxic, hypolimnetic river water
sample which has a pe value of
-4.625, a pH of 8.00 and a SO4²-
concentration of 103 M.
Select one:
a. 1.00 x 103 M.
b. 1.00 x 10-4 M.
O c. 1.00 x 10-2 M.
O d. 1.00 x 10-5 M.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning