Q: Hz o h. xs acetic acid + sodium hydroxide in water Na 04 CH3- - OH HzO Please Do these 2 Question…
A: When there is a possibility of acid base reaction, acid-base reaction occurs primary. The reaction…
Q: QUESTION 1 For the aldehyde shown below, which çarbon İs commonly referred to as the "a-carbon"? O…
A: Aldehydes and ketones have alpha hydrogen.
Q: Write all resonance structures H₂N - Due to resonance structure(s); is the amine more or less basic,…
A: Let us first draw the structures and then solve the questions.
Q: Rank the following compounds from strongest acid to weakest acid
A: The presence of electron withdrawing group in parent acid increases its acidity. The reason is that…
Q: What is the conjugate acid of HPO4 - 2? A. H3PO4 B. PO4 -3 C. HPO4 -2 D. H2PO4-
A: Applying concept of conjugate acid and base.
Q: 1. Acidity CI COOH COOH HO CI 1 2. Basicity NH2 NH2 NH2 NH2 CH3 CH3 H3C H3C 3. Stability 2. 4.
A:
Q: Which of the following is a diprotic acid? HCl H2SO4 CH3COOH H3PO4
A: Which of the following is a diprotic acid? HCl H2SO4 CH3COOH H3PO4
Q: H2AsO4 + NH3 ---> NH4+ + HAsO4- What is the Bronsted acid in the reaction above? a. H2AsO4 b. NH 3…
A: According to the Bronsted Lowry concept : Acid is the substance that can donate protons.
Q: What is the conjugate acid of NH2- ? A. NH3 B. HCl C. NH4+ D. NH2- E. N3-
A: The conjugate acid of NH2- is: Acid + Base ⇌ Conjugate Base + Conjugate Acid(known as conjugate…
Q: Rank the given compounds based on their relative acidities. strongest acid HF HC=CH H,C=CH, NH,…
A: The question is related to finding out the acidity of given molecules. Some points to remember 1.…
Q: ank the indicated atoms from the most (1) to the least (3) basic. ONH NH2 B A
A: We have to identify the order of basicity. More unstable the anion is , more it will be basic.
Q: 1. KMNO4, OH- (warm) 2. H3O* Select to Draw
A:
Q: Which is the strongest acid? В. CI 0 FO | CH;CHCH,COH CH;CH,CHCOH CH;CH¿CHČOH CHCHC-COH F CI
A: We are having four organic compound that are acidic in nature, we have to predict the most acidic…
Q: rrange the follwoing in order of decreasing basicity. OH-, CH3COOH, CH3CH2-, NH2-, NH3, Cl-
A: Basicity: Tendency to donate electron pair. Greater the tendency to donate electron pair, greater…
Q: In HNO3 + H2O —> H3O+ + NO3- , who is the Bronsted base?
A:
Q: redict the conjugate acid of C₅H₅N.
A: According to bronsted- lowry acid theory, acid is defined as that species which donate H+ and base…
Q: Which hydrogen in pseudoephedrine, the nasal decongestant in the commercial medication Sudafed, is…
A:
Q: CaCl2 (aq) + Na2CO3 (aq) --> CaCO3 (s) + HCl (aq) --> CuO (s) + HCl (aq) -->
A: CaCl2 (aq) + Na2CO3 (aq) -----> CaCO3 (s) + 2 NaCl (aq) CaCO3 (s) + 2 HCl (aq) ----->…
Q: Rank the compound of the attached group from strongest acid to weakest acid:
A: Lone-pair anions prefer to stay in orbitals with more s character than p character because s…
Q: Rank the acidity of the following from 1 (MOST) to 4 (LEAST). H N- -CH3 ČH3 a. H- b. -s-CH3 H-…
A: The acids given are,
Q: | Rank the following compounds from strongest acid to weakest acid. `OH F3C *OH A D
A: Carboxylic acids and ketones both belong to the class of carbonyl group. But carboxylic acids are…
Q: In acetic acid, CH3COOH, the OH hydrogen is more acidic than the CH3 hydrogens. Explain
A: Arrhenius gave the basic definition of an acid or base. A substance which loses a proton is known an…
Q: Review opics) Arrange the following carboxylic acids in order of acidity. CICH,-COOH ICH,-COOH FCH,…
