Q: The following carbohydrate tests have been performed on a sample containing an unknown sugar. The…
A: Ans. From the given data above, the results are as follow: Characteristic Unknown sugar(Answer…
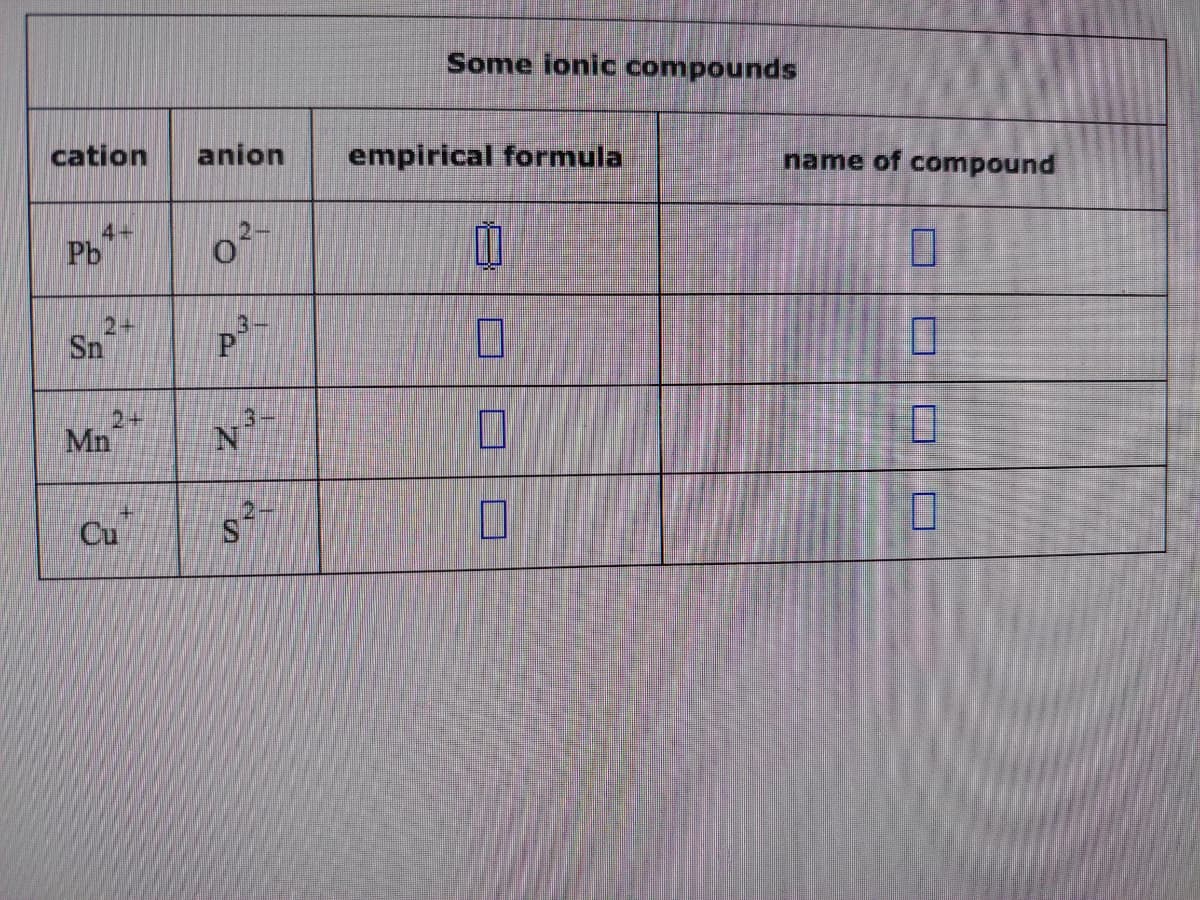
Q: Fill in the name and empirical formula of each ionic compound that could be formed from the ions in…
A: Write name and empirical formula of the given ions---
Q: how to write the enthalpy cycle?
A: The previous answer has shown all the necessary calculations and enthalpy changes. In case you want…
Q: Provide the organic products of the following and also show the mechanism involved in (a)? (1) Ph3P…
A: Dear student,the given question is a multiple question, hence I solved first question according to…
Q: There are two substances with the molecular formula C2H7N. Draw them and describe how they differ.
