Calculate the solubility at 25 °C of Zn(OH), in pure water and in a 0.0180M ZnSO, solution. You'll find Ken data in the sp ALEKS Data tab. Round both of your answers to 2 significant digits. 믐 solubility in pure water: solubility in 0.0180 M ZnSO4 solution: :
Calculate the solubility at 25 °C of Zn(OH), in pure water and in a 0.0180M ZnSO, solution. You'll find Ken data in the sp ALEKS Data tab. Round both of your answers to 2 significant digits. 믐 solubility in pure water: solubility in 0.0180 M ZnSO4 solution: :
Chapter10: Effect Of Electrolytes On Chemical Equilibria
Section: Chapter Questions
Problem 10.16QAP
Related questions
Question
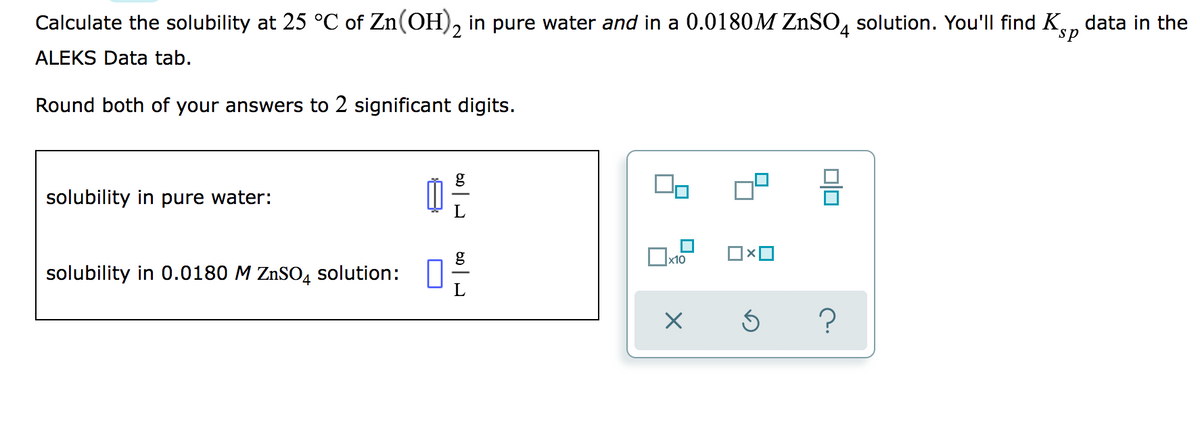
Transcribed Image Text:Calculate the solubility at 25 °C of Zn(OH), in pure water and in a 0.0180M ZNSO, solution. You'll find K, data in the
sp
ALEKS Data tab.
Round both of your answers to 2 significant digits.
g
solubility in pure water:
x10
solubility in 0.0180 M ZnSO4 solution:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning