Q: [Review Topics] [References] Devise a synthesis of butane using one of the starting materials and…
A: Synthesise the butane from suitable alkyne and select suitable reagents On hydrogenation alkynes are…
Q: Which of the following statements correctly describe(s) SN2 reactions of alkyl halides (RX)? I. Rate…
A: SN 2 reaction is nucleophilic substitution reaction.
Q: Use VSEPR theory to predict the electron-pair geometry and the molecular geometry of XeF5+.
A:
Q: In an acid-base titration, the neutralization of 20.00 mL of a solution of HClO (perchloric acid)…
A: Given, Note: 1000 mL = 1 L Perchloric acid (HClO) solution Molarity of perchloric acid (HClO)…
Q: For the following reaction, 20.9 grams of iron are allowed to react with 9.42 grams of oxygen gas.…
A:
Q: Zinc was analysed in acid mine drainage (AMD). The signal obtained from an AAS for the sample was…
A:
Q: QUESTION 25 When 2.5 moles of calcium carbonate is added to 4.8 moles of hydrochloric acid, calcium…
A:
Q: Arrange the following compound in the order of increasing acidity (least acidic first) I)…
A: Acidic character of the given organic compounds can be explained by the presence of replaceable…
Q: (a) Calculate the energy of a single photon of light with a frequency of 6.38×108 s-¹. Energy = J…
A: Given, The frequency of a light is 6.38 × 108 s-1 The energy of a single photon is:
Q: Given the following reactions N2 (g) +202 (g) → 2NO2 (g) 2NO(g) + O2 (g) → 2NO2 (g) ΔΗ = -114.2 kJ…
A:
Q: (a) What is the main difference between a UV-Vis spectrophotometer and a spectrofluorometer? (b)…
A:
Q: 6) Calculate the molar mass of Na₂SO4. OA) 110.1 g/mol OB) 119.1 g/mol OC) 94.05 g/mol O D) 71.06…
A: We have to calculate molar mass of Na2SO4 .
Q: 2. MS: a. Identify and give the m/z for the base peak and molecular ions b. Determine n appropriate…
A: This question is from mass spectrometry. Base peak : The tallest peak in the spectrum is known as…
Q: For Water at 15 bar and 400°C What is the specific internal energy in kJ/kg?
A:
Q: show the synthetic scheme to achive a final product L R-C-0-0-4 !! OH он 4 DR. CO₂ H 2) H₂ot Ⓒ
A:
Q: Calculate the hydronium ion concentration in an aqueous solution that has a pH of 6.65
A:
Q: What is the correct IUPAC name of the following compound? Cl Problem viewing the image, Click Here…
A:
Q: Acid-catalyzed dehydration of 3,3-dimethyl-2-butanol gives three alkenes: 2,3-dimethyl-2-butene,…
A: -> Reactant -> 3,3-dimethyl-2-butanol -> Products-> 2,3-dimethyl-2-butene ,…
Q: E 107 100- 90 $840.57cm-195.73 % 1 3819 20cm-1.95.92 T 3355.60cm-1, 96.01% T 80- 70- 60 50 3691…
A: Answer. The vampire fang like structure in the above given spectra at 3480 cm-1 and 3357 cm-1…
Q: a. Identify whether the reaction of Na(s) and Br₂ (l) is exothermic or endothermic. Use the…
A: Exothermic reaction and endothermic reaction: Exothermic reaction: Exothermic reactions are reaction…
