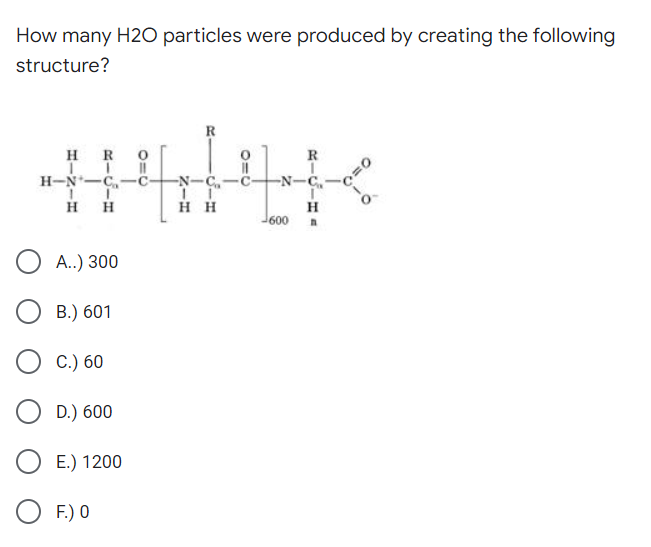
How many H2O particles were produced by creating the following structure? R H R R -N-Ç, H-N-C,- H H -N-C,- нн H J600 А.) 300 B.) 601 С.) 60 O D.) 600 E.) 1200 O F.) 0
Q: In Gel filtration chromatography, when will you stop collecting eluents if sample is not colored?
A: Gel-filtration chromatography is a type of partition chromatography that is used to separate molecul...
Q: . To which class does each enzyme belong? Explain your answers. a.) pyruvate decarboxylase b...
A: As you have posted multiple questions we are supposed to solve only the first question ..If u want a...
Q: MRI What does the "induced free decay" curve describe?
A: The term "free induction decay" refers to a short-lived sinusoidal electromagnetic signal that devel...
Q: Which of the following statements is FALSE? Select one: a. Secondary structure of a protein determin...
A: Amino acids are monomers of protein they are linked with each other by forming peptide b...
Q: The order of elution of amino acids X, Y, VW and Z in a cation exchanger eluted with a buffer of inc...
A: Ion exchange chromatography is used to separate molecules based on the charge. In the ion exchange c...
Q: prepare 0.7% potassium Chloride (KCI) in 0.3 liter? * 0.21 gm O 21 gm O 210 gm 0.021 gm
A: Diluting or mixing two or more substances or even compounds refer as 'Dilution'. The ratio of...
Q: In what order would the following proteins eluted from a gel filtration column of Sephadex G200: Myo...
A: Chromatography is a method of separation of specific compound from the mixture of compounds. This ca...
Q: Biological activity of proteins is dependent on which? A. peptide bonds B. thermodynamic properties...
A: Proteins are composed of a linear chain of amino acids attached together via peptide bonds. Proteins...
Q: List down the 25 different antibiotics and give the mechanism of action
A: Antibiotics, also known as antibacterials, are medications that either kill or slow the growth of ba...
Q: Drawn below is a schematic of a transmembrane protein. AN Extracellular Cell membrane Cytosolic side...
A: Introduction: A membrane protein is a protein molecule that is connected or linked with the membran...
Q: The gene for the primase enzyme is mutated and is nonfunctional. What is the result of such a mutati...
A: DNA replication unlike transcription cannot start ab initio and requires a primer that is elongated ...
Q: Which of the following could be the DNA template for the following protein primary structum Methioni...
A: In order to form a polypeptide chain, amino acids are linked together in a sequence. During protein ...
Q: 23. In the concerted model, the most active enzyme form will be when a. All subunits are in the R st...
A: Enzyme can have it's subunits in 2 conformations : Low-activity state -(Tense)T state High-activity...
Q: 32. Which one of the following is NOT a typical characteristic of the active site of an enzyme? A) T...
A: Substrate molecules bind to the enzyme, which then undergoes a chemical reaction. The active site is...
Q: In the second reaction of the pentose phosphate pathway that generates NADPH, What are the subtrates...
A: The pentose phosphate pathway also called the hexose monophosphate shunt is an alternative pathway f...
Q: How can we use software to identify restriction-enzyme cutting sites in sequenced DNA ?
A: Restriction analysis is the process of identifying restriction mapping sites in DNA sequences by emp...
Q: Which of the following statements concerning structural characteristics of bile acids is CORRECT? * ...
A: Bile acids are a collection of acidic steroid molecular entities with unusual physical, chemical, an...
Q: the different individuals involved in a clinical laboratory (their job specification -qualification,...
A: JOB SPECIFICATION Laboratory Director QUALIFICATION and TRAINING Doctoral degree (e.g., MD or ...
Q: Assume that mitochondria contain 0.35 Molar KCL and 0.012 Molar NaCl. Calculate the grams per liter,...
A: Osmolarity is defined as the concentration of a solution expressed as the total number of solute par...
Q: Name the four different types of interactions responsible for creating tertiary structures in protei...
A: Tertiary structures shape is determined via the different bonding interactions between side chains o...
Q: Our body is compost of about 70% water which means that majority of the chemical processes happens i...
A: The amino acids are classified based on the polarity as, Polar amino acids (hydrophilic)- These are...
Q: Why phosphoglycerides can be found in the cell membrane?
A: A cell membrane surrounds all live cells. Plant and animal cells both have a cell nucleus, which is ...
Q: What are the other methods of detection for blood glucose determination? Explain the principle invol...
A: There are different methods for estimating the levels of glucose in the blood. The different methods...
Q: C. Deepen (Pagpapalalim ng Kaalaman) Let us do the activity below. (50 mins. with provision for anal...
A: Transcription is the process of synthesis of mRNA from DNA. And the process of synthesis of protein ...
Q: Assume that Compound A is an inhibitor of the electron transport chain's Complex II. On the other ha...
A: Electron transport chain is the transport of electrons through a chain of electron carriers to the m...
Q: Modified true or false. Write the correct answer if the underlined word is false.
A: Any organic molecule that consists of a basic amino group, an acidic carboxyl group, and a distinct ...
Q: COO COO HC- CO0 C-H CH HO-CH COO CO0 COO O ligase O transferase O lyase O isomerase O hydrolase oxid...
A: The enzymes can be classified based on the type of reactions that they catalyze (example, ligase, ly...
Q: Describe the role that protein domains play within a protein’s three-dimensional structure
A: The domains are in a protein are distinct structural units in the three dimensional structure of the...
Q: How much ATP and CO2 can be derived from 500 g of lactose?
A: Lactose is a disaccharide made up of galactose and glucose ...
Q: Aldosterone is glucocorticoid hormone with Na+/K+ balance i the human body. Cortisol is an adrenocor...
A: The hormones in the body play a central role in maintaining key homeostatic processes in the body.
Q: Write the net ATP in glyconeogenesis and write the net reaction of it...
A: Glyconeogenesis is the synthesis of sugars like glucose and glycogen from sources other than carbohy...
Q: The main role of carbohydrate in the cell membrane is ____ Select one: a. recognition b. reception c...
A: Carbohydrates are composed of carbon, hydrogen, and oxygen in the ratio of 1:2:1. Carbohydrates are ...
Q: What are the changes occur in oxidative randicity? Explain.
A: Oxidative rancidity is a chemical process when lipids oxidize through a complex series of reactions ...
Q: A group of BS Biology students were tasked to design and fabricate different synthetic cell membrane...
A: Several factors determine the fluidity of the cell membrane membrane like membrane lipid chemistry, ...
Q: In the first cycle of fatty acid synthesis via the acyl carrier protein system, the acetyl group on ...
A: Fatty acid synthesis is the formation of fatty acids from acetyl co A and NADH using fatty acid synt...
Q: Which of the following monosaccharides structural relationship is CORRECT? * сно сно сно но- -H H- -...
