Can you please summarise this photo just dividing it into main points in the preparatory phase and the pay off stage.
Q: You are presented with the following clinical scenario: "A 12 month old male child presents with…
A: Flow cytometry is a technology used for rapidly analyzing population of cells or particles. It…
Q: Define enantiomers or optical isomers. Give examples
A: Introduction: The above answer explains the concept of enantiomers, also known as optical isomers,…
Q: Q.4. What are monosaccharides? Give two examples.
A: Introduction: Monosaccharides are the simplest type of carbohydrates, also known as simple sugars,…
Q: Which of the following will happen if perchloric acid is added to a neutral polysaccharide gel?…
A: Polysaccharide gels are used as sieve to separate molecules on the basis of charge and size. The…
Q: 0) Stereoisomerism and amino acids 31) Protein and nucleotide based electrophoresis and blotting…
A: Stereoisomerism is the phenomenon in which two or more molecules have the same molecular formula and…
Q: Is proton concentration higher in the intermembrane space or the matrix of the mitochondria?
A: A proton gradient is the differential in proton (H+) concentration between two locations separated…
Q: Which of the following is true about the Z-DNA helix? a) It has fewer base pairs per turn than B-DNA…
A: DNA is a double helix and can exist in 3 different three-dimensional forms:- B-DNA: the structure…
Q: 33. In bacterial cells, a) glucose-6-phosphate; ATP b) ADP; phosphoenolpyruvate c)…
A: Since you have posted multiple questions, we will provide the solutiononly to the first three…
Q: what about proteins and fats
A: Fatty acids and phospholipids are the most common building blocks of lipids and are composed of a…
Q: Which of the following amino acid cannot undergo transamination reaction? A. Alanine B. Proline C.…
A: Transamination is the reaction where amino group is removed from a amino acid (donor) and…
Q: ATP Structure H- N NH₂ N Adenine, (Nitrogenous base) N N H -H H H CH, O H Alpha Beta phosphate…
A: ATP (adenosine triphosphate) is a molecule that plays a crucial role in cellular energy transfer.…
Q: Describe how soluble, luminal ER proteins that "leak" out of the ER into the cis-Golgi are retrieved…
A: Proteins are transported between ER, Golgi and every other cellular compartment by first packing…
Q: Can you help me understand how to measure both protein A and B please
A: PAGE is performed by loading a sample containing the molecules of interest onto the gel and…
Q: A protein solution is prepared by dissolving 350 µg of protein in 200 µL of water. A 150 µL sample…
A: The four types of biological macromolecules are nucleic acids, proteins, lipids and carbohydrates.…
Q: Identify the other test used for calcium determination and discuss its principle.
A: Calcium is an essential mineral in the human body, playing a critical role in many physiological…
Q: 1. What is koji and how is it used and prepared as starter enzyme in certain production of food…
A: 1. What is koji and how is it used and prepared as starter enzyme in certain production of food…
Q: 23. is used in the first reaction of the citric acid cycle and regenerated upon completion of one…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: The partial sequence of one strand of a double-stranded DNA molecule is…
A: EcoR1 and Pst1 are types of restriction endonuclease, that have the property of cleaving the DNA at…
Q: Which of the following are important for white blood cell recognition? a) Glycoproteins b)…
A: Glycoproteins are an important type of molecule that are involved in white blood cell recognition.…
Q: 3. Draw the structure of the catalytic triad found in the active site of chymotrypsin and explain…
A: Lineweaver Burk (LB) plot has 1/v0 as Y-axis and 1/[S0] as X-axis. Competitive, Uncompetitive and…
Q: At pH 7, draw the structure of Arg-Tyr-Gln-Glu-Lys. 2. What’s the charge of this peptide at pH 12?…
A: 1. At pH 7, the Arg-Tyr-Gln-Glu-Lys peptide would have the following structure: H H H…
Q: 1. outline the principles and procedures of isoelectric focusing. 2. Explain the four levels of…
A: As per the central dogma of molecular biology, DNA contains the code that is necessary for the…
Q: discuss the difference between ‘immunoreactivity’ and ‘bioactivity’.
A: Immunoreactivity and bioactivity are two terms used in immunology and biochemistry to describe…
Q: 45. What is a term for an assembly of antenna pigments? a) thylakoid b) granum c) light-harvesting…
A: Since you have posted multiple MCQs, we will provide the solutiononly to the first three MCQs as per…
Q: What types of bonds are important for each level of protens
A: Protein structure have 4 levels. They are; primary, secondary , tertiary and quaternary.