Q: Under standard state conditions, the complete combustion of a hydrocarbon, primarily produces: O…
A: -> Combustion is a chemical reaction between fuel and an oxidant that produces an oxidized…
Q: Which of the following molecular formulas is reasonable for a stable compound? O a. C7H10NCI b.…
A: -> For stable compound the reasonable molecular formula must contains integer degree of…
Q: Glutamine is a OA. basic OB. acid O c. neutral O D. racemin amino acid:
A:
Q: Arrange the following compound in the order of increasing acidity (least acidic first) I)…
A: Given compounds: I) Cyclohexanol II) 4-Nitrophenol III) Phenol IV) 4-Chlorophenol The acidity…
Q: [Review Topics] The functional groups in an organic compound can frequently be deduced from its…
A: IR spectroscopy is useful to determine functional groups of organic compoundc
Q: addition to oxygen, what other substance is produced by this reaction? Where is this substance…
A:
Q: B. The copper concentration in a contaminated salt sample was determined by flame atomic absorption…
A: Here standard addition technique is used to standardize the copper sample in which to a fixed volume…
Q: 2.1) Draw the structure of "major organic final product" of the following Addition of H₂O to alkyne…
A: Here we have to predict the major product formed in the hydration of the following given alkyne in…
Q: What alkene would you use to create 2-propoxy-(s)-propane. Prove your answer by showing a chemical…
A:
Q: Unshared, or lone, electron pairs play an important role in determining the chemical and physical…
A: Elements have electrons in valence shell called valence electrons . In formation of compounds…
Q: A Calculate aqueous in the following. E [OH-] solution at 25 °C: [1/₂0+] = 1.0 × 10-8 M. © Calculate…
A:
Q: Element Atomic Symbol Symbol Be 121 Sb 51 Atomic Mass Number Number 8 Protons Neutrons Electrons 51…
A: -> Atomic number = number of proton = number of electron (for element) -> Atomic mass = proton…
Q: When the Pb2+ concentration is 1.35x104 M, the observed cell potential at 298K for an…
A:
Q: This question contains 2 Parts. Provide your answers to the following questions. Indicate your…
A: Synthon is the charged fragment produced by disconnection. Synthetic equivalent is the actual…
Q: Which of the following molecules has an R configuration? COOH CH CH OH З H₂C CH₂ HOCH COOH CH cooH…
A: We have to determine the configuration and select the molecules having R configuration
Q: Which of the following represents the correct Cahn-Ingold-Prelog priority ranking of C5H11…
A:
Q: What are the coefficients?
A: Given that, a redox reaction is H5IO6 + Cr → IO3- + Cr3+. We have to write the coefficients of the…
Q: αcarboxyl and α-amine are 2.2 and 9.1 perspectively. Calculate isoelectric point of tyrosine. i want…
A:
