Chapter17: Alcohols And Phenols
Section17.SE: Something Extra
Problem 22VC
Related questions
Question
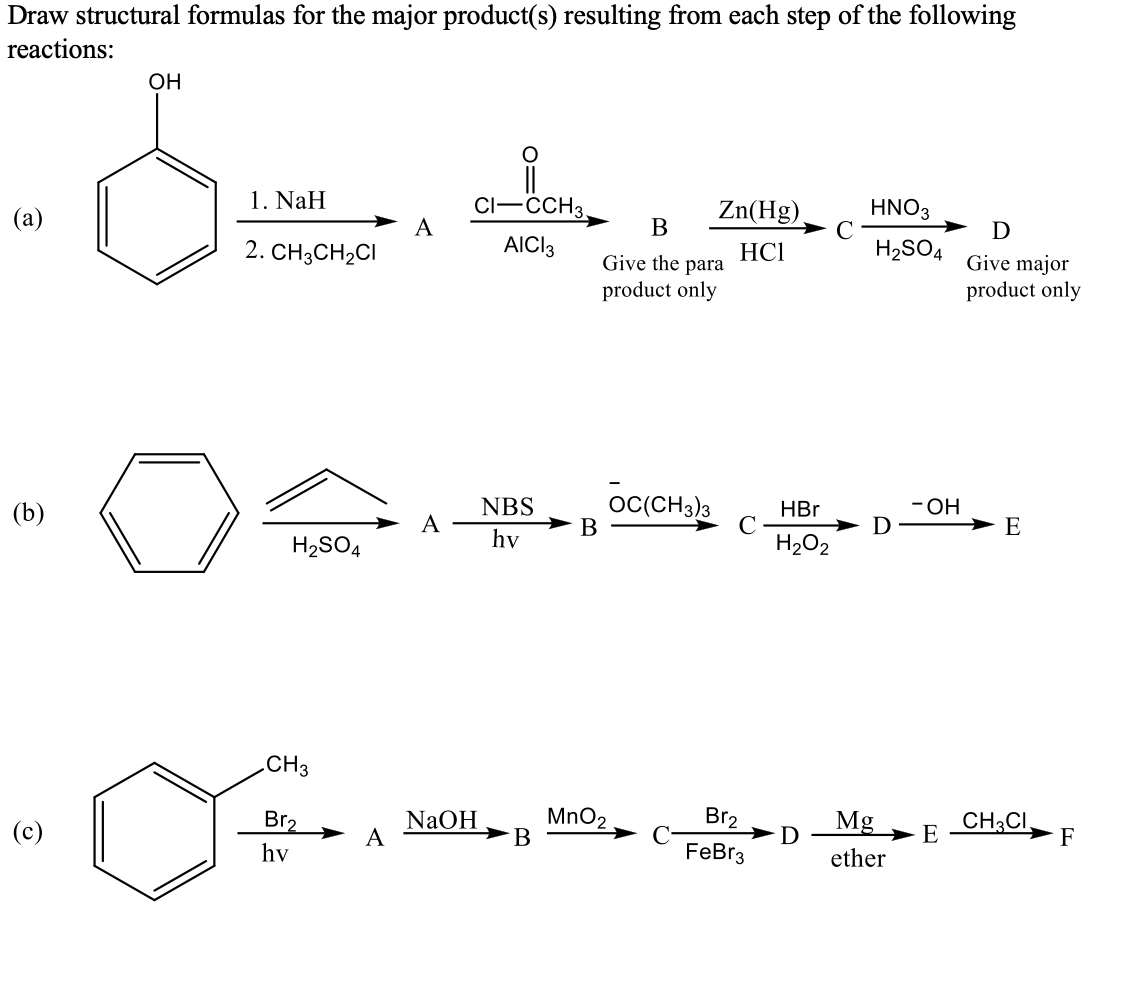
Transcribed Image Text:Draw structural formulas for the major product(s) resulting from each step of the following
reactions:
OH
1. NaH
CI-CCH3.
Zn(Hg),
HNO3
(a)
A
В
D
AICI3
HC1
H2SO4
Give major
product only
2. CH3CH2CI
Give the para
product only
- ОН
OC(CH3)3
В
HBr
C
H202
NBS
E
(b)
A
hv
H2SO4
CH3
NaOH
A
MnO2
В
Br2
Mg
CH3CI.
E
Br2
F
(c)
D
ether
FeBr3
hv
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 3 images

Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning