Chapter22: Carbonyl Alpha-substitution Reactions
Section22.SE: Something Extra
Problem 57AP: All attempts to isolate primary and secondary nitroso compounds result solely in the formation of...
Related questions
Question
1
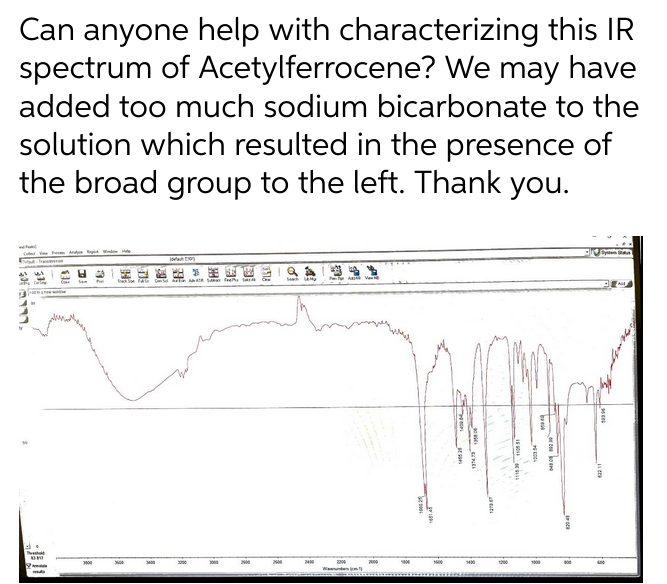
Transcribed Image Text:Can anyone help with characterizing this IR
spectrum of Acetylferrocene? We may have
added too much sodium bicarbonate to the
solution which resulted in the presence of
the broad group to the left. Thank you.
GM
A
7400
3600
ja
幽幽影圈四
FAGARTH AWARS Free Sa
3400
IQ A
LANG
2400
2009
Waber
2004
be
M
13747313806
1400
12:00
111.3 110851
1000
be
1200
000
System Sta
LIZE
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning