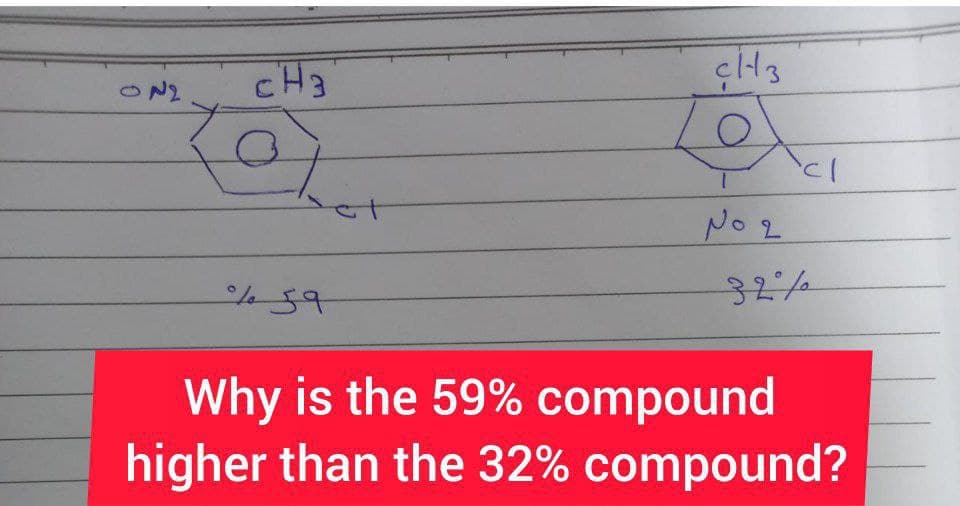
CH3 N2 No 2 32/ Why is the 59% compound higher than the 32% compound?
Q: How many moles of Na,SO4 are in 100.0 mL of a 0.2500 M solution ? The GFW is 142.07 g. O A. 0.0500…
A: 0.25 M Na2SO4 solution. Molarity = number of moles in 1L of solution So, 0.25 moles of Na2SO4 in 1 L…
Q: How many total atoms are in (NH4)3PO4
A: An atom is the smallest unit of matter.
Q: 8 7- 6- 2- 1 405 180 225 270 315 [Substrate] (nM) 45 90 135 360 450 495 540 58! What are the units…
A: Solution Michaelis menten kinetics is one of the best models of enzyme kinetics. Value of…
Q: How many moles of sodium hypochlorite can be prepared from 32.5 g of NaOH?
A: Moles of Sodium hydroxide equals moles of Sodium
Q: IDENTIFY THE FOLLOWING PARTS - A B C D E F G H I J K
A: The given diagram is a 3D model of a eukaryotic cell as we can see the membrane-bound organelles…
Q: How do I name the following compounds:? a. Fe3(PO4)2 b. P2O6 c. F2O d. Mo(SO4)3…
A: A chemical compound is a chemical substance that is composed of many identical molecules which are…
Q: Br-CH2CH2CH2-Br + Zn its give propane I C Cyclo-propene II C propene.II 0 Cyclo-propane .IV O
A: The chemical reaction is the reaction in which one or more reactants are converted to form one or…
Q: Give the noble gas shortened electron configuration for Hf 16 4 5 Give the noble gas shortened…
A: Hf or Hafinum atom have 72 electrons.
Q: What does the formula DPD = SF - TP mean?
A: The above mentioned question is related to cell membrane. It is asking the meaning of mentioned…
Q: 11 of 11 What volume (in uL to 1 decimal place) of stock SybrSafe solution (3.4 mg/mL) would you add…
A: Given- 1. Concentration of stock solution = 3.4 mg/ml 2. Volume of agarose solution = 120 ml 3. The…
Q: NH O purine O pyridine H. Consider compound 2. Compound 2 is classified as a purine NH pyrimidine…
A: Introduction: Compound one is pyrimidine. (Uracil) Compound two is a purine. (Guanine) Compound…
Q: How many total diastereomers is possible for nonulose?
A:
Q: Explain why H is 5' and E is 3'
A: DNA replication is the process of the copying of the DNA strand by the use of a DNA polymerase…
Q: Given: 2KI +Pb(NO3)2 --> PbI2 + 2KNO3. How many grams of lead (II) iodide are produced if 0.250…
A: 2KI +Pb(NO3)2 --> PbI2 + 2KNO3 Thus, 2moles of KI with 1mole of Pb(NO3)2 gives 1 mole of PbI2…
Q: In the given formula, p+q=1, what does q stand for?
A: Hardy - Weinberg law - It is an equation which is used to calculate the frequencies of different…
Q: What coefficient must be placed in the following blanks so that all atoms are accounted for in the…
A: As per the law of conservation of mass, mass can neither be created nor destroyed. This also applies…
Q: There are three isomers of chloropropane. (1-chloropropane, 2-chloropropane, and 3- chloropropane.)…
A: Chloropropane is a chemical compound formed by the reaction of n-propyl alcohol with phosphorous…
Q: If you prepare a different prescription using 5 g of zinc sulfate [ZnSO4), how many mmols of zinc…
A: Milli mole iss 1000th part of mole. So 1 mole= 1000 millimoles or we can also write it as 1…
Q: Determine the number of atoms in 30.0 g of P, with the correct amount of sig figs.
A: One mol of any substance is equal to avogadro number of molecules or atoms. In such cases you are…
Q: How many grams of CH3OH can be made from 10.0 g of CO if it all reacts?
A: Methanol (CH3OHCHX3OH) is made by reacting carbon monoxide (CO) and hydrogen (H2) at high pressure:…
Q: How many moles of Na2S2O3 would be required to react completely with 0.450 g of AgBr?
A: As it is given, Na2S2O3 is reacted with the AgBr, the balanced reaction is shown below: 2Na2S2O3 +…
Q: 1. Prepare 50 ml of 50% v /v alcohol with 95% v / v alcohol. 2. Prepare 50 g of 40% w / w alcohol…
A: Multiple subparts are asked. I will answer the first three subparts, as per guidelines. Asked :…
Q: What coefficients must be placed in the following blanks so that all atoms are accounted for in the…
A: When balancing chemical reaction the number of atoms of each element in left hand and right hand…
Q: 10 12 3 13 4 9 9 10 8 14 What is the structure labeled "5" in the image?
A: In the biological system, two types of cells are defined as eukaryotic and prokaryotic. The…
Q: What type of alkyl halide is this? (CH3)2CHB"
A: Alkyl halides are chemical compounds that are often derived from alkanes that contain one or more…
Q: +2 H3N O:
A: The compounds that contain COOH as functional groups are called acids. The compounds that contain…
Q: 9. Draw the Electron Structure for Rb & O 10. Draw the Electron Structure for Sr & F Formula Unit:…
A:
Q: typical ratio of C:H:O
A: Carbohydrate is a biomolecule consisting of carbon, hydrogen, and oxygen. They are polymers of the…
Q: How many grams of Cl2 are needed to react with 50.0 g of CH4?
A: As we know to react with the CH4, 2moles of Cl2 are required. So following is the balanced chemical…
Q: How many g of dextrose are in 3 L of D5W
A: Dextrose is a simple sugar. it is made from substance chemically identical to glucose. it is used…
Q: The colour change for the test reaction of an alkene with KMNO4 is from: A. Purple to black/brown O…
A: Potassium permanganate is an inorganic compound and is of purple colour due to permanganate ion. It…
Q: H C-C-N-H エ-
A: Condensed structure formula is written in a line text and it is less extended form than normal…
Q: What is the Mass of 3.5 L of H2O
A: Given: The Mass of 3.5 L of H2O.
Q: How many moles of HCl are there in 75.0 mL of 0.160 M HCl?
A: Molarity can be stated as the soluble moles of solute, which is dissolved per liter of solution.
Q: 10 12 3 1 2 13 4 24 9 10 8. 14 What is the structure labeled "6" in the image?
A: In the given image, structure of cell is represented. Cell is a basic structural and fundamental…
Q: Select the product for the following cyclization CHO H- OH OH H н он HÓ HỒ но HO но HO OH но H OH он…
A: Sugars such as glucose exist in two forms in closed chain form or open-chain form, which is shown in…
Q: Indicate the COOH-pKa value of the HAY tripeptide on the titration curve. Titration curve of…
A: The titration curve of peptides indicates the buffering zones and the pKa value of each ionic…
Q: the titration are designated I to VII. 12 12 II 10 97 VI 8 pH 3.9 4 20 IV 0.5 1.5 2.5 3 Equivalents…
A: Amino acids are the basic unit that form proteins. Amino acids contain an amino (NH3+)group, a…
Q: In 6O2 how many oxygen (O) atoms are present
A: 6O2 stands for six oxygen molecules. 6H2O = six water molecules The plant will use the glucose as…
Q: What is the half-reactions and the Ecell to this equation: 2Ag (s)+H2S (g)+ 1⁄2O2 (g) →Ag2S+H2O (l)
A: An oxidation-reduction reaction is also known as a redox reaction, is a type of chemical reaction…
Q: choices for question 1: H LOH NH2 INH3* HO H 'N' `NH2 `NH2 a b H LOH LOH NH2 INH2 `NH2 `NH2 H A H A…
A: Hi! Since you have posted multiple questions, we will answer the first one for you because we are…
Q: How many H2O particles were produced by creating the following structure? R H R R -N-Ç, H-N-C,- H H…
A: Extention of a polypeptide chain is a typical dehydration reaction where OH from carboxyl group and…
Q: Why is it difficult to determine an exact number for P/O ratios?
A: P/O ratio refers to the Phosphate/Oxygen ratio. P/O ratio signifies the number of ATP produced for…
Q: A reaction at 23°C has ∆G = 1 kJ mol–¹. Why might this reaction become spontaneous at 37°C?
A: A reaction at 23 degree celecius has 1 KJ mol-1 This would make the G value negative and hence the…
Q: Why 3' end should be G and C and 3' should not have more than 3 consecutive G and C?
A: Polymerase Chain Reaction ( PCR) is a technique of making numerous copies of genetic material ( DNA…
Q: Which of the following Fischer structure is repres structure? CHOH
A: The Fischer and Haworth projection are the two different ways to represent the organic molecules. As…
Q: How many stereoisomers are possible in Aldopentoses? Select one: a. 6 O b. 8 с. 2 d. 4
A: Stereoisomers are molecules with the same chemical formula and bound atom sequence but different…
Q: Which of the following shows B-D-Gulose? A) B) CH2OH CH2OH OH OH OH OH H. OH OH H H ÓH C) D) E)…
A: Carbohydrates are polyhydroxy aldehydes or ketones which are associated with reducing property and…
Q: If I put 80g of NaOH in 0.5L of H2O, what is the molar out of solution?
A: Molarity is the concentration of the solution that is defined as the number of moles of the solute…
Step by step
Solved in 2 steps


