Q: Consider the compound Hesperetin which belongs to the flavanone class of flavonoids. Hesperetin, in…
A:
Q: Draw the product of this reaction. Ignore inorganic byproducts. H₂/Pt
A: Alkynes are hydrogenated by the reaction with hydrogen in the presence of finely divided nickel,…
Q: Draw the major and minor product that could be formed when 1- chloro-1,3-butadiene reacts with…
A: IntroductionThe Diels-Alder reaction is a cycloaddition reaction that involves the reaction of a…
Q: 3)2-CH2 The molecular formula for both n-hexane (or simply hexane) and 2,2-dimethylbutane is C6H14.…
A: The compounds with the same molecular formula but different bond connectivity are structural…
Q: When hydrogen sulfide gas is bubbled into a solution of NaOH, the reaction forms sodium sulfide and…
A: Number of moles = Mass/molar massBalanced chemical equation:H2S + 2NaOH -----> Na2S +…
Q: 6. Sketch a graph that shows the following relationships. For each graph, state whether the…
A: The objective of the question is to identify the relationship between-Volume and Temperature of a…
Q: ion For separately excited dc motor, if the main flux is divided to four with VA remaining constant.…
A:
Q: Draw the SN2 mechanisms for reaction equation below. In addition, show the reaction energy diagram…
A: The above reaction is SN2 type substitution reaction. This reaction involves a single step mechanism…
Q: H3CC=CH₂ Br Mg ether A H₂O B
A:
Q: Draw significant resonance structures for the following compound:
A: Given
Q: Amino acids are biological compounds with the following structure, where the R group can vary. The…
A: Amino acids are important biological compound. The given Amino acid is
Q: The following reaction is electrophilic substitution: 3- [Pt(CH₂)(CI)(PMePh₂)₂] + N° [Pt(CH3) (N)…
A: Substitution reaction can be classified into two types they are electrophilic and nucleophilic…
Q: At a local pawn shop a student finds a medallion that the shop owner insists is pure platinum.…
A: We have been give the densities of different metals.We have been given the mass of the metal.We have…
Q: How many ml of 25% dextrose are needed to prepare 500 ml of 40% dextrose if you are to prepare 40%…
A: Let's denote the volume of 25% dextrose solution needed as V1 and the volume of 60% dextrose…
Q: Observe the following picture and choose the correct statement. aa aa aa-R aa aa aa aa aa aa R-aa aa…
A: Proteins are large polymers of amino acids. The structure of a protein is determined at different…
Q: These are polymorph. which one has lower melting point and better dissolution?, and which one is…
A: Melting point is defined as the temperature at which a solid starts melting and becomes liquid.it…
Q: 3. Draw an orbital overlap model of the following molecule and indicate the most reactive electron…
A: The valence bond theory (VBT) was developed to explain some properties of covalent bond especially…
Q: Draw the structure of the product(s) that are formed when C5H₁0, is treated with Br₂ and heat. + B1₂…
A: This is an free radical substitution reaction. Cyclopentane reaction with Br2 produced…
Q: Determine the product(s) of the following electrophilic addition reaction. Draw the low temperature…
A: The addition of strong acid like HBr to the conjugated diene is regioselective reaction which…
Q: t of estion BIO—H Select one: O a. 2 b. 4 O c. 3 O d. 1 Br CI How many chiral center in…
A: The objective of the question is to determine the number of chiral centers in the given molecule…
Q: 2. Identify the products of the following reaction: Br NaOEt
A: The given reaction is between bromide ion (Br-) and sodium ethoxide (NaOEt).
