Q: How do I show my work for (4 x 103) / (2 x 106)?
A: Given,
Q: Suppose you were given a glass tube with mercury in it but no scale markings, and you were given…
A: A thermometer is an instrument that is used to measure the temperature of any substance. A…
Q: Given that 1 cm = 102 m, 3 A. 1 cm³ = 1 x 102 m B. 1 cm2 = 1 x 102 m3 C. 1 cm² = 1 x 10ª m² D. 1 cm3…
A: Given : 1 cm = 10-2 m
Q: If X and Y are quantities that are related to each other by inverse proportion, what will the value…
A: If two quantities x and y are related to each other by inverse proportion Then an increase in x…
Q: A column of liquid is found to expand linearly on heating 5.42 cm for a 14.7°F rise in temperature.…
A: It is given that the liquid rises 5.42 cm by a change of temperature of 14.7 degree fahrenheit.Thus,…
Q: Which expression is correct? O All of them are correct O 1/v = Vmax/Km 1/[S] + 1/Vmax %3D O Km =…
A: Correct equation are ➡️Km = (k2 + k-1)/k1 ➡️V = Vmax [S]/(Km+[S]) is the Michaelis-Menten…
Q: 6. Assume P₁V₁ n₁T₁ that V₁ = V₂ and solve for T₁ 2 P, V₁ niT, P₂V₂ 1₂T₂ T2 7. Solve for m₁ P2 V₂…
A:
Q: All physical changes can be identified at a macroscopic level. True or False
A: Physical changes are the changes affecting the form of a chemical substance, but not its chemical…
Q: We use the . difference to separate a liquid mixture by distillation. O Melting point Flash point…
A: The temperature at which the solid substance is converted to liquid substance is known as melting…
Q: Is this correct or does the true value need the same sig figs as the estimated value?
A: Number of significant figures gives an idea on the accuracy of the measured value.
Q: Which section(s) would contain liquid water? What does the length of the horizontal line (in 2…
A: The given graph is the heating curve of water where temperature is plotted in x-axis and time is…
Q: The density answers are so close that I need help trying g to figure which substance goes with what
A: Substance 8 d= m/v = 8.5/15.6 = 0.55 g/ cm3 substance 9 d= m/v = 8.0/15.6 = 0.51…
Q: The density of water is about 1 g/cm³. Predict whether the following objects will sink or float.…
A:
Q: In Beer's Law, if the unit of b is cm, and unit of c is g/mL, what is the unit of ε? ( ε is Epsilon)…
A: Beer Lambert law provide a relationship between absorbance and concentration of absorbing species.…
Q: cube welg pe. Caleu late the densite of a metal 0.4 98 Hg length of 4.21 cm in 3/mL. 1 mL = 1 cm²…
A: Given : Mass = 0.498 kg Length = 4.21 cm
Q: Memorize that 1mL=1cm3 Convert 2000mL to cm3 Use a conversion factor of 1cm3/1mL
A: If we are having, 1mL=1 cm3
Q: A cubical box with a length of 0.1 m contains le20 02 molecules. What is the density
A: Length of cubicle box = 0.1 m Number of O2 molecules = 1 x 1020 Density = mass ÷ volume
Q: large spherical object that orbits a star
A: Planet is a large spherical object that orbits a star
Q: gth in nm of 7.88 x 1014 s1 light. your answer. Do use proper significant figures in your answer.…
A: We will calculate wavelength
Q: what is the size of the smallest increment shown on my ruler?
A: Hi, as the scale diagram is not given hence I am providing the scale and also the solution for the…
Q: Which parts of Earth are most similar to the plate with an axis angle of 80°?
A:
Q: Evaluate the following expression: 1.723 × 10² + 7.38 × 10³
A: Rule of significant figures (SF) in mathematical operations:(i) Addition and Substraction round by…
Q: CFCs break down to form chemicals that react with ozone. Is this a macroscopic or a microscopic…
A: CFCs break down to form chemicals that react with ozone. Is this a macroscopic or a microscopic…
Q: Here are two figures representing two diatomic molecules interacting:
A: Given : figures
Q: Q16. What is the mass % of A) 60%A, 10%C C) 30%A, 20%B components A, B and C at the point x? B) 10…
A: Given is a ternary phase diagram, also called the Gibb's phase triangle, consisting of three…
Q: 2. Indicate the number of sigma & pi-bom H HHH || | H-C-C-C-N-C%3D0: | | · H HH H HHH H-C-C-C-0- ..…
A: The single bonds in a compound are known as the sigma bonds. If there are double bonds in a compound…
Q: Convert 554 cm2 to in2
A: 1 inch = 2.54 cm Or, 1 cm = (1/2.54) in
Q: Direction: Complete the table of the substances below by filling out the necessary information that…
A:
Q: b. What is the radius of Neptune's moon Triton (assuming it to be spherical) if it has a mass of…
A:
Q: 13. If a 91.8 cm³ block o
A: Density (d) of a substance is equal to its total mass divided by its total volume i.e d = M/V…
Q: Using the table from image 1 D-H can you please fill in the information for the second image
A: Atoms are forming chemical bond by donating, accepting or sharing of electrons to form molecule.…
Q: There are two substances with the molecular formula C2H7N. Draw them and describe how they differ.
A:
Q: Which option is a correct representation of the missing information in Figure 8? * 1- A…
A: In an organic reaction mechanism, a curved arrow represents the transfer of electrons. In an organic…
Q: Convert the value for R, 0.068Latm mol K1, to one that has units of J mol1 K-1. ROUND ANSWER TO 2…
A:
Q: PV 4) Solve for T: =DR %3D nT 5) Solve for b: a2 + b² = c2 6) Solve for r: A = ar2
A: The given equations are as follows: 4) PVnT = R5) a2 + b2 = c2and6) A = πr2
Q: Part B 125. ft to cm Express your answer using three significant figures. L2 = %3D
A:
Q: A student finds that 24.96 g of water at 24.9 C(density=0.9971 g/cm3) is required to completly fill…
A: Given data,Mass of water to completely fill the flask=24.96gDensity of water=0.9971g/cm3Mass of…
Q: Solid matter is made up of particles that are moving faster then the particle that make up Liquids.…
A: Solid state is a state of matter in which the inter molecular force of attraction is very strong due…
Q: What is the density of water in g/cm3?
A: To find: Density of water in g/cm3 = ?? experimentally
Q: The distance from the Earth to the moon is 239,000 miles. The distance from the Earth to the moon is…
A: Distance between earth and moon = 385000 km = 385000×103 m Speed of Apollo 10 = 11080 m/s
Q: How did you get the intercept's value and slope value in 1/n
