Q: The table provides information about the solubility of sucrose. Temperature Solubility of Sucrose…
A: Given, Solubility of sucrose at 20°C is 230 g/100g H2O.
Q: The data below apply to the reaction. A > 2B+ C. If there are no B and C present initiany. t/h 3 12…
A: Formula used K= 2.303/t × log (a/a-x) where a = initial concentration t = time in hour x= amount of…
Q: A chemist wishing to do an experiment requiring 4' Cat (half-life = 4.5 days) needs 5.0 µg of the…
A: Given : Half life (t1/2) = 4.5 days Mass of 47Ca+2 required = 5.0 ug Time for delivery = 96 hour…
Q: Which substance gets reduced in: Pb(s) + PbO2(s) + 2H2SO4(aq) = 2PbSO4(s) + 2H2O(+)?
A: Given :- Pb(s) + PbO2(s) + 2H2SO4(aq) → 2PbSO4(s) + 2H2O(l) To identify :- reduced species
Q: *76. Using data from Table 16.3, calculate the concentrations of Hg²+(aq), HgCl*(aq), and HgCl2(aq)…
A: We have find out concentration of Hg2+, HgCl+ and HgCl2.
Q: REAGENT AMOUNTS In this section, you must record the actual amounts and volumes of reagens use in…
A:
Q: Table 1 Simple Molecules Molecule CH4 NH3 H2O CCl4 NCl3 Number of Bond Pairs…
A:
Q: How many moles of NaCl are in a 880-mL sample of 1.50M NaCl?
A: Given :- Molarity of NaCl solution = 1.50 M Volume of solution = 880 mL To calculate :- No.of…
Q: 2. In a titration experiment, 50mL of HCI was needed to completely neutralize 20mL of 0.25 M NaOH.…
A:
Q: 6.3. Calculate the higher and lower heating values of methyl alcohol, CH3OH, by using the following…
A:
Q: How will you prepare 3.5 mL of 45 mM hydrogen peroxide from a 600 mM stock
A: Dilution of the solution: When we dilute a concentrated solution with pure water, the mole of solute…
Q: 6. Suppose 1.0 x 102 grams of ice absorbs 1255 J of heat, reaching a final temperature of -2.0°C.…
A: Given: The mass of ice = 1.0×102 g The amount of heat absorbed by ice = 1255 J The final temperature…
Q: Draw the major product of the reaction. H3PO4 он heat Draw ALL of the beta hydrogens on the…
A:
Q: Calculate the De Broglie wavelength of a 1.50 ton ca x a speed of 12.0 m/s. h = 6.626 × 1034 J· sec,…
A:
Q: X Incorrect. Compute the standard free energy change for the following reaction: C2H4(g) + 302(g) →…
A:
Q: Pb(NO3)2 (aq) + Nal (aq)
A: Given :- Pb(NO3)2(aq) + NaI(aq) → To complete :- Given reaction
Q: prepare 0.1 M and 0.1 N solution of sodium thiosulphate in 100 ml of volumetric flask.
A: Molarity is the number of moles of solutes present in one liter of solution while normality is the…
Q: What amino acid residues interact with methylphenidate in DAT and NET protein active sites? please…
A: The three human monoamine transporters (MATs) i.e. the dopamine transporter (DAT), the serotonin…
Q: How is the pH scale of acidity/basicity described? It's a vector function of the [H+]. It's a…
A:
Q: I am following this procedure to extract ibuprofen from tablets and was wondering if someone could…
A: Ibuprofen have Functional Group which are acidic in nature ( carboxylic Acid )
Q: What volume in mL of O2 gas collected over water is obtained from heating 852.7 mg KClO3 (122.55) at…
A: Given- Pressure =1atm Weight of KClO3=852.7mg=0 .5427g Temperature= 25°C=(273+25)K=298K
Q: Methanol is known to be the simplest alcohol, and is used as an antifreeze agent, a solvent, a fuel…
A:
Q: Identify the type of chemical reaction
A:
Q: 3. What is the mass of a silver coin if 5.2 J is required to raise its temperature from 21.0 to 25.0…
A:
Q: solution with [H+]= 1 x 10-3 M has pH . Is this solution an acid, base, or neutral? A…
A: Given-> [H+] = 1× 10-3 pH =-log[H+] = -log(1× 10-3) = -log10-3 = 3log 10 = 3 pH =3<7 =>…
Q: same thermodynamic properties as graphite. Substance AH°(kJ/mol) S°(J/(mol*K)) C(graphite) 0 5.7…
A:
Q: basketball is inflated to a pressure of 1.75 atm in a garage at a temperature of 18.0°C. What will…
A:
Q: What is the maximum amount of electrons that can be placed in the 7th shell? A 14 B 49 98 D 140…
A: Here, we have to find the maximum number of electrons that can be placed in the 7th shell.
Q: Bond H-O H-S H-Te i Energy (kJ/mol) 458.9 i 363 238 There is a trend + : moving down the column, the…
A: Bond energy: It is the amount of energy require to break the bond.
Q: Identify which of the following are carbohydrates. Check all that apply. • View Available Hint(s) OH…
A: We have to predict the carbohydrates among given choices .