Q: Answering the following about the organic compound butane 1) The functional groups present on the…
A: The objective of the question is to understand the properties of the organic compound butane,…
Q: :The following data were obtained in a study of an enzyme known to follow Michaelis-Menten kinetics…
A:
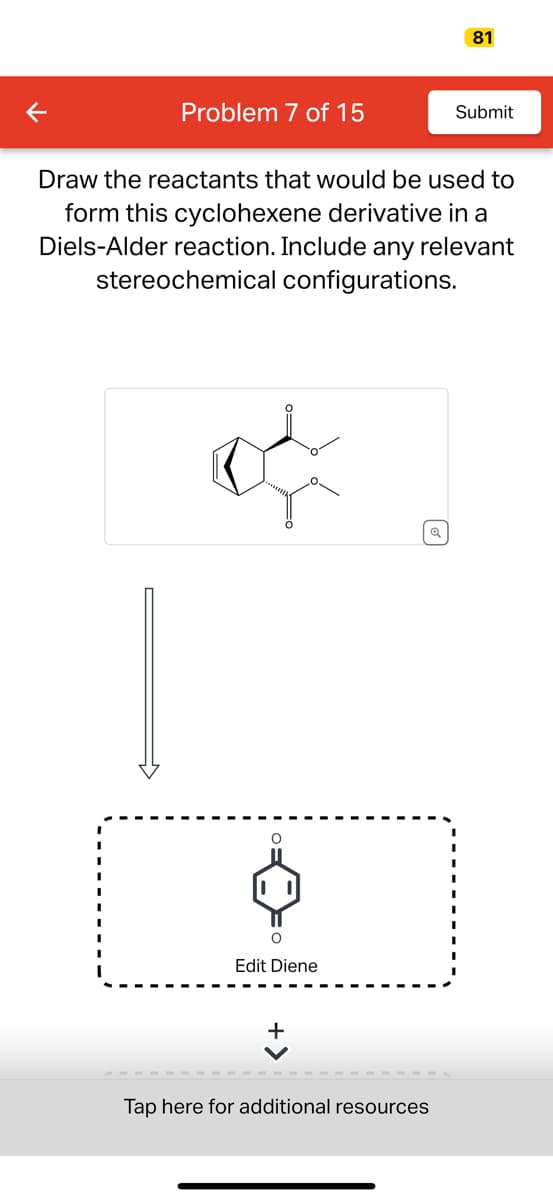
Q: 2) Draw the structure of the major product (most stable) that results when the diene shown is…
A:
Q: Draw the major and minor product that could be formed when 1-chloro-1,3- butadiene reacts with…
A: The Diels-Alder reaction is a cycloaddition reaction that involves the reaction of a conjugated…
Q: 1. Li (2 equiv), THF, -78 °C 2. Cul (0.5 equiv) 3. cyclohexenone 4. H3O+ Q
A: In the given reaction first of all there occur formation of nucleophile with help of given reagents…
Q: Give the IUPAC systematic names for each of the following. OH OH OH
A:
Q: Food dyes have very high LD50 values and they are consumed in very small quantities. Blue Dye #1 has…
A:
Q: Highlight in red each acidic location on the organic molecule at left. Highlight in blue each basic…
A: According to Bronsted-Lowry concept an acid is defined as a proton donor and base is defined as…
Q: Can you include the diene and dieneophile
A: Retro-Diels-Alder reaction is the reverse of the forward Diels-Alder reaction. The forward…
Q: How many different molecules are drawn below? 1 X Ś
A: The basic skeleton of IUPAC nomenclature is "prefix + parent + suffix".Parent is the longest carbon…
Q: Draw structural formulas for organic products A and B in the window below. -CH₂CI ***** ? Li pentane…
A: A question based on reactions of alkanes. Two successive steps of a reaction scheme are given that…
Q: If 35.92 g CH4 and 70.86 g O2 combust, how many grams of CO2 are produced? CH4 (g) + 2 O2 (g) → CO2…
A:
Q: INTERACTIVE EXAMPLE Determining Density At a local pawn shop a student finds a medallion that the…
A: We have been give the densities of different metals.We have been given the mass of the metal.We have…
Q: What is pH of a 0.0988 M solution of NaOH? Give answer rounded to two decimal places please.
A: Given,[NaOH] = 0.0988 Mwe know that NaOH is a strong base and it will dissociate completely into…
Q: Based on a grade 11 chemistry student, answer the following question based on the provided tables:…
A: The objective of the question is to estimate the toxic dose for Mary, who weighs 50 kg, for…
Q: Question 10 Listen If you have not weighed out enough solid compound on the balance, you should ...…
A: Place your scale on a smooth plane surface. Put the weigh boat on the weighing pad and switch on the…
Q: Use the following key to classify each of the elements below in its elemental form: A. Discrete…
A: Element: An element is formed by only one type of atom. All the atoms in the element are the…
Q: Briefly describe your understanding of the following terms using any typical element in the periodic…
A: The principal quantum number (n) is akin to an element like Hydrogen (H) on the periodic table and…
Q: Exactly 10.5 mL of water at 31.0 \deg C is added to a hot iron skillet. All of the water is…
A: Information about the question
Q: Experiment 1: Synthesis of a Flavone This experiment will illustrate a multistep synthetic sequence…
A: To solve these type of problems always see the reaction and mole ratio to form the compound.If mole…
Q: Predict the major organic product formed when the compound shown below undergoes reaction with…
A: The given reaction is based on nucleophilic addition reaction.
