Q: [H+] 11 [OH™] 2[CO-] + Answer Bank 2[H+] [HCO3] + 3[H+] [CO] [H,CO, ]
A: The balanced chemical equation is the equation in which the number of atoms on the both sides of…
Q: Why is diethylamine more basic that methylation? ( provide structures)
A: Basicity of an organic amine compound is determined by the availability of lone pair electrons on…
Q: Functional group (Name) Hydroxyl General: Specific: Carboxyl Chemical formula or Structural formula…
A: Since, Functional group represent the characteristics of the compound. Example- For methanol or…
Q: g of Mg(NO3): ?
A: Molarity of a solution is defined as numbers of solute dissolved per litre of the solution. Given…
Q: 3.1. Consider 12C, 13C and 13C- 3.1.1 How many neutrons are found on each? 3.1.2 How many electrons…
A: Carbon has atomic number = 6 In a nucleus of atom , proton and neutrons are present. Electrons…
Q: Used 24.38 mL of a 0.1014M NaOH solution to titrate the dilute HCL solution to the endpoint in one…
A: Molarity of the concentrated HCl stock solution, C = 0.9889 M Volume of stock solution used to…
Q: Q2 State the number of significant figures in each of the following measurements: 4.5 m 0.0004 L 805…
A: Rule of significant digit-1) In non decimal number, 0 comes after non-zero not count in significant…
Q: Consider a tripeptide (a peptide that's only 3 amino acids long) composed of one molecule each of…
A:
Q: The rate constant for the first order reaction AB+C is k-44 x 103 min at 57 K. What is the half-life…
A: As per student's request I am answering only 12th question.
Q: Glutamic acid (Glutamate) has an R group that has the following pKa values 2.19, 4.25 and 9.6. Draw…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 24) What is the density of NH3 at 5 atm pressure and a temperature of 35°C? B) 3.36 g/L A) 16.6 g/L…
A:
Q: Write the oxidation half reaction for the ionization of Zn atom to a Zn+2 ion.
A:
Q: What is the half-life of a first-order reaction if k =0.050 s¨¹? Your Answer: Answer units
A: Given that The rate constant of the first order reaction, k = 0.050 s-1 The half-life period of the…
Q: 2.1. Give a detailed mechanism of following products. Show the major product(s) and g the IUPAC name…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Name of the following compound.
A:
Q: 3. Determine the values of K that correspond to the following values of K₂. (a) SO₂Cl₂(g) → ← SO₂(g)…
A:
Q: Given the decomposition reaction: 2C1O2(aq) Experiment [CIO₂], (in M) 0.0114 0.0228 0.0114 0.0228 1…
A:
Q: Activity 1.2 Rice that Sinks! Materials: small transparent bowl or glass, 1 tablespoonful of cooked…
A: Mixture: A mixture can be defined as a substance that is made of more than one element or compound.…
Q: 3 Serotonin (C10H12N₂O) is a neurotransmitter that when balanced is responsible for happiness,…
A:
Q: 3.) Vanilla ice cream is made with vanillin (C3H8O3), which is an artificial flavor. If I have 186…
A:
Q: An ion from a given element has 24 protons and 21 electrons. What is the charge on the ion? What is…
A:
Q: Which of the following is correct concerning the orbital energy? O4s <4p < 4d < 4f O4s <3s<2s< 1s…
A: Orbital energy:- Orbital energy is decided by (n+l) rule. •Here n=> Principal quantum number…
Q: Select all that apply. Which of the following bases are strong enough to deprotonate…
A:
Q: Prepare each compound from CH₂CH₂CH₂CH₂OH. More than one step may be needed. a. CH₂CH₂CH₂CH₂Br C.…
A: (a) 1-Bromobutane can be prepared from Butan-1-ol by reacting Butan-1-ol with HBr. (b)…
Q: Homogeneous or Heterogeneous. Write the name of the mixtures on the correct column. Homogeneous…
A: Homogeneous solution is solution with uniform composition and properties. Heterogeneous solution is…
Q: Why is diethylamine more basic than methylamine? (Provide structures)