A:
Q: 7 # 3 G 8 4 1 mole CH₁₂0= 1 mole Fe₂(Cro), = g Fe, (CrO₂), -9 C₂H,₂06 10. Avogadro's constant was…
A:
Q: Rank the following elements in terms of increasing atomic radius and ionization energy: sulfur,…
A: In this question, we will arranged all the elements in increasing order of their atomic radius and…
Q: Avogadro's constant was not actually covered by Avogadro. Who actually calculated ogadro's constant…
A:
Q: Consider the following two reactions: A →→ 2Β ΔΗ° rxn = 456.7 kJ/mol A → C ΔΗ° rxn -22.1 kJ/mol…
A: The reactions given are, A→2B (∆Horxn=456.7kJ/mol)----i)A→C (∆Horxn=-22.1kJ/mol)----ii) As enthalpy…
Q: between the compounds or not. (i) Compounds A and B, using UV-Vis spectroscopy with a detection…
A:
Q: packed columns the stationary phase is: (a) packed in the column (b) coated inside the column (c) a…
A:
Q: Barium-122 has a half-life of 2 minutes. If 10.0 g of Ba-122 are produced in a nuclear reactor how…
A: Radioactive decay follows the first-order kinetics. The half-life of the first-order reaction does…
Q: For the molecule shown below, provide the names for all of the indicated functional groups: ger NH₂,…
A: We have find out the names of indicated functional groups in the given molecule.
Q: CH₂ Br a. 98-15 and 2 4 3 Br CH₂ C1- C2- C3- C4- Related- b. Me 1 Me OH and Et OH C1- C2- Related-…
A: Here we have to determine R and S configurations of chiral centres and relationships between the…
Q: 3. Consider the following reaction where Kc = 3900 at 2000 K 2SO2(g) + O2(g) = 250³(g) a Write the…
A:
Q: The theoretical yield of a reaction is the amount of product obtained if the limiting reactant is…
A:
Q: Calculate the mass defect, Dm, and the corresponding energy change, ΔE (in joules) per mole of…
A: Given that - Protactinium-231, Pa-231 It has Atomic Number of 91, thus Number of protons are 91…
Q: For a mixture of 40% nitrogen, 35% oxygen, and 25% carbon dioxide on a molar basis, what is the…
A:
Q: Which of the following can form hydrogen bonds in a pure sample? A) CH₃F B) CH₂F₂ C) HF D) HBr
A: Given compounds are : A) CH₃F B) CH₂F₂ C) HF D) HBr Which of the following can form hydrogen…
Q: What volume of water must be added to 10 .0 mL of 12.0 M HCl to make the final concentration of the…
A: Given data :
Q: 1.2 2(1) In the contact process: 2SO₂(g) + O₂(g)2SO3(g) AH = -197 kJ mol-¹ Predict the position of…
A:
Q: In a molecular collection, either iomic or covalent, the Oxidation number of a central atom has to…
A: Belong to redox reaction that is oxidation number. We calculate oxidation State or number by 2 ways.…
Q: Complete the following diagram for the carbon molecule (C2): 2p 2s 1s 30 1g 30 1 200 20 100⁰ 10g 2p…
A:
Q: Q4/ Six 25 ml volumetric flasks containing 2, 4,6, 8 and 10 ml of a standard solution containing 20…
A: We firstly need to determine the concentration of NiCl2 in each flask and then plot a calibration…
Q: Which of the following is/are characteristic of silica as stationary phase in chromatography? O It…
A:
Q: What is the relationship between the following two molecules? НО -Н ОН ОН -ОН НО H н-он ОН Н -ОН O…
A: We have to find the relationship between these two molecules.
Q: 1. What is the specific gravity of N2 * at 80°F and 745 mm Hg compared to air at 80°F and 745 mm Hg?
A:
Q: 6. Calculate the mass of M₂X that will dissolve in 250 mL of water at 273 K. the molar mass of M₂X…
A:
Q: Methanol is a highly toxic compound that can cause permanent damage in the optic nerve, leading to…
A: Here we have to predict the inter-molecular forces of attraction present in methanol.
Q: Phosphorous pentachloride decomposes according to the reaction PC15 (g) = PC13(g) + Cl₂ (g) A 15.7 g…
A: Here we have to determine the equilibrium constant of the following given reactions.
Q: When a 0.01 M KCl solution was filled into a conductivity cell at 25°C and its resistance was…
A:
Q: Consider the following reflux set up for the preparation of unknown substance "P". If you add…
A: Question 67) ans. Option B- Soap Question 68) ans. Option A - Colloidal particles can scatter…
Q: Which of the following reaction conditions can be used to synthesize an ester (RCOOR)? O A…
A:
Q: 1 Label the diagram below, identify the anode, cathode, direction of electron flow, and direction of…
A: This type of cell is called concentration cell in which both the anode and cathode are made up of…
Q: The galvanic cell described below has potential (E) of 0.23 V at 298 K. The rate constant for the…
A:
Q: Which of the following separation methods can be used to separate stones from water
A: Centrifugation: This is a method of molecule separation which having different densities by spinning…
Q: T 2 HCI + 2 456 mL 18.00 M 100% Ca(OH)₂ 个工 I 2 Mole ratio Volume, m Molarity Excess moles FW, g/mol…
A:
Q: Consider an ionic compound, MX3, composed of generic metal M and generic gaseous halogen X.…
A:
Q: A 10-ml sample of lake water was obtained and a titration was performed on it to determine the…
A:
Q: From the following structures, select a reasonable resonance contributing structure for the molecule…
A:
Q: A volume of 115 mL of H₂O is initially at room temperature (22.00 °C). A chilled steel rod at 2.00…
A:
Q: Is soap or detergent) was more acidic, Why? Is soap or detergent was able to dissolve oil, Why? Does…
A: Answer - According to the question - Detergents are more acidic because ,soap is always basic.…
Q: Q2: Indicate whether the following statements are true or false, correct the false and give a proper…
A: Q2: Indicate whether the following statements are true or false, correct the false and give a proper…
Q: A mixture of 0.592 M H₂O, 0.480 M Cl₂O, and 0.747 M HClO are enclosed in a vessel at 25 °C. H₂O(g) +…
A: Given, H2O (g) + Cl2O (g) ⇌ 2HOCl Kc = 0.0900 at 25 ℃ [H2O] = 0.592 M,…
Q: I had having trouble understanding the TLC labs. Could someone help explain how to tell which…
A:
Q: 0.800g of X combines with 0.200g of oxygen to give X2O5. Find the atomic mass of X. A)120 B)140…
A: Given data 0.800g of X 0.200g of oxygen Give X2O5
Q: The units for the answer is KJ/mol. But this answer says joules. why is that?
A: Previous answer is incorrect.
Q: 16. In this chromatographic technique the mobile should consists of buffer solution or with known…
A: The mobile phase of Ion Exchange Chromatography (IEC) is usually a buffer solution or a pH known…
Q: your choice. CH₂ NH CH₂ H₂C-Ö H H₂C-N H₂C-6: + H₂C-N: CH₂ CH₂ The forward reaction is favored…
A:
Q: Molybdenum (Mo) is element 42 on the periodic table. A sample contains 7.31x1022 atoms of Mo.…
A:
Step by step
Solved in 6 steps

- The electronic configuration of 15 X 3- ion is a. [Ne]3s23p2 b. [Ne]3s2 c. [Ne]3s23p6 d. [Ne]3s23p1Given is teh mass spectogram of an unknown compoound. This unstable cyclic compund is a colorless liquid with a sharp odor and reacts with light and air with long exposure. What is the molar mass of the target mecule? What is the Molecular formula of the target molecule? What IHD /U ? What could be the description of the structure? Give the name of the structure.Show how to use the microanalysis data to determine the number of cod (C8H12) ligands per Pd. C, 33.65%; H, 4.24%; N, 0.00%; Pd, 37.27%.
- Select one: True or False 1-The mesomeric form of succimer is the active chelating form that is used to chelate trivalent metals through its COOH groups. 2-Anions with large radius particularly those having less than two negative charge are more easily polarized by the small size cations while those of ≥1 charge like sulfide S-2 and selenium Se-2 are much harder to be polarized 3-Covalent bond is encountered in case of NaCl and KBr while ionic bond is encountered with BeCl2 and BF3 4- Besides providing the largest suitable number of valance orbitals for covalent bond formation it can determine the final shape of the molecule for example Sp hybridization leads to a diagonal planner molecule, SP2 hybridization while Sp3 hybridization leads a pyramidal (tetrahydral shape) 5-About the underlying compound ( in the images ) , it it’s a calcium complex of EDTA used to treat metal toxicities like pulmbism in order to avoid hypocalcaemia.Calculate the energy(kJ)whenphotons areejected from 3molesofhydrogen atomsupon an electron transition from n = 4to n = 2. ∆?=−??(1??2−1??How is CCl2F2 polar given that is tetrahedral( highschool level explanation pls)?
- What are the redcuible representation for C4V of [MnI(CO4)] and for C2v of IF3?Which of the following species: C6H5Cl ; Cl2 ; FeCl3 ; (CH3)3C+; HCl ; C6H6 ; CH3+ ; C6H5CH3 ; has or can display the following properties:You can only list a specie once (v) electron withdrawing group : ……………………………………………(vi) electron donating group : ………………………………………………..Use resonance forms to show that the dipolar sigma complex shown in the sulfonationof benzene has its positive charge delocalized over three carbon atoms and its negativecharge delocalized over three oxygen atoms.