A: The strongest acid is the acid in which electronegative atom is attached. Here there are 4 acids,…
Q: Arrange the given compounds based on their relative Brønsted acidities. Strongest Brønsted acid H-SH…
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: H A В
A: Carbonyl compounds are those functional groups that contains (C=O) bond. The carbon adjacent to…
Q: Which acid is stronger, propanoate or lactic acid, and why? lactic acid, because the Ka of lactic…
A: Ka is the equilibrium constant for the dissociation of an acid. The higher the Ka value, the…
Q: Rank the compound of the attached group from strongest acid to weakest acid:
A: The acidity depends on the stability of conjugate base formed after release of proton. Higher the…
Q: Below is a compound in its neutral from, draw the form that would predominate at pH 7.4 H N. хонг
A:
Q: но но NO2 `CH3 1 2 3 HO но но N= ČH3 6
A: The acid and base are defined on the basis of hydrogen ion acceptor or donor. If a compound accepts…
Q: Rank the compounds in order of decreasing acidity. Most acidic Least acidic Answer Bank BRCH, CO,H…
A:
Q: Strongest base H3C. 3
A:
Q: 3. Which of the following is the strongest acid? Why? A F3C F3C OH F3C B D H3C H3C F3C OH F3C H3C…
A:
Q: Draw each molecule given its name and the following information. (a) Nitroglycerin, also known as…
A: Nitroglycerin is also known as 1,2,3-trinitroxyglycerin and is a compound which is used in dynamites…
Q: Which of the following is a strong acid? Select one: a. NH3 b. All of these c. NaOH d. CH3CO2H e.…
A: NH3 base NaOH is base CH3COOH is weak acid HNO3 is Strong acid
Q: Rank the following in order of increasing basicity. The first in your list should be the least…
A: OH- is a conjugate base of H2O. F- is a conjugate base of HF. Cl- is a conjugate base of HCl.
Q: me the following two acids in both delta as well as omega menclature: COOH A. 1. A- 2. w- e and B.…
A: Welcome to bartleby ! We have to give naming for delta as well as omega for these fatty acids
Q: Which of the following is a triprotic acid? a) H2SO3; b) HC2H3O2; c)HBrO3; d) CH3Cl; e) H3P O4
A:
Q: Rank the following compounds from least acidic to most. Rank from least acidic to most acidic. To…
A: We know that terminal alkynes are most acidic and middle alkynes are least acidic and acidity of…
Q: Question 3: Which of the following is the most acidic? Explain why. a) CH H3C b) H3C H. a bm minrbom…
A: Alkyne has general formula CnH2n-2. Example: Propyne Alkane has general formula CnH2n. Example:…
Q: Be sure to answer all parts. Select the three most acidic hydrogen atoms in lactic acid,…
A: Given , Lactic acid : OH O | ||H3C-CH-C-OH
Q: Question 1 Which of the following is the strongest acid? a. CH3CH3 b. CH3NH2 c. CH3OH d. CH3F a O b…
A: Ans. Option c. CH3OH
Q: Is NH2 stronger or weaker base than OH-? explain why?
A: Answer: NH2 - is stronger base than OH- Explanation: Proton donar is Bronsted acid . Weak acid…
Q: The boiling points of a range of organic compounds (A-D) of similar Mol. Wt.s are given below. OH…
A: Four questions based on intermolecular force of interaction, which are to be accomplished.
Q: 1.) H-B- 2.) H2O2, NaOH (xs) a) b.) d)
A:
Q: Which is a stronger base: 1. F-NH2 or Cl-NH2 2. CH3-NH2 or Cl-NH2
A: The bases given are, 1) F-NH2 or Cl-NH2 2) CH3-NH2 or Cl-NH2
Q: Q.20 SINGLE CHOICE CH3 I H₂O/H+ H₂C O 18 CH3 Where A and B are, respectively, A O 18 H₂C and Me,C-OH…
A: ->Acidic hydrolysis of ester give acid and Alcohol as product .