Q: (a) Fill in the blank regarding the mechanism of chymotrypsin below. (1) Polypeptide substrate binds…
A: Serine proteases are protein cleaving enzymes that have Serine, Histidine and Aspartate (the…
Q: Which of the following involves carrying genetic information from DNA for protein synthesis? a)…
A: The molecule that carries genetic information from DNA for protein synthesis is mRNA (messenger…
Q: How about for the T form affinity sites
A: Hemoglobin is a globular protein, ie it is roughly spherical. It is a tetramer of two types of…
Q: There are multiple fates of pyruvate. If you were studying pyruvate entering the citric acid cycle,…
A: Introduction: In the field of science, there are many pathways and cycles that are involved in…
Q: 25. Alternative routes . The most common metabolic sign of mitochondrial disorders is lactic…
A: When lactic acid builds up in the bloodstream, it forms lactic acidosis. Generally, when oxygen…
Q: Chymotrypsin, trypsin, and elastase are digestive enzymes called serine proteases. The serine…
A: Aminoacids are classified based on the nature of their side chain. They are Acidic aminoacids :…
Q: 1. What are the major differences in both meals? 2. Is the turkey sub healthier when looking at the…
A: The macromolecules present in the turkey sub and burger are: Turkey Sub: 47 grams of carbohydrates,…
Q: What is the role of Sucrose in DNA isolation? A.Cell Lysis B. Maintain osmotic pressure C.…
A: DNA isolation is a process by which DNA portion in a cell is collected from the rest of the contents…
Q: 4. Enzymes with a kcat / Km ratio of about 10³ M-¹s-1 are considered to show optimal catalytic…
A: The Kcat/Km ratio is a measure of the catalytic efficiency of an enzyme. Kcat (also known as…
Q: Why are a higher number of sugar molecules displaced when sucrose concentration levels are high?
A: Sugar and sucrose are types of carbohydrates (can be monosaccharides, disaccharides,…
Q: 8. A type of gene therapy called RNA interference (RNAI) is being investigated to treat Huntington's…
A: During transcription, the DNA double helix unwinds and the RNA polymerase enzyme reads the template…
Q: you discover that myoglobin has an isoelectric point of 6.8 and chymotrypsin has an isoelectric…
A: Isoelectric focusing separates proteins based on their isoelectric point (pI), which is the pH at…
Q: 5. Consider the thermodynamics of DNA replication. What are the different thermodynamic parameters…
A: Central dogma states that the flow of information is unidirectional and irreversible, i.e.…
Q: CARBOHYDRATES CONCEPT MAP
A: A concept map is a diagrammatic representation of a topic using arrows and terms that describe that…
Q: mechanism and what we learned about acid/base chemistry, why chymotrypsin would not be able to…
A: Chymotrypsin is an enzyme that is optimized to function at a neutral pH of around 7.4, which is the…
Q: Describe the bonds stabilizing protein structure.
A: Introduction: Proteins are complex macromolecules composed of amino acids that fold into specific…
Q: As more inhibitor is added to the enzyme reaction depicted in the graph below, the shift in the…
A: Enzymes are high molecular weight proteins that catalyse biochemical reactions. They contain an…
Q: What is the ∆G naught' for a phosphoryl transfer from ATP to glycerol? The hydrolysis of…
A: The phosphoryl transfer reaction from ATP to glycerol can be represented as: ATP + glycerol → ADP +…
Q: How many ATP are produced when a 18-carbon fatty acid is completely oxidized by a cell that has a…
A: Fatty acids are carboxylic acids with a hydrocarbon chain ranging from 4 carbon to 36 carbons.…
Q: Clearly explain the location, the metabolic role and the way in which its function is regulated (if…
A: Introduction: The below answer describes the location, metabolic role, and regulation of five…
Q: How do you calculate the amount of ammonium sulfate necessary to get a 20% solution?
A: To calculate the amount of ammonium sulfate necessary to make a 20% solution, you need to use the…
Q: Q6. You measure the kinetics of an enzyme as a function of substrate concentration, first without…
A: Inhibitors are those molecules which slows down a reaction. In case of enzymes, the inhibitors bind…
Q: Q. 5. Give one example each of di-, tri- and tetra-saccharides.
