Q: Determine the oxidation state and coordination number of the metal ion in each complex ion. a. (Co(N...
A: To solve the given problems related to coordination compounds, we must be aware of the following ter...
Q: Carbon tetrachloride can be produced by the following reaction: CS (g) +3 Ca (g) S,Ch (g) + CC4 (g) ...
A: Kc is also known as the equilibrium constant. It is defined as the ratio of the concentration of pro...
Q: 5) The table below shows AHvalues for 3 reactions. Reaction DHran 1 C,H;(g) + 5/2 0,(g) → 2CO;(g) + ...
A: Enthalpy of a reaction is the heat energy released or absorbed from the reaction. The enthalpy of fo...
Q: Produce a bar graph that shows the effect of substrate concentration on the rate of reaction for amy...
A: The graph shows that how the enzymes act on the substances to form products. It would be observed th...
Q: 4.00 140 1.6 120 -4.25 1.4 100 1.2 -4 50 80 1.0 4.75 0.8 60 0.6 -5.00 o1 2 3 Time (s x10 2. 3. 15 6....
A: From the graph we can get the idea about the order of the reaction .
Q: 7) Propane is a gas commonly used to fuel gas grills and camping stoves. The structure of propane is...
A: Combustion Reaction: It is the reaction in which a substance reacts with the oxygen gas and releases...
Q: c) A student titrated 50.0 mL of a 0.10 M solution of a certain weak acid with NaOH. The results are...
A:
Q: Identify any chiral centers within the 2 molecules and identify whether each molecule is chiral or a...
A:
Q: How may grams of chlorine are in 7.74 moles of Calcium chloride?
A: First we have to calculate no. Of grams of chlorine present per mole from their molar masses and the...
Q: Answer the following questions about weak acids. Use the Ka values in the following table to help an...
A: In the given question we have to predict which one of the acid has lowered pH and acidic by using th...
Q: Explain if a hydrogen from the OH group and a hydrogen from the carbon atom with the OH group are re...
A: A compound having a hydroxyl group bonded to a carbon atom is known as alcohol. Some of the examples...
Q: The following reaction is second order in NONO and first order in O2O2: 2NO(g)+O2(g)→2NO2(g)2NO(g)+O...
A:
Q: Give any commọn name for the following compound.
A: IUPAC is the universally-recognized authority on chemical nomenclature and terminology. IUPAC is I...
Q: What is the chemical formula for both, dichlorine heptabromide and trinitrogen pentaphosphide?
A: Different type of compounds have different formula according to their respective valencies and Oxida...
Q: Lactic acid, HC3H5O3, is a monoprotic acid that dissociates in aqueous solution, as represented by t...
A: The number of moles is the ratio of the mass of a substance per molar mass of that substance. In a c...
Q: Fill in the blanks: Part 1: subscripts, ratio, compound The ______________ show the ___________ of ...
A: Given question is based on concept and related to inorganic chemical bonding chemistry. Ionic bonds ...
Q: When you convert feet to inches, how do you decide which portion of the conversion factor should be ...
A: Feet and inches are the measuring units of length. Both the units can be converted from one form to ...
Q: Predict the major organic product
A:
Q: Which statement on planes is correct? Part of (300) planes are (100) planes. Part of (100) planes ar...
A: Planes are drawn by the use of miller indices as shown below: Let us take an example of miller indic...
Q: Please draw the structure of dopamine and list all the wavenumbers you would expect in the IR spectr...
A: A dopamine molecule consists of a catechol structure i.e. benzene ring with two hydroxyl side group...
Q: The reaction of benzene with (CH);CCH;CI in the presence of anhydrous aluminum dlnide produces princ...
A: Benzene undergoes electrophilic substitution reactions with the given reagents. Aluminium chloride a...
Q: Q2: the solubility products for a series of hydroxides are: BIOOH Be(OH)2 Tm(OH); Hf OH), Which of t...
A: Solubility product is useful quantity to measure the condition of precipitation .
Q: Write the correct chemical formula of each of the following salts. solution 1: 0.075M potassium phos...
A: We can write chemical formula of an ionic salt by criss-crossing the valencies of ions.
Q: A 10.00-ml. solution containing cr was treated with excess AgNO to precipitate 0.4368 g AgCl. What w...
A: According to the mole concept, in terms of mass, the amount of substance in moles is equal to the ra...
Q: Naphthalene has a log Kow of 3.33. Estimate its soil-water partition coefficient normalized to organ...
A: For the soil-water partition coefficient normalized to organic carbon and the 95 per cent confidence...
Q: 3. A mixture of Calcium carbonate and ammonium carbonate is 61.9% carbonate by mass. Find the mass p...
A:
Q: Sodium chloride can be produced from reacting sodium metal with chlorine gas. Carly reacts 2.6 g of ...
A:
Q: (a) How many milliliters of a stock solution of 13.0 M HNO3 would you have to use to prepare 0.500 L...
A:
Q: Consider the following equilibrium: 2H2(g) + X2(g) 2H,X(g) + energy Addition of X2 to a system descr...
A: The reaction in equilibrium can be understood by Le Chatelier's principle. It says that any reaction...