Q: Complete the following table, which lists information about the measured acid dissociation constants…
A: The relationship between the Ka ( acid dissociation constant) and pKa of the acid is given by,=>…
Q: Balance the following equation with the smallest whole number coefficients. Choose the answer that…
A: The question is based on concept of chemical reaction.We need to balance the given reaction.
Q: What is the structure for 2,2-dimerhylbutane and ethane?
A: The objective of the question is to understand the structure of 2,2-dimethylbutane and ethane. These…
Q: -1 Compound X has a molar mass of 135.12 g mol element carbon mass % 44.44% hydrogen 3.73% nitrogen…
A: Molecular formula: In a molecular formula we describe the number of atoms of all the elements…
Q: What is the major organic product of the dehydration reaction shown. OH IV H₂SO4 11 V |||
A: Dehydration of alcohol is a reaction that removes water molecules from alcohol to form alkenes. It…
Q: Give the IUPAC name for the following compound: Interactive 3D display mode Ⓡ CH3 H₂C-C- -C-C-OH H₂…
A: The full name of the compound can be determined by IUPAC rule of nomenclature are as follows –…
Q: Liontic acid is a weak acid with a pKa of 7.2. A 400-mL solution of 0.3 M liontate buffer was…
A: Given,pKa of liontic acid = 7.2Molarity of liontate buffer = 0.3 MVolume of buffer solution = 400…
Q: Be sure to answer all parts. Draw the products formed, disregarding stereoisomers, when the…
A: The question is based on organic reactions.We need to identify the product and explain its…
Step by step
Solved in 3 steps with 2 images

- From letters, C to G predict the products for the following reactions. Indicate NR if there is NO REACTION.27. Which of the following reactions gives the product shown? Select all that apply.HURRY ASAP I WILL RATE NO NEED FOR EXPLANATION WHICH OPTIN According to the given reaction below, which of the following statement(s) are true when nitrobenzene (C6H5-NO2 ) is brominated with Br2 and FeBr3? C6H5-NO2 + Br2 → I. meta-bromo nitrobenzene is formed. II. ortho and para bromo nitrobenzene are formed. III. -NO2 group is a passivating substituent and directs the ring to ortho and para positions. IV. -NO2 group is an activating substituent and directs the ring to meta position. I, IV II, III II, III, IV I II, IV
- 18. Which of the following reactions from the products shown? Choose all correct answers.From what alkenes might the following alcohols have been prepared?Compound A has the formula C10HI6. On catalytic hydrogenation over palladium, it reacts with only 1 molar equivalent of H2. Compound A also undergoes reaction with ozone, followed by zinc treatment, to yield a symmetrical diketone, B (C10H16O2). (a) How many rings does A have? (b) What are the structures of A and B? (c) Write the reactions.
- problem10.10 a I want to know this answer and mechanismUnder forcing conditions, sp2 CH bonds can be deprotonated, and sp2 chlorides can be eliminated. Draw two different mechanisms to account for the formation of the isomeric products C3H4 from the same chloride starting material.choice. b. (4 pts) Assign configurations ( R or S ) to all stereocenters in the products. vs. (1 pt) Unlike NaBH4, reductions carried out with DIBAL require quantities equimolarof DIBAL and substrate. Can you explain this difference? MinOR Help wirh part B please, please show working out
- trans-2-Butene is more stable than cis-2-butene by only 4 kJ/mol, but trans-2, 2, 5, 5-tetramethyl-3-hexene is more stable than its cis isomer by 39 kJ/mol. Explain.please help with OChem question Predict the oxidative product(s) of 2-methyl-6-octyn-2-ene after undergoing ozonolysis with O3, H2O2Show synthetic strategy to convert 1-bromo-1-methycyclohexane into 1-(2-methylpropyl)cyclohexene. Neither mechanism nor stereochemistry are required for this question.

