Chapter80: Crystallization: Purification Of Solids
Section: Chapter Questions
Problem 4P
Related questions
Question
Calculate the number of moles of NaOH used in the reaction . 20% NaOH solution was used .
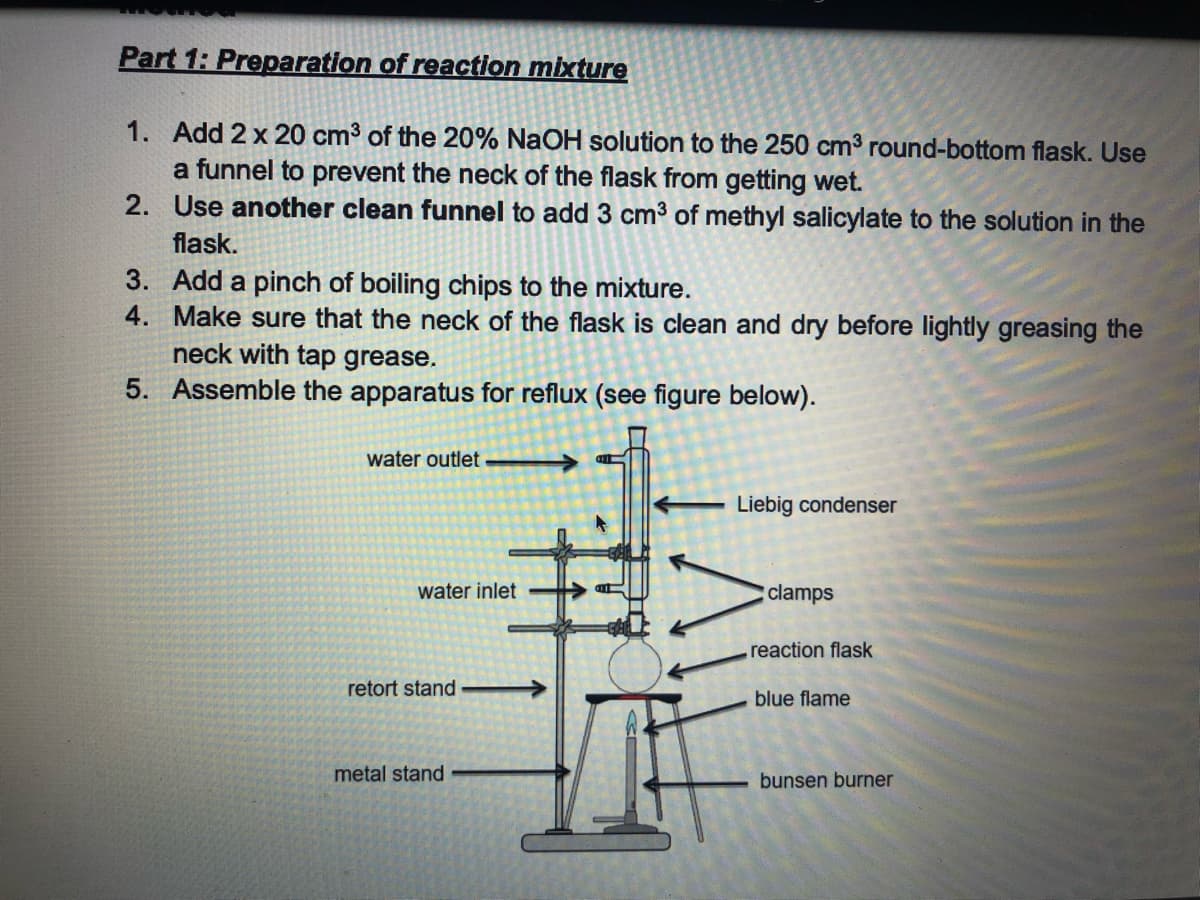
Transcribed Image Text:Part 1: Preparation of reaction mixture
1. Add 2 x 20 cm3 of the 20% NaOH solution to the 250 cm3 round-bottom flask. Use
a funnel to prevent the neck of the flask from getting wet.
2. Use another clean funnel to add 3 cm3 of methyl salicylate to the solution in the
flask.
3. Add a pinch of boiling chips to the mixture.
4. Make sure that the neck of the flask is clean and dry before lightly greasing the
neck with tap grease.
5. Assemble the apparatus for reflux (see figure below).
water outlet
Liebig condenser
water inlet
clamps
reaction flask
retort stand
blue flame
metal stand
bunsen burner

Transcribed Image Text:cical Manual_Praktika Handleiding_2021.pdf
28 / 86
136%
Part 2: Reflux reaction
6. Open the tap and ensure that the Liebig condenser is completely filled with running
water. The piping and water will be already switched on using the water saving
system. Ensure that the pipes are handled with care as water can easily leak out.
7. Light the Bunsen burner and place it + 2 cm below the round-bottom flask to heat
the solution.
8. Boil the mixture under reflux for 30 minutes.
9. Extinguish the flame and allow the flask and its contents to cool to room
temperature.
10. Quantitatively transfer the reaction mixture to a 250 cm3 glass beaker.
Work-up of product
11.Acidify the reaction mixture by adding 3 M H2SO4 (approximately +5 cm3 at a time).
Use Congo red paper to determine when the mixture is acidic.
12.After confirming that the reaction mixture is acidic add a further 15 cm3 of H2S04.
13.Place the beaker in an ice bath (a larger beaker containing ice and water) to cool
down the mixture. A white precipitate should form as your final product.
26
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole