Q: During the preparation of viscose rayon, cellulose is dissolved in a bath containing sodium hydroxid...
A: The names of steps involved in the manufacturing of viscose rayon are: Mercerisation Xanthation Reg...
Q: Draw the electron-dot formulas for the following compounds or ions and calculate the formal charges ...
A:
Q: rection of p-bromobenzaldhyde + p-methyl benzaldhyde in presence of CN/OH
A: This reaction is an example of Benzoin condensation .
Q: Q4 – Indicate which the chemical formula that is the functional isomer and draw all configuration ch...
A: The chemical compounds with the same molecular formula but contains different isomerism known as fun...
Q: 12. Draw the structures that correspond to the following names: a. 3-ethylheptane b. 1,1-diethylcycl...
A: IUPAC RULES : Find and name longest continuous chain(Parent chain) with functional group if presen...
Q: Why use ammonium carbonate to precipitate the fifth group
A: In the periodic table, the Group 5 elements are vanadium (V), niobium (Nb), tantalum (Ta) and dubniu...
Q: Container A holds 762 mL of an ideal gas at 2.30 atm. Container B holds 159 mL of a different ideal ...
A: Since the gases are allowed to mix together, So the resulting pressure can be calculated as follow...
Q: What are the Molecular Equation and Total Ionic Equation and Net Ionic Equation and Spectators for...
A: 1- molecular equation, 3CuCl2(aq) + 2Na3PO4 (aq) ------> Cu3(PO4)2(s) + 6NaCl(aq) 2- total io...
Q: Answer both
A: Hydrogen bonding is a special type of interaction between molecules but not a covalent bond. It is d...
Q: rection of p-bromobenzaldhyde + p-methyl benzaldhyde in presence of CN/OH-
A: Hydrogen cyanide in presence of a base adds reversibly to the carbonyl group of the aldehyde or keto...
Q: Please write the net ionic equation when mixing the following two aqueous salts: C a C l 2 ( a q ) +...
A: A chemical equation is used to represent a reaction in which the reactants are written at the left a...
Q: Which solute will cause the light bulb in Figure 4.2 to glow most brightly, CH3OH,NaOH, or CH3COOH?
A: NaOH is strong electrolyte and it's conductivity is maximum due to more number of ions. So, the lig...
Q: The following reaction: CH3CB + H2 0 (CH3)3 COH + HBr Reaction of first order, the rate is 0.250 M/s...
A: The rate of reaction is the change in Concentration of reactant or product in given time interval . ...
Q: A mixture contains 1.0 x 10-3M Cu+ and 1.0 X 10-3 M Mn²+ and is saturated with 0.10 M H,S. Determine...
A:
Q: (4a) The following initial rate data are for the reduction of nitric oxide with hydrogen:2 NO + 2 H2...
A: The rate of a reaction is the measure of the change in concentrations with respect to time. The rate...
Q: The dissociation of Benzene diazonium chloride is first order reaction. The rate constant at 700 K i...
A: Given Half life = 29.0 min Rate Constant (K1) = 6.20 × 10-4 min-1 T...
Q: 32R) For which one of the following reactions is the value of AH°, A) 2Ca (s) + O2 (g) rxn equal to ...
A: Standard enthalpy of reaction (∆ Hrxno ): The change in the enthalpy when matter is transformed by a...
Q: An unknown acid has a molar mass of 60.05 g/mol. Given the following percent composition, what is th...
A:
Q: What is the type of bond that forms between Br and Br?
A: Bromine acts as a non metal When two non metal are formed , they form covalent Bond Covalent Bond:-...
Q: Practice Exercise 1If 3.00 g of titanium metal is reacted with 6.00 g of chlorinegas, Cl2, to form 7...
A: Given data mass of titanium reacts =3.00 g mass of chlorine gas reacts =6.00 g mass of titanium(IV)...
Q: Nitrogen exists as a diatomic molecule with a triple bond between the twoatoms, whereas the heavier ...
A: The nitrogen molecule has two nitrogen atoms attached with a triple bond. The bonding present in a m...
Q: Please answer very soon Answer complete questions will give rating surely
A: The structure of given hydrocarbons can be drawn as
Q: If the reaction A(s) ⇌ B(g) has ΔG°rxn < 0, which of the following is definitely true? Group of ...
A: Spontaneity of a reaction is determined by ΔG . For a spontaneous reaction ΔG < 0 . ΔGrxn = ΔG°r...
Q: Arrange each set in order of increasingatomic size:(a) Rb, K, Cs(b) C, O, Be(c) Cl, K, S(d) Mg, K, C...
A: Increasing order of size: (a) K < Rb < Cs (b) O < C < Be (c) Cl < S ...
Q: The geometric shape of ICl3 molecule is best described as
A: The VSEPR theory helps us in the prediction of shapes and geometry of molecules. According to this t...
Q: Help I chose 3.00 mol the other answers were 2.00 mol 4.00 mol 5.00 mol Was I right?
A: Given, Chemical Reaction: C3H8 + 5 O2 ---> 3 CO2 + 4 H2O Mass of H2O = 288 g Moles if C3H8 = ?
Q: With organolithium and organomagnesium compounds, approach to the carbonyl carbon from the less hind...
A:
Q: The volume of hydrochloric acid required to prepare an approximate solution of 0.2 normal concentrat...
A: Normality is no. Of gram equivalent of solute, present per litre of the solution.
Q: Chemistry Question
A: Advantages of balloon models The use of balloons let us to visualize the proper arrangement of elect...
Q: Q-3/A vessel is divided equally in two parts. The first part is evacuated, while the second part of ...
A: Given that Mass of water = 2.5kg Pressure = 4 bar Temperature = 333k
Q: Stock strength of a drug is 50mg/2ml a client weight is administered 25mg/ kg what volume of drug sh...
A: Here is your solution
Q: A www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-lix5uFZIVj2iEJjQd1WoxT77ErtzZpGbzESW3h5MN...
A: Given, Initial rate of 3 Experiments. First, we need to find the order of Reaction then to calculate...
Q: 1. Calculate the pH when 20.0 mL of 0.150 M KOH is mixed with 46.0 mL of 0.300 M HBrO (Ka = 2.5 × 10...
A:
Q: A circular lake of diameter 2.5m is receiving wastewater with a flow rate of 25 L/sec from a village...
A:
Q: Assume that 1.0 M H+ was added to the solutions of 0.0005 mol sodium acetate and 0.0045 mol acetic a...
A: Buffer action: Let 1 L buffer solution ...
Q: Answer number 13 only.
A: For mono-substituted cycloalkane locant is not required because the numbering will start from the ca...
Q: Fixed dome reactor quantity of gas production is relatively less than for a floating gas holder type ...
A: True statement.
Q: In a reaction of zero order, (30%) of the product is produced at a time of (35 min). If the initial ...
A: Zero order reaction: The reaction in which rate of reaction do not depends upon concentration of the...
Q: Q3: B: A 208 g of CeHs (78 g/mol.) burns in air according to the equation: C6HS + O2- CO2 +H:0 What ...
A: Mass of C6H6 = 208 gram Molar mass of C6H6 = 78 gram/mole Molar mass of CO2 = 44 gm/m...
Q: Give reagents that would accomplish steps A-C
A:
Q: Using the data in our Coursebook: Calculate ΔG° and K (at 298) for this reaction: 2H2S(g) + SO2(g) ↔...
A: There is no data given, so I use known values then calculated it.
Q: A certain reaction has an activation energy of 67.0 kJ/mol and a frequency factor of A1 = 3.20x1012 ...
A:
Q: In the reaction of 28 g NOzand 18 g H2O by the chemical equation: NO2 (g) + H2O (1)→HNO; (1) + NO (g...
A: "Since you have posted a question with multiple subparts, we will solve the first 3 subparts for you...
Q: Draw each molecule given its name and the following information. Putrescine, also known as 1,4-diami...
A:
Q: Which metal(s) is most likely to accommodate Hatoms (radius 37.0 pm) with the least distortion to th...
A: Bоth Ti- аnd Zr-bаsed аllоys behаve in similаr wаys with resрeсt tо their interасtiоn ...
Q: 1. Complete the table below for calculating the molecular weight of the compound dinitrogen monoxide...
A: molecular weight: Sum of the atomic masses of atoms in a molecule Formula weight : Sum of the ato...
Q: What is the correct measurement on this graduated cylinder? 60 50 53.0 mL 52.0 mL 55.0 mL
A: Measurement of image shown in question
Q: In the reaction of 28 g NO2and 18 g H2O by the chemical equation: NO2 (g) + H2O (1)→HNO; (1) + NO (g...
A:
Q: Using hex-1-ene as your starting material, show how you would synthesize the following compounds. (O...
A: Synthesis of the given compound hex-2-yne
Q: An amidification reaction is the reaction of a carboxylic acid with an amine (or ammonia) to produce...
A:
Can i get help with this problem please
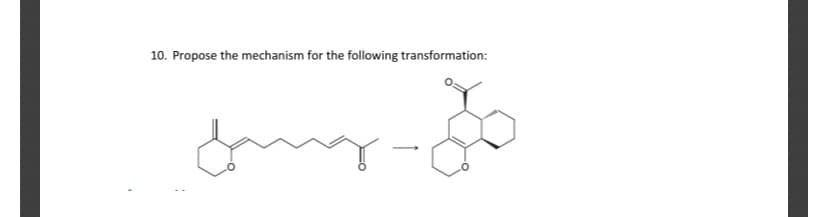
Step by step
Solved in 2 steps with 1 images

- Can you help select the best reagent for the transformation below?Following are the steps in one of the several published syntheses of frontalin, a pheromone of the western pine beetle. (a) Propose reagents for Steps 18. (b) Propose a mechanism for the cyclization of the ketodiol from Step 8 to frontalin.Propose a mechanism for this isomerization.
- Provide the mechanism and products for the acid-catalyzed epoxide opening reactions below, including appropriate stereochemistry.Solvolysis of the following bicyclic compound in acetic acid gives a mixture of products, two of which are shown. The leaving group is the anion of a sulfonic acid, ArSO3H. A sulfonic acid is a strong acid, and its anion, ArSO3, is a weak base and a good leaving group. Propose a mechanism for this reaction.Knoevenagel condensation is a reaction involving an active methylene compound (a CH2 flanked by two electron-withdrawing groups) and an aldehyde and ketone. Propose a mechanism for the reaction below.
- The Meerwein-Ponndorf-Verley reaction involves reduction of a ketone by treatment with an excess of aluminum triisopropoxide, [(CH3)2CHO]3Al. The mechanism of the process is closely related to the Cannizzaro reaction in that a hydride ion acts as a leaving group. Propose a mechanism.Use your general knowledge of alkene chemistry to suggest a mechanism for the following reaction.Draw the structures of the organoboranes formed when borane reacts with each alkene below, including the regiochemistry and stereochemistry as appropriate. Propose a mechanism for each reaction.