Q: In a Chemistry lab experiment, typically, a solid chloride unknown (not the AgCl product) for…
A: The undried sample of solid chloride will consist of water , metal and chlorine in it. Since the…
Q: All of the following alkyl bromides react by SN2 substitution when treated with sodium cyanide.…
A: The correct option is option C.
Q: A piece of metal weighs 8.25 g. When a student places it into a graduated cylinder containing water,…
A: we have to calculate the density of metal
Q: 20. Which of the pairs has the member with the greater molar entropy listed first? Select all that…
A:
Q: Calculate AH-AU C(s) + 1/2 O₂ (g) ----> CO (g) A) 1.24 kJ/mol B -2.48 kJ/mol C D E -3.72 kJ/mol…
A:
Q: There are two radioactive elements, elements A and B. Element A decays into element B with a decay…
A:
Q: A 0.03 m³ tank contains 13.5 kg of air at a temperature of 200 K. Using the van de Waal's equation,…
A:
Q: How many chiral carbons are there in each of the following molecules? Type a numerical number in…
A:
Q: Which of the following contributes most substantially to rate acceleration produced by an enzyme? O…
A: The solution is given below -
Q: Which two Fischer projections represent a pair of enantiomers? CH3 CH3 CH3 HOH H- H- CI но- C₂H5 A O…
A:
Q: The following compounds have similar molecular weights. Which has the highest boiling point? O a.…
A:
Q: QUESTION 3 The enthalpy of vaporization is typically much greater than the enthalpy of fusion…
A: #3: Enthalpy of fusion is required to convert a solid into liquid and the phase changes from solid…
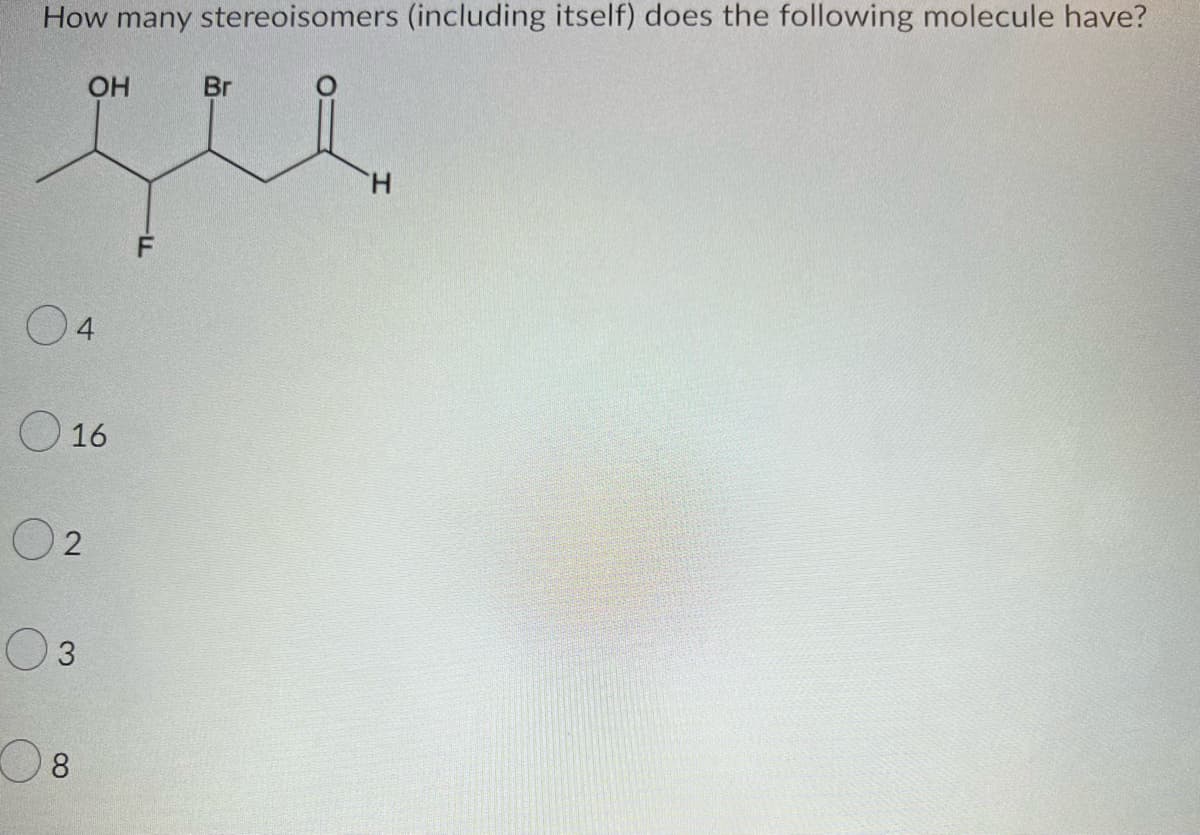
Step by step
Solved in 2 steps with 2 images

- How many asymmetric centers does the following compound have? How many stereocenters does it have?5. How many stereocenters does this compound have? How many stereoisomers are possible for this compounda. How many asymmetric centers does the following compound have? b. How many stereocenters does it have? CH3CH-ClCH CHCH3
- Does the following compound have an enantiomer?What is the maximum number of stereoisomers possible for each compound?Shown below is Streptomycin, an antibiotic medication used to treat a number of bacterial infections, including tuberculosis, plague, and endocarditis. Neomycin B has broad-spectrum antibacterial activity. Circle and label as many functional groups in these molecules as you can. Label each chiral carbon in Streptomycin. How many total stereoisomers exist for Streptomycin? Label each chiral carbon in Neomycin B. How many total stereoisomers exist for Neomycin B?
- The following compound has only one asymmetric center. Why then does it have four stereoisomers? CH3CH2CH-BrCH2CH=CHCH3Which of the following structures represent the same stereoisomer? A. only 1 and 2 B. only 1 and 3 C. only 2 and 3 D. 1, 2 and 3 Please explain.How many different possible stereoisomers could exist in the following molecule?