A: A disaccharide is a complex carbohydrate that upon hydrolysis yields two molecules of same or…
Q: The initial rate (v0) data as a function of substrate concentration [S] for an enzyme (Enzyme1) that…
A: Enzymes kinetics - is the study of the reaction rate of enzyme catalyzed reactions. The reaction…
Q: Select all statements that are correct. Enzyme inhibitors more than 1 correct statement. prevent the…
A: An enzyme is a biocatalyst that increase the rate of chemical reaction without itself being changed…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

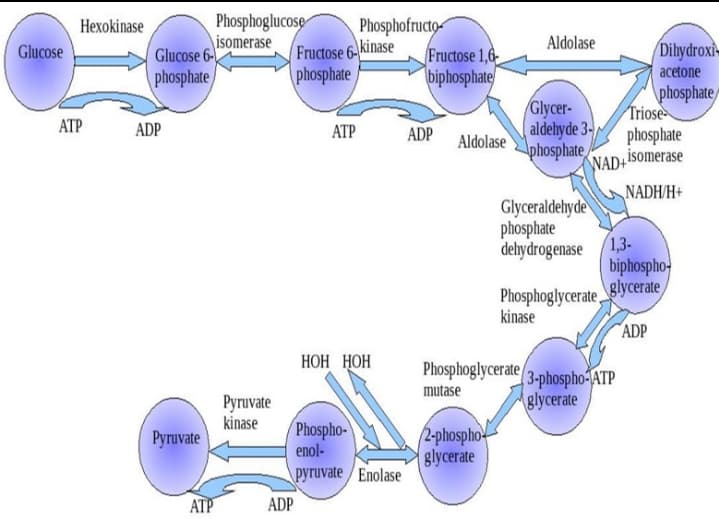
- Fructose-2,6-bisphosphate is an activator of and an inhibitor of O phosphofructokinase; hexokinase O fructose bisphosphatase; phosphofructokinase O phosphofructokinase; fructose bisphosphatase O glucose-6-phosphatase; pyruvate kinase O pyruvate kinase; phosphoenolpyruvate carboxykinase3-Phosphoglycerate is a metabolite in glycolysis. Which glycolytic enzyme generates this metabolite?How many ATP may be produced from 1 mole of the following compounds on complete oxidation using the glycerol-3-phosphate shuttle? COMPOUNDSa.) Mannoseb.) Dihydroxyacetone phosphatec.) Citrated.) Malatee.) Succinate
- Individuals with Pompe disease lack the lysosomal _______. a. Glycogen phosphorylase b. Amylo-α(1→6)-glucosidase c. α(1→4)-glucosidase d. Glucose-6-phosphataseVon Gierke’s disease is also known as glycogen storage disease type I. Patients with von Gierke’s disease lackglucose 6-phosphatase activity. Two prominent symptoms of this disorder are fasting hypoglycemia and lactic acidosis (elevated lactate levels in the blood), especially during strenuous exercise. Explain why these symptoms occur. What chemical reaction does this enzyme catalyze? Which pathways involve this enzyme? Lacking thisthe enzyme will cause impairment of which pathways?• Pls consider what pathways are affected by Von Gierke’s disease. Include in your explanation involving Cori’s cycle. can you please do not write by your hand? I mean computer if you can. thank youSelect the following enzymes that utilize a mechanism where an enediol intermediate is formed. Check all that apply: a)phosphoglucoisomerase b)triose phosphate isomerase c)aldolase d) glyceraldehyde 3-P dehydrogenase e) hexokinase
- For the pyruvate dehydrogenase complex, please identify the oxidation states of every atom and prove whether the reaction is oxidized or not Pyruvate --> Hydroxyethyl TPP B. Hydroexythl TPP --> Acetyl Lipoylysine C. Acetyl Lipoylysine --> Acetyl-CoAGlycogen synthase catalyzes glycogen synthesis. Determine whether each intermediate is associated with an “increase” or “decrease” and or “no effect” on the activity of glycogen synthase. Glucose 6-phosphate – cAMP –Phosphorylation of phosphatidylinositol yieldsa) Phosphatidylinositol 4, 5-biphosphateb) Phosphatidylinositol 3, 5-biphosphatec) Phosphatidylinositol 3, 4-biphosphated) Phosphatidylinositol 5, 6-biphosphate
- Glycolysis produces_____and_______through oxidizing_____ and ____choose from the following:GlucosePyruvateNADHNADPHATPMany components of the glycolytic pathway and the citricacid cycle are direct exit or entry points to metabolic pathways ofother substances. Indicate another pathway available to the following compounds:(a) Fructose-6-phosphate(b) Oxaloacetate(c) Glucose-6-phosphate(d) Acetyl-CoA(e) Glyceraldehyde-3-phosphate(f) -Ketoglutarate(g) Dihydroxyacetone phosphate(h) Succinyl-CoA(i) 3-Phosphoglycerate(j) Fumarate(k) Phosphoenolpyruvate(l) Citrate(m) PyruvateFor myristic acid, C 13H 27CO 2H: (a) How many molecules of acetyl CoA are formed from complete β-oxidation? (b) How many cycles of β-oxidation are needed for complete oxidation?

