Calculate the values expected for the total alkalinity and for the phenol- phthalein alkalinity of a 25°C saturated solution of calcium carbonate in water, and compare them to the values for a solution that also is in equilib- rium with atmospheric carbon dioxide. Use the concentrations quoted in the last column of Table 10-4.
Calculate the values expected for the total alkalinity and for the phenol- phthalein alkalinity of a 25°C saturated solution of calcium carbonate in water, and compare them to the values for a solution that also is in equilib- rium with atmospheric carbon dioxide. Use the concentrations quoted in the last column of Table 10-4.
Chapter10: Effect Of Electrolytes On Chemical Equilibria
Section: Chapter Questions
Problem 10.11QAP
Related questions
Question
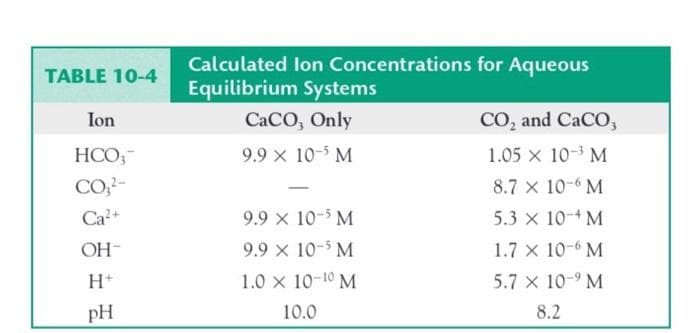
Transcribed Image Text:Calculated lon Concentrations for Aqueous
Equilibrium Systems
TABLE 10-4
Ion
CaCO, Only
CO, and CaCO,
HCO,-
9.9 x 10-5 M
1.05 x 10-3 M
CO,?-
8.7 x 10-6 M
Ca?+
9.9 x 10-5 M
5.3 x 10-4 M
OH-
9.9 x 10-5 M
1.7 x 10-6 M
H*
1.0 x 10-10 M
5.7 x 10-9 M
pH
10.0
8.2

Transcribed Image Text:Calculate the values expected for the total alkalinity and for the phenol-
phthalein alkalinity of a 25°C saturated solution of calcium carbonate in
water, and compare them to the values for a solution that also is in equilib-
rium with atmospheric carbon dioxide. Use the concentrations quoted in the
last column of Table 10-4.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

