11 of 11 What volume (in uL to 1 decimal place) of stock SybrSafe solution (3.4 mg/mL) would you add to 120 mL of agarose solution to give a final concentration of 0.4 pg/mL? Answer:
11 of 11 What volume (in uL to 1 decimal place) of stock SybrSafe solution (3.4 mg/mL) would you add to 120 mL of agarose solution to give a final concentration of 0.4 pg/mL? Answer:
Chapter10: Reconstitution Of Powdered Drugs
Section: Chapter Questions
Problem 7.5P
Related questions
Question
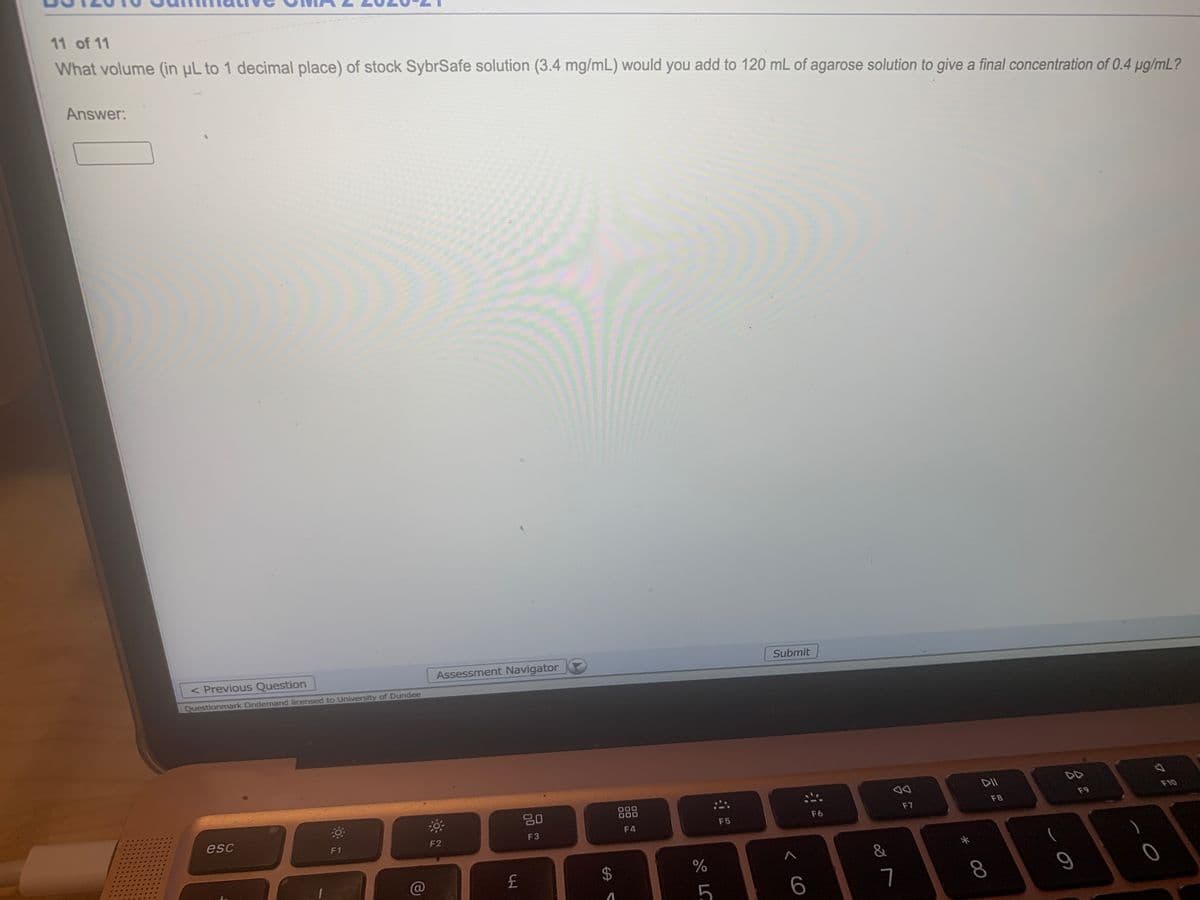
Transcribed Image Text:11 of 11
What volume (in µL to 1 decimal place) of stock SybrSafe solution (3.4 mg/mL) would you add to 120 mL of agarose solution to give a final concentration of 0.4 pg/mL?
Answer:
Submit
< Previous Question
Assessment Navigator
Questionmark Ondemand licensed to University of Dundee
DII
DD
80
000
000
F10
F8
F9
F7
esc
F5
F6
F3
F4
F1
F2
24
...
6
7
* 00
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you


Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage


Essentials of Pharmacology for Health Professions
Nursing
ISBN:
9781305441620
Author:
WOODROW
Publisher:
Cengage