Q: Which of the following is not an acid A) NH3 B) HC2H302 C) HF D) HCIO
A: Acids have different definations: Lewis acid : one which accepts lone pair. Bronsted acid : one…
Q: Write “most" under the most basic compound. Write “least" under the least basic compound. Locts NHz…
A:
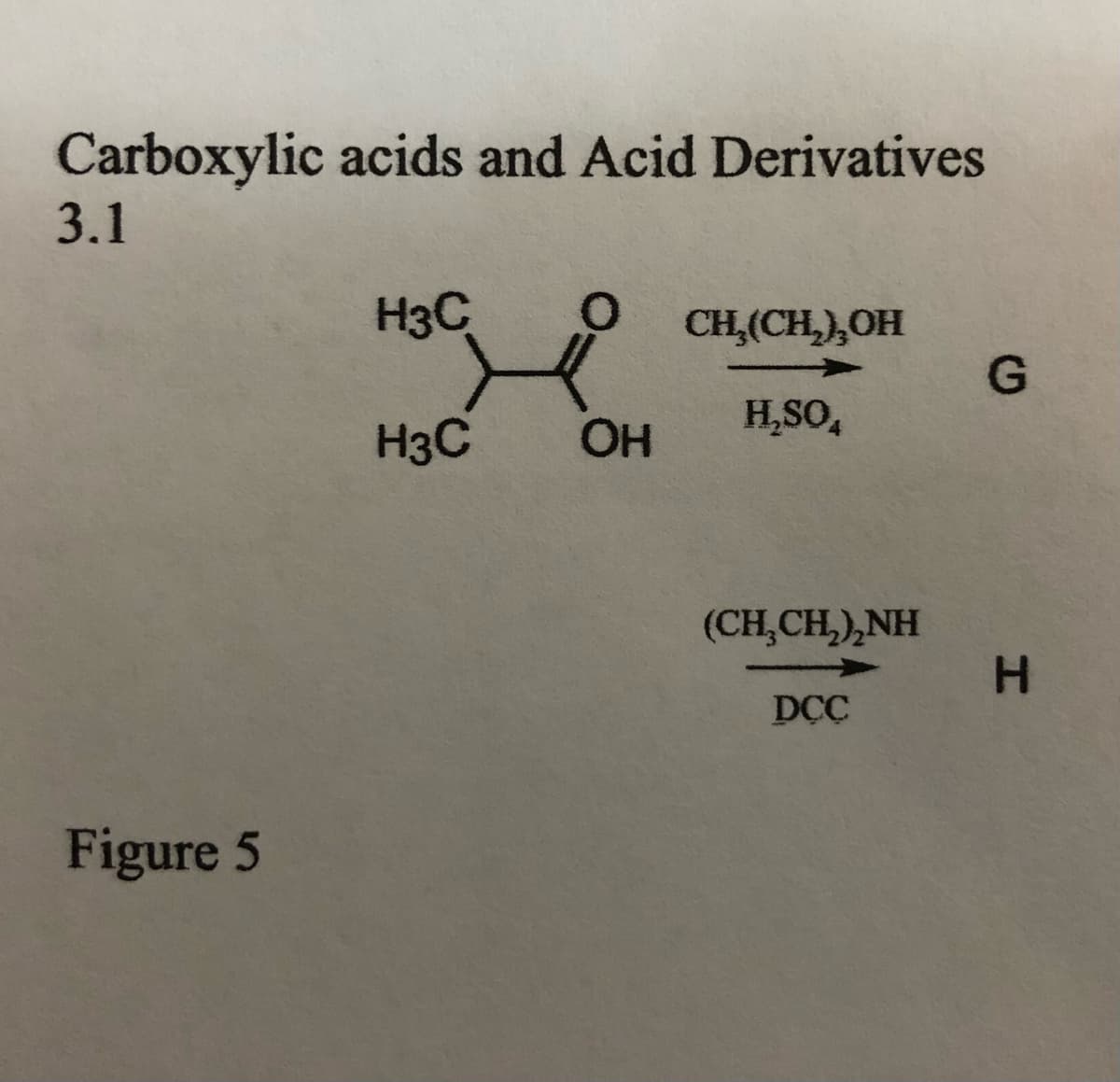
Step by step
Solved in 3 steps with 3 images