A: here we are required to provide an explanation for the statement that diethyl amine is more basic…
Q: What is the weight percent of toluene in a mixture of toluene and n-heaxne that has a spevific…
A: Here we have to find weight percentage of toluene in a mixture of toluene and n- hexane . And given…
Q: How many grams of Mn are there in 4.51 moles of Mn? grams
A:
Q: 100.0mL of 0.45M HBr are mixed with 50.0mL of 0.85M CaCl2 and 100.0mL of 0.80M NH3. Determine…
A: According to the question we have, The volume of HBr = 100.0 ml The molarity of the 100.0 ml HBr =…
Q: 13) A solution was made by dissolving 4.05 g of a solute in 1369 g of acetone. The solution boiled…
A:
Q: Which of the following corresponds to a second order reaction? a) [A]²= [A]o - kt Ob) [A] = [A]o -…
A: Second ordered reaction of kinetics : kt = 1/[A]t - 1/[A]0 .... where, k = rate constant of…
Q: 3.1. Consider ¹2C, 13C and 13C- How many neutrons are found on each? How many electrons are found on…
A:
Q: A possible product of chlorine gas added to 3-methylhaxane is: O hexane O 3-chloro-3-methylhexane O…
A:
Q: Based on your observations in Part III, which reagent is present in excess? Which reagent is the…
A: To determine the limiting reagent, reagent present in excess, theoretical yield based on limiting…
Q: Discussion of the compound intermolecular forces, and how (and why) they change as your compounds…
A: Intermolecular force between the two molecules of compound is a weak attraction force between the…
Q: Consider the equilibrium for the transesterification reaction. mmmmmmmmmmmm triacylglycerol +…
A: The given equilibrium for transesterification is…
Q: Br D=Ú C-H
A: Organic compound are those compounds which are made up from carbon and hydrogen. A functional group…
Q: Chemistry A solution is 0.010 M in both Br- and SO4^-2. A 0.250 M solution of lead(II) nitrate is…
A:
Q: 5. Heparin has a molar mass of 600g/mol. For a patient on a blood thinner regimen, you need 100 ml…
A: Given, Note: 1 molar = 106 micro molar Concentration of Heparin solution = 500 micro molar = 0.0005…
Q: In your experiment you have 0.2 M solutions of HCl, NaOH, and the uncharged free base form of…
A: Given Data: Volume of TEA = 2.00 L Concentration of TEA = 0.040 M pKa of triethanolamine = 7.8 pH =…
Q: Select the compound that has the highest boiling point, based on that compound's dominant…
A: Answer: Value of boiling point is depends on the strength of intermolecular forces. Higher the…
Q: The pH of a 0.1 M acetic acid solution is 2.885. What is the dissociation constant of acetic acid?
A: Given, that the pH of a 0.1 M acetic acid solution = 2.885 We have to find the dissociation constant…
Q: An experiment produces evidence that the evaporation of 4.00 g of liquid butane, C4H10 (l), requires…
A:
Q: abel each carbon atom with the appropriate geometry. CH₂ ||
A: Given compound s organic compound as it contain carbon and hydrogen atoms.We have to tell geometry…
Q: 1.1 Give the correct IUPAC names of the following compounds: 1.1.1. 1.1.2. OCH3 å OCH3
A:
Q: B) 105 C) 12.7 D) 140 14) The decomposition of N₂Os(g)-NO₂(g) + NO3(g) proceeds as a first order…
A: #Q.14: The first order reaction is: N2O5(g) → NO2(g) + NO3(g) half-life of the first order…
Q: The table below gives the plasma drug concentrations (Cp) obtained following an intravenous bolus…
A: In the given process the concentration of plasma drug varies with time and rate of decomposition is…
Q: Use the following information to answer the next question: Mass of aluminum calorimeter: 470.0 g…
A:
Q: Use the relationship equation of state, P= a a RT a 1 Vm-b VTV (V+b) Drag the appropriate labels to…
A:
Q: solution of carbonate (pKa = 6.3, 10.3) is at pH 10.1. Calculate the base:acid ratio at this pH.
A:
Name the compound below:
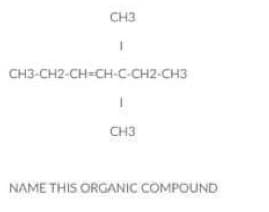
Step by step
Solved in 2 steps with 2 images


