Q: Water Molecule ii. Shown below is a Cu2+ ion. Draw at least three water molecules as they wouid be a...
A: Here we have to draw the diagram in which Cu+2 ion will be surrounded by atleast three water molecul...
Q: Correct The balanced equation is: CH4 (g) + 2 O2 (g) CO2 (g) + 2 H2O(g) c What mass of O2, in grams,...
A: Number of moles is calculated by dividing the given mass with molecular mass.
Q: HA weak acid has (K = 0.0056), a concentration of 0.480 M. what is pH of the acid
A: Weak acid dissociate less in solution
Q: Give the condensed formula of the following alkane. Ex: CH3CH(CH3)CH2CH2CH(CH3)2 1. 2,3-Dimethylpent...
A: The condensed formula is one of the ways to express the structural formula of organic compounds. To ...
Q: of the reducing agent and the formula of the oxidizing agent. redox reaction? yes no 2Fe(s) + 3NICI,...
A: Redox reaction : It is also known as oxidation-reduction reaction
Q: 6. Provide the complete mechanism (arrow, lone pairs, formal charges) for the following transformati...
A: Protonation of alcoholic oxygen Loss of leaving group Carbocation rearrangement for the formation ...
Q: Aspirin, CsH4(0COCH3)CO2H, is produced by the reaction of salicylic acid, C6H4 (OH)CO2H, and acetic ...
A:
Q: OH CrO H,SO, ОН CH3 f) H3C- Br2 -OCH3- FeBr, g) CH, NBS Na*O-t-Bu A h)
A:
Q: Question 5 of 21> An aqueous solution at 25 C is 13.9% HNO, by mass and has a density of 1.053 g/ml....
A:
Q: он CH, `CH, H,C
A: Friedel crafts acylation and clemmenson reduction and reduction with LiAlH4 can be used to prepare t...
Q: 5. 8. 9. 10 11 12 13 An analytical chemist weighs out 0.033 g of an unknown monoprotic acid into a 2...
A:
Q: 3. Provide a mechanism for the following: acetyl CaA COASH co a. b.
A: 1. In presence of acid, esterification occurs when carboxylic acid and alcohol reacts. 2. In presen...
Q: A 25mg/L solution is prepared by diluting ul of a 50g/L solution into 10ml of water.
A: To determine the concentration of diluted solution, the following equation is used. M1V1 = M2V2 On r...
Q: 1) Write the balanced equilibrium reaction for the dissolution of barium nitrate in a saturated solu...
A: 1. Sol :- Given compound is : Ba(NO3)2 1) Write the balanced equilibrium reaction for the dissolut...
Q: A chemistry student weighs out 0.0977 g of phosphoric acid (H,PO,), a triprotic acid, into a 250. mL...
A: First find the molarity of phosphoric acid and then by using the formula of concentration, find volu...
Q: H3C CI NaBH, CH3 AICI,
A: CH3COCl, AlCl3 ====> Friedel Craft acylation reaction NaBH4 ====> Reduction
Q: What is the pKa of HC4H4O6 ? 2.17 10.26 O 6.59 8.15 4.34
A: generally pka gives the acidic nature of the molecule lower the pka more will be the acidic nature ...
Q: SPh SPh Br H- H. c) d) :CH3 CH3 H-ö:
A:
Q: Determine A and B to complete the reaction below: CH,OH CH,OH H+ OH H,0 OH OH OH r-D-glucopyranose a...
A: This reaction involves glycosidation. Glycosidation means formation of glycoside from aldose or keto...
Q: An aqueous solution is made by mixing 4.3 mL of 0.40 M Co(NO3)2 and 8.7 ml of 12.0 M HCI, and the fo...
A: A question based on equilibrium concept that is to be accomplished.
Q: Consider an isothermal column of an ideal gas at 25oC. What must be the molar mass of this gas be if...
A:
Q: OH H* (еxcess
A: Find the major product for the following reaction.
Q: The trans isomer of the compound below is more stable than its cis isomer. True or false?
A: Stereochemistry is branch of chemistry in which we deal with arrangement of atoms in molecules.
Q: 3. Determine the 3D structure of the molecule of formula C2H,O (H3CCHO). Indicate the geometry aroun...
A: CH3CHO - acetaldehyde Contains two carbon single bonded 1is methyl carbon, sp3 hybridised tetrahe...
Q: 2. Draw the curved arrow notation for each elementary step of the reaction sequence described below....
A:
Q: The resulting chromatogram showed the separation of two component in the sample. Component A is clos...
A: Given that, for a chromatogram separation of two components in the sample, where component A is clos...
Q: Measurements show that the enthalpy of a mixture of gaseous reactants decreases by 394, kJ during a ...
A:
Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them 1 atm pr...
A: Answer: This question is based on the understanding of system, surrounding and first law of thermody...
Q: Consider the reaction: ACC9)t 2B(g)<- ABa(g) * Ceg) AH = +56.9KJ The System is sitling at equilibriu...
A: According to Le Chatelier's principle when stress is applied on reaction at equilibrium then the rea...
Q: Indicate the 2 carbanions that this compound can give. Point out the most stable and explain it.
A: Carbanion is species in which carbon carrying negative charge. In carbanion, carbon atom is sp³ hybr...
Q: Formal Charge Distribution vs Oxidation States -ci: :ČI: For the Lewis diagram, above, determine: +1...
A: Oxidation state is defined total number of electrons that an atom either gain or loss in order to fo...
Q: Predict the splitting pattern(s) you would expect for the proton(s) in the molecule below that will ...
A:
Q: Nitric acid is often manufactured from the atmospheric gases nitrogen and oxygen, plus hydrogen prep...
A: Given two steps for the manufacturing of HNO3 is : Step.1 :- N2 (g) + 3H2 (g) --------> 2NH3 (g) ...
Q: Part A A weather balloon is inflated to a volume of 25.5 L at a pressure of 738 mmHg and a temperatu...
A:
Q: An aqueous solution is made by mixing 4.3 mL of 0.40M Co(NO3)2 and 8.7 mL of 12.0 M HCI, and the fol...
A:
Q: In lab, you add 1.985 g salicylic acid to a 125 mL Erlenmeyer flask, as listed in the procedure. You...
A:
Q: mulae of any reactants that will be reduced in the third column. reaction reactants reactants oxidiz...
A: 2Al(s) + 3Cl2(g) ---> 2AlCl3(s) 4Pb(s) + S8(s) ---> 4PbS2(s) 2Ni(s) + O2(g) ---> 2NiO(s) ...
Q: 8. 1MIEndel eats a coökié at lunch. Which of the following actions during the digestive process is a...
A: Please find your solution below : Digestion is a chemical change in which the enzymes breakdown the ...
Q: 6 ST4E.1 - Calculate the boiling point temperature of phenol (in the Celsius scale) if 4.3 grams of ...
A:
Q: Part A MISSED THIS? Read Section 6,9 (Pages 245 - 246) ; Watch IWE 6.15. A sample of N20 effuses fro...
A: Time of effusion for N2O = 50 s
Q: o-dichlorobenzene and p-dichlorobenzene can be separated by TLC. Which compound is more polar? Which...
A: Since you have posted multiple questions, the answer for first question is given below. Kindly repos...
Q: Indicate the 2 carbanions that this compound can give. Point out the most stable and explain it.
A:
Q: Calculate the pH of 0.50 L of a buffer solution consisting of 0.70 M formic acid () and 0.50 M sodiu...
A:
Q: Calculate potentials after the addition of 10.00, 25.00,50.00, and 50.10 mL of the reagent. Where ne...
A: Given: Volume of reagent: 10.00, 25.00,50.00, and 50.10 mL Cell: 50.00 mL of 0.1000 M V2+ with 0....
Q: The following initial rate data are for the reduction of nitric oxide with hydrogen: 2 NO + 2 H2 N2 ...
A: 2 NO + 2H2 -------> N2 + 2 H2O Rate = k [NO]m[H2]n k = rate constant m = order of reaction wit...
Q: on 1 of 3 The decomposition of N,O, can be described by the equation 2N,0, (soln) 4 NO, (soln) + O,(...
A:
Q: Which of the following will be soluble with water? Group of answer choices Vegetable Oil Desi Ghee...
A: The solubility of a compound mainly depends on the following factors; Temperature Nature of solute ...
Q: Consider the 25.0 mL portion of pH=4.45 buffer solution prepared as described in question 4. Calcula...
A: Solution: We will solve this question using henderson equation,
Q: Which of the following is a sugar alcohol? Mannitol Xylulose Trehalose
A: Sugars refer the carbohydrates.Carbohydrates can be defined as the organic compounds made up of carb...
Q: The formula for calculating the energies of an electron in a hydrogenlike ion is given in the follow...
A: For muli electron atom ,z is replaced with z-s. S is shielding constant value.
Prove the product of the reaction in the given image.
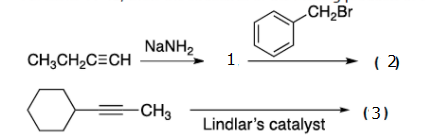
Step by step
Solved in 2 steps with 2 images

- Draw the organic products formed when cyclopentene is treated withfollowing reagent. (CH3)3COOH, Ti[OCH(CH3)2]4, (−)-DETWould 7b show stereochemistry? It looks like it has 2 chiral centers.DHA is a fatty acid derived from sh oil and an abundant fatty acid in vertebrate brains. Hydrogenation of DHA forms docosanoic acid [CH3(CH2)20CO2H] and ozonolysis forms CH3CH2CHO, CH2(CHO)2 (ve equivalents), and HCOCH2CH2CO2H. What is the structure of DHA if all double bonds have the Z conguration?
- Which double bonds in the attached natural products can exhibitstereoisomerism? Nerolidol is isolated from the angel's trumpet plant,caryophyllene is present in hemp, and humulene comes from hops.Ignoring stereoisomers, draw the two possible enols for butan-2-one (CH3COCH2CH3), and predict which one is more stable.(a) Why does p-dichlorobenzene have a higher m.p. than its o- and m-isomers?(b) Why is (±)-Butan-2-ol optically inactive?
- Draw the organic products formed when cyclopentene is treated withfollowing reagent. [1] CH3CO3H; [2] H2O, HO−Draw the structure of the unbranched isomer of C5H11Br that is most reactive in an SN1 reaction.What are the reagents used to convert 2-butyne to C4H8? What are the products when that C4H8 is mixed with OsO4, and H2O (the formula will be C4H10O2 racemic)? What is the product when C4H8 is mixed with RCO3H, and H2O (the formula will be C4H10O2 meso)?
- 4-chloro-2-pentene has a double bond that can have either the E or The Z configuration and a sterogenic center that can have either the R or The S configuration. How many steroisomers are possible ? Draw the structure of each?Draw the products formed when both cis- and trans-but-2-ene are treated with OsO4, followed by hydrolysis with NaHSO3 + H2O. Explain how these reactions illustrate that syn dihydroxylation is stereospecic.Name the alkene below.Use only E/Z designators to indicate stereochemistry.