Q: Draw an electron pushing mechanism for proline racemization. Draw a reaction coordinate diagram of…
A:
Q: same volume of ethyl ether vapor to pass through the same hole? b) What volume of chlorine will…
A: Molar mass of O2(M1) = 32 g Molar mass of ethyl ether(M2) = 74 Rate of effusion = Volume…
Q: Chemistry Compound CS2 has a dipole moment u=0 1.Write the type of compound that CS2 is (ionic,…
A: Ionic compound is generated by two opposite ions where covalent molecule is formed by sharing of…
Q: The reaction A(aq) + B(aq) C(aq) + D(aq) has a ΔHrxn = -85 kJ/mol. This reaction is…
A:
Q: How is 250 mL of 0.5M CsCl prepared? MW: Cs: 132.91 g/mol; Cl: 35.45 g/mol
A: Given, Molarity of CsCl solution = 0.5 M = 0.5 mol/L Volume of the solution = 250 mL = 0.250 L…
Q: the tocal heat energD required or produced when a salt n.soop disolves in water s pven by agtotal…
A: The heat of the solution of 0.500 g of salt is = 1000.01 J The molar mass of the salt is = 24.305…
Q: sources of error of determination of iron in iron ore
A: Impurities in the iron from the Blast Furnace include carbon, sulphur, phosphorus and silicon. These…
Q: Estimate AG° at -77°C for the following reaction. 302(g) → 203(g) AG° = i kJ eTextbook and Media…
A:
Q: Concentration of NaOH in HCl is 2.57% by mass. Density of HCl is 1.2g/cc. Determine molality of…
A: Relationship between %by mass and molality m=%by weight×1000/Molar mass of NaOH×(100−%by weight)…
Q: Reler to the phase diagram below when answenng the questions on this worksheet. Begin by adding S,…
A: A substance can exist in 3 different phases- solid,liquid,gas.Phase diagram describes effect of…
Q: How does the buffer react to the addition of acids and bases? In comparison, how does water react to…
A: Buffer solutions are aqueous solutions that resist the change in pH on addition of small amount of…
Q: Consider the three flasks in the diagram below. What is the partial pressure of each gas and the…
A: Given : For He gas : Volume = 1.0 L Pressure = 200 torr For Ne gas : Volume = 1.0 L Pressure =…
Q: Why can’t an organic molecule have the formula C2H7?
A: The bonding in main-group elements (especially carbon, nitrogen, oxygen, and halogens,) can be…
Q: 1. Draw the complete structural formula of the following: 3-Ethylpropene 2,3-Cyclohexadiene…
A: structural formula : Structural formula can be defined as the arrangements of the atom in the…
Q: Calculate the pH of a 0.105 M solution of ethylenediamine (H,NCH,CH,NH, ). The pKa values for the…
A:
Q: A two-liter sample of mineral water was evaporated to a small volume, following which the potassium…
A:
Q: Suggest two possible structures of an unsaturated ketone that will be produced from the…
A: IR spectra deals results from the interaction of infrared radiation with matter by absorption. IR…
Q: Strike anywhere" matches contain P4S3, a compound that ignites when heated by friction. It reacts…
A: Given- P4S3(s)+8O2(g) ->P4O10(s)+3SO2(g) ∆H°reaction=-3677kJ ->Answer may differ slightly…
Q: Bud N. Chemist mixes 125.0 mL of 0.0250 M sodium phosphate (aq) with 75.0 mL of 0.0500 M calcium…
A:
Q: A 0.4126-g sample of primary-standard Na₂CO₃ was treated with 40.00 mL of dilute perchloric acid.…
A:
Draw the structure of a wax formed from stearic acid and the alcohol.
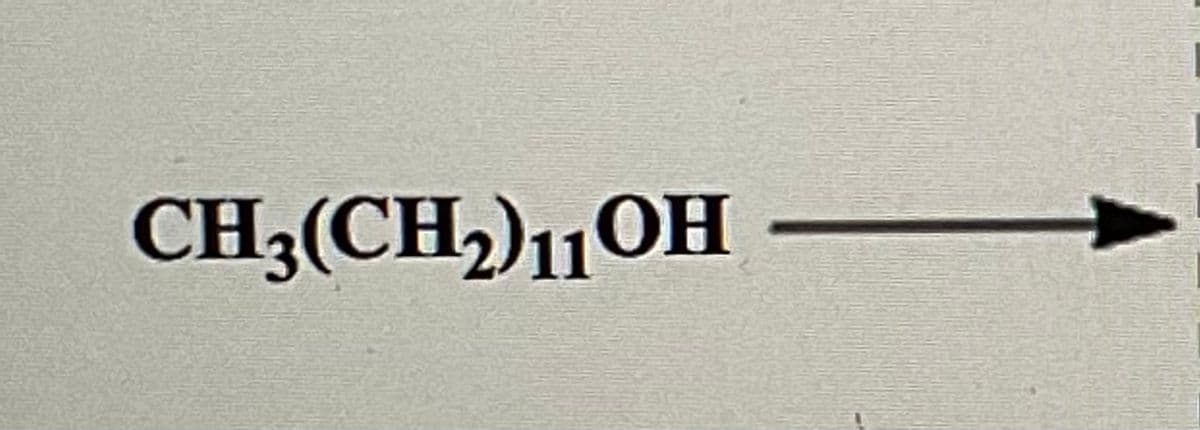
Step by step
Solved in 2 steps with 1 images