A: A question based on adsorption that is to be accomplished.
Q: 2. Convert the following Hermann-Mauguin to Schonflies notation c) 2/m b) 2/m 2/m 2/m c) 422 d)…
A:
Q: 5 Please explain these peaks
A: The structural formula and the notable functional group that exists in an unknown molecule can be…
Q: X and Y are quantities that are related to each other by inverse proportion, what will the value of…
A: Welcome to bartleby !
Q: "NH₂ over 3000 cm -1 Peak 2- and 3 d 2
A: According to the question, we need to choose the correct option.
Q: Which of the following is a (011) plane? (A) (B) (C) y A None of the above B. O OO
A:
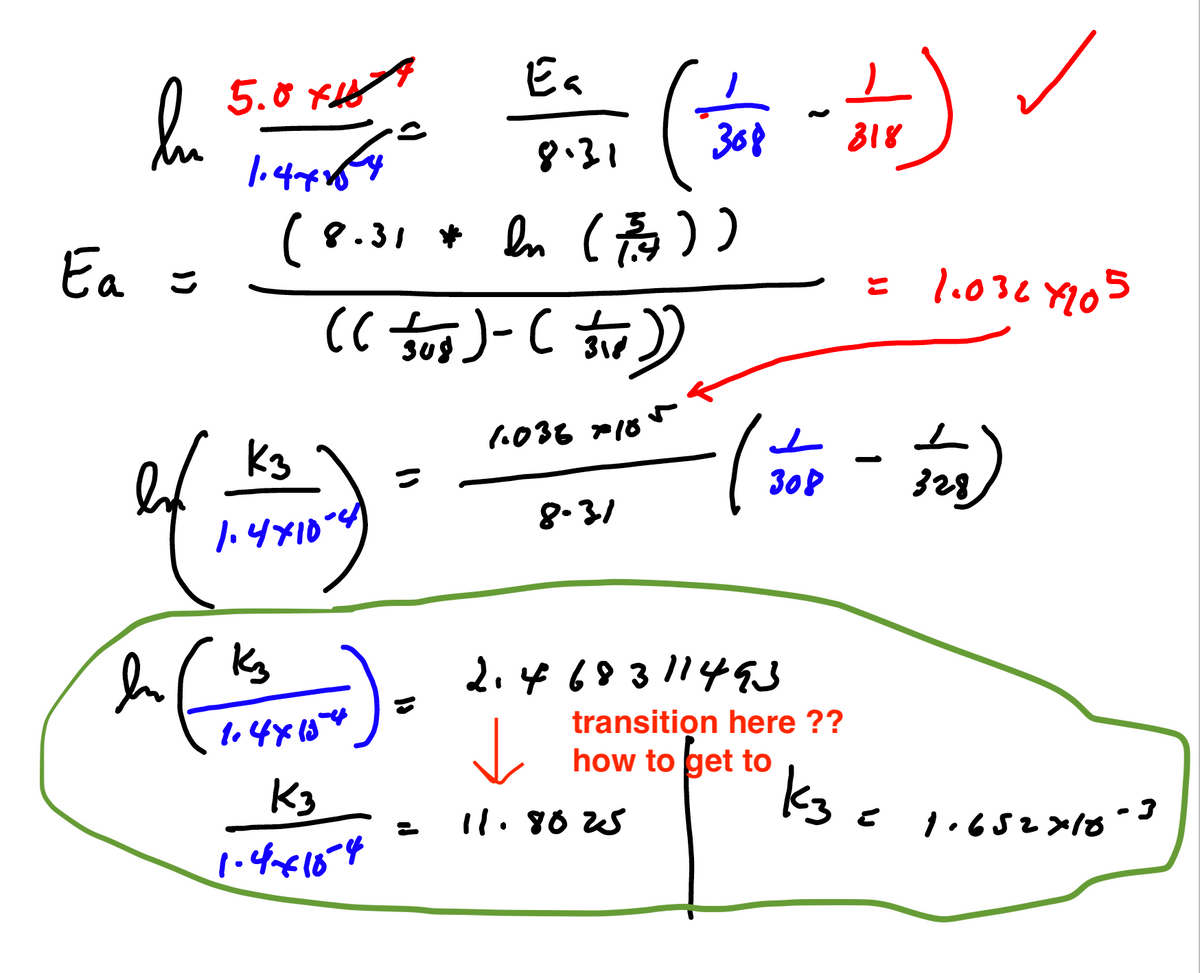
Can somebody help me with this problem by explaining the transition there at the bottom? How it went 2.468.. to 11.8025?
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Pls solve this problem and I upvote. Do not reject if it's hard for you skip it so one of the expert can do it. Thank you. I pray for your wellbeingPlease answer atleast 4 sub-parts. Thank you.An aqueous solution of sucrose (C12H22O11) is prepared by dissolving 7.4045 g in sufficient deionized water to form a 100.00 mL solution. Calculate the molarity of the solution. Do you mind showing work? I would assume that (given) gram weight (times moles present/divided by mole(s) of compound) then divided - again by compound weight will give a percentage, then divded by the .....that's where I'm a little lost. thanks
- Show all steps leading to the final answer po. Here’s a pdf file in accordance with the topic po: https://drive.google.com/file/d/1_FnDtXCrFKSol3RNWIG_9tNQ7IxgxD6t/view?usp=drivesdkCalculate the gravimetric factor of: FeO in Fe2O3 Bi2S3 in BaSO4 BaSO4 in (NH4)2SO4 Mn2O3 in Mn3O4 NOTE: Present complete solution and include the detailed computation of the molecular weight. Express your final answers up to FOUR decimal places.Calculate the amount of phycocyanin in Sample 1 in mg where A620=0.211 and A650=0.086, taking into account the dilution factor as per question 6 (100ul), and the total volume of extract as per question 4 (140ml) . Note your answer to 2 decimal places
- Please show steps and answer accordingly. Thanks! X3A.There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…There's 1 drink (and you are asked to determine the glucose concentration in the drink in the units of g/100mL. (Why these units? Well, once you have the concentrations in g/100mL you will be able to compare your values with the nutritional values given on the drink bottles’ labels). The sample of the drink was diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for you is that the dilution factor will need to be taken into consideration in your calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). You measured the concentration of their diluted drink using the spectrophotometer and their results were provided to them in the units mM (millimolar). Glucose Concentration in mM of drink =…