A: The stereoisomers have the same composition but differ in the orientation of parts in space. Two typ...
Q: What would happen to the functionality of cholic acid if the 3 hydroxy groups were removed? A) Ther...
A: Fats are water insoluble amphipathic molecules. More than 90% of the structure of a fat is hydrophob...
Q: With regard to Na+ and K+ equilibrium potentials and the resting and active membrane potentials, wri...
A: Under all conditions of negative and positive membrane potentials; the concentration of Na+ is highe...
Q: Starting with pyruvate (CH3COCOO—) as the precursor, the first step of gluconeogenesis is activatio...
A: Gluconeogenesis is a metabolic process that results in the transformation of non-carbohydrate carbo...
Q: Explain the enzymes.
A: An enzyme is a biocatalyst that increases the rate of chemical reaction without itself being changed...
Q: What biological rationale can explain why there are so few variants observed at position 65 of the h...
A: The iron atom in the heme group can form a total of 6 coordinate bonds . 4 of these are formed with ...
Q: A. Calculate the final percent concentration of the erythrocytes in each well: (NaCl, CaCl2, MgCl2)...
A: This is classical example of chemical calculation is lab. I am going to do all calculation step wise...
Q: Write the structure formula, three-letter and one-letter abbreviation for each essential amino acid ...
A: Amino acids contain amino group and carboxyl group along with R side chain. The R side chain defines...
Q: 8. In patients With Albrighl-Butler syndrome (kidney acidoSIS) secretion of H* into distal tubule lu...
A: Butler-Albright Syndrome is a kind of primary distal renal tubular acidosis. Undetermined. Females a...
Q: Explain the Donnan Membrane Phenomenon. Why is it important for the homeostasis of the cell?
A: There occurs different biochemical reactions in the human body. Since a chemical reaction is the con...
Q: Choosing the proper buffer solution In Protein Precipitation, two liters of 5mM buffer solution wit...
A: The processes that occur in the body require a specific and stable pH range. Buffers are solutions t...
Q: A mixture containing arginine, glutamic acid and isoleucine was subjected to electrophoresis at pH 7...
A: Amino acids differ from each other on the basis of their side-chain properties. There are twenty sta...
Q: Experimental results describing a protein's amino acid composition are useful for estimating the mol...
A: Proteins are biological macromolecules composed of amino acids linked together by peptide bonds. The...
Q: Can the iodine test distinguish between amylose and amylopectin? What would you look for?
A: Carbohydrates are an essential source of energy. It is found in almost all foods and is largely used...
Q: A. Heteroglycans are polysaccharides with only one type of monosaccharide unit. Heparin is a heterog...
A: Polysaccharides that are comprised of same type of monosacharide units, then it is termed as homopol...
Step by step
Solved in 2 steps

- Given the equation of the line, y=150x-8.1221 (cells per mL is in 10^8), what is the CFU per mL of the original suspension if the thousand-fold dilution of that suspension has an absorbance of 0.3 at 600nm? * A. 3.7 x 10^12 B. 3.7 x 10^9 C. 3.7 x 10^1 D. None of these is correctC6H8 + O2 ------à CO2 + H2O How many grams of C6H8 are needed to from 72grams of H2O ?Why is Hydrogen peroxide toxic to cells in high concentrations?
- Why does eluent strength increase as solvent becomesless polar in reversed-phase chromatography, whereas eluentstrength increases as solvent becomes more polar in normal-phase chromatography? What kind of gradient is used in supercritical fluid chromatography?How many moles of Na2S2O3 would be required to react completely with 0.450 g of AgBr?What volume of 0.10NH2 SO3 will be required to neutralize a solution containing 10.0grams of Ca(OH)2?a) 0.27Lb) 27Lc) 2.7Ld) 270L What volume of 1.5N NaOH is needed to react with 25ml 4.0N HCl?a) 66.67mlb) 6.67mlc) 50mld) 70ml