Q: Arrange the reactions in increasing order of rate. What type of reactions are these?
A: Given: some reactions To find: there reactivity order. Solution: SN1 reaction means nucleophilic su...
Q: Calculate the boiling point of a 1.75 m Mg(NO3)2 aqueous solution.
A: When a solute is dissolved in the water, the vapor pressure of the solution is lower than the pure s...
Q: A sample of a gas at room temperature occupies a volume of 36.0 L at a pressure of 702 torr. If the ...
A:
Q: Convert 7.84 x 104 from scientific notation to decimal form.. Give the answer in the correct format ...
A: Scientific notation is a means of representing quantities that are too big or too small (usually wou...
Q: Suggest a synthesis for the following alcohol starting from an aldehyde or a ketone and an appropria...
A:
Q: part b and c
A:
Q: Determine the equilibrium concentration of gas A.
A:
Q: can someone calculate the 10 concentrations please ?
A: Given that: the volume of potassium permanganate solution = 50 cm3 the volume of potassium dichroma...
Q: What is the reaction mechanism for the major product? Please draw.
A: Please find the attachment
Q: The density of a material is 2 kg/m. What is its specific volume?
A:
Q: If a solution of 1.00g of unknown solute is added to 9.116 g of steric acid and the solution freezes...
A: Molality of solution is defined as number of moles of solute dissolves in per kg of solvent. Molalit...
Q: At 25 °C the vapor pressures of benzene (C6H6) and toluene (C>Hg) are 93.4 and 26.9 torr, respective...
A: Given data : Mass of benzene in the solution = 57 gms Mass of toluene in the solution = 79 gms Molar...
Q: Can someone help pls I don't understand thanks
A: Introduction: NMR stands for nuclear magnetic resonance spectroscopy. In this, the molecules are stu...
Q: Heat of formation values for four compounds are given in the table above. a) Sketch an energy diagra...
A: Enthalpy of reaction is equal to the difference between total formation enthalpies of products and t...
Q: Consider the following system at equilibrium where K. = 5.10×10-0 and AH° = 268 kJ/mol at 548 K. %3D...
A: From the given data 1) True Justification: The value ∆H° for a forward reaction that is productio...
Q: Draw the expected products of the following reactions. Mgl PCI5 DCM THE then H30*
A: Since you have posted multiple independent questions in the same request, we will solve the first qu...
Q: For the following electronic transitions in the hydrogen atom, calculate the energy, frequency, and ...
A:
Q: [References Cyclohexane, C6H12, a hydrocarbon, can isomerize or change into methylcyclopentane, a co...
A: The equilibrium is established between cyclohexane and methylcyclohexane. cyclohexane ⇔ methylcycloh...
Q: The chemical environment of carbons can be deduced form their chemical shifts. Deduce the relative c...
A:
Q: The questions below refer to the following system: Co(H,O)6 +4 Cl =C0C1* + 6H0 (pink) (blue) When co...
A:
Q: Hello I am having some trouble understanding how to balance the equation on letter (g) using the hal...
A: Answer:- This question is answered by using the simple concept of balancing the chemical equation us...
Give the: mechanism, product(s), intermediate, and twll if there is a hydride shift or methyl shift
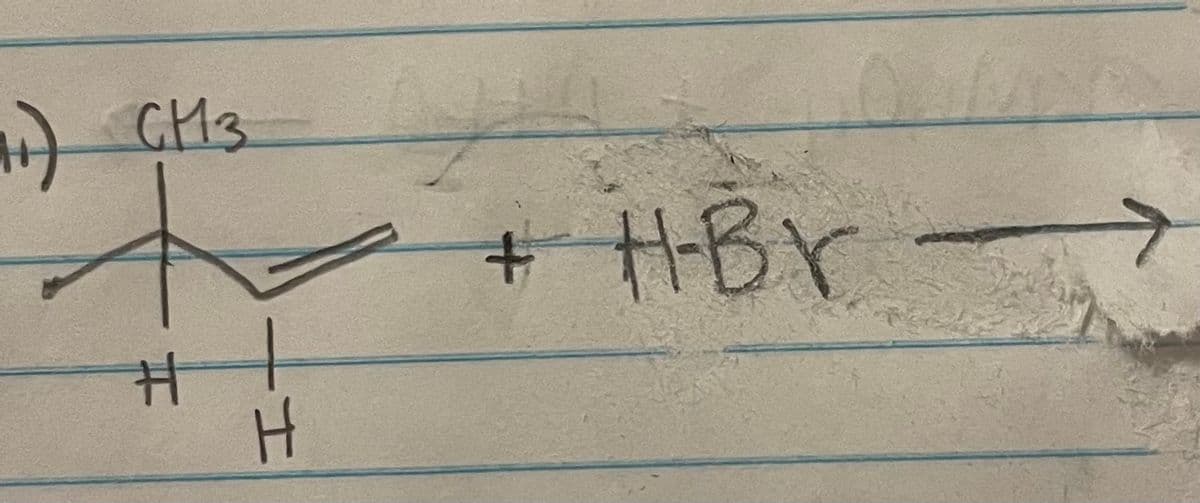
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images


