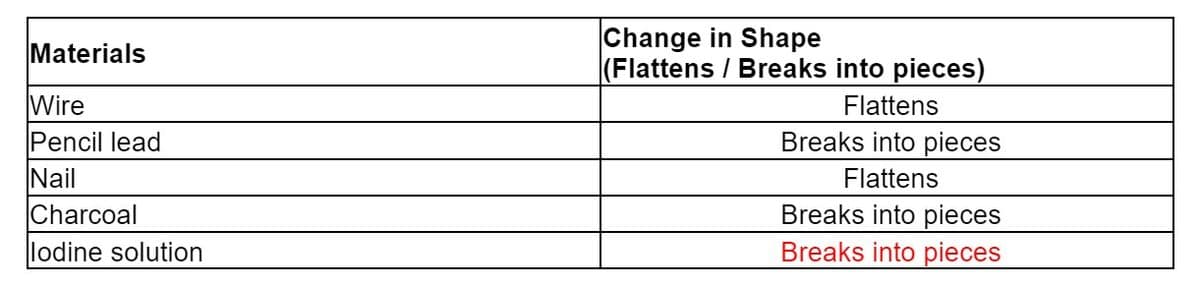
Change in Shape (Flattens / Breaks into pieces) Materials Wire Flattens Pencil lead Breaks into pieces Nail Flattens Charcoal Breaks into pieces Breaks into pieces lodine solution
Q: Part B. Below are chemical equations, by inspection fill in the missing coefficient on the blank and...
A: We need to count the atoms. We have to make sure to balance the number of each individual atom is ba...
Q: Salt bridge A concentration cell similar to the one shown is composed of two Ni electrodes and solut...
A:
Q: functional groups
A:
Q: Compare Lavoisier's definition of acids to the modern one. How did his fall short? Why was his defin...
A: Lavoisier definition : Lavoisier hypothesized that the compounds which contain oxygen as a constitue...
Q: 1. CI AICI, followed by hydrolysis 2. CI AICI, 3. NH,NH2 followed by NaOH (heat) O:
A: ->benzene give electrophilic aromatic substitution reaction. ->NH2-NH2 used for reduction of c...
Q: Use standard reduction potentials to calculate the equilibrium constant for the reaction: Pb2+(aq) +...
A: Pb^2+(aq) + Sn(s) -> Pb(s) + Sn^2+(aq)
Q: Nitrogen (N,) gas and hydrogen (H,) gas react to form ammonia (NH,) gas. Suppose you have 13.0 mol o...
A: Nitrogen reacts with hydrogen to form ammonia. The equation for the balanced chemical reaction is gi...
Q: Find the H30* in a Sauvignon Blanc with a pH of 3.25. ? .... [H3 O]+ = M Submit Request Answer
A: pH = 3.25
Q: A buffer solution is 0.500 M in acetic acid and 0.500 M in sodium acetate. Its pH is 4.74. What is i...
A:
Q: Using Table 2.1, calculate the required amount (in g) of ammonium sulfate to bring the solution from...
A: A numerical problem based on concentration terms that is to be accomplished.
Q: Answer no.2 only show complete based from the book, if y’ll gonna upload a photo make sure it is cle...
A: We know that for a reaction is to be spontaneous., If ΔG<0, the process occurs spontaneously. If ...
Q: 9. What is the normality of a solution containing 150g of HCI in a 4.5L solution? MW- H:1, Cl=35
A: Normality is number of gram equivalents presents in 1 liter of solution called normality Molar mas...
Q: C4) Write the molecular, complete ionic, and net ionic equations for the reaction between nickel chl...
A: The reaction between nickel chloride and sodium carbonate is : a) Molecular
Q: The Lewis representation above depicts a reaction between a halogen (blue) and a main-group eleme. g...
A:
Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gas...
A:
Q: Classify each of the following acids as strong or weak. Drag the appropriate items to their respecti...
A: The strong acid are easily dissociates in water and release Hydronium ion along with their conjugate...
Q: Which of the following is a BrØnsted-Lowry acid? O C Cl 4 Ο ΝΗ O PH, Ο ΝΗ5 O Br 2
A:
Q: What major product (from Figure #15) results from the following ester hydrolysis reaction (from Reac...
A:
Q: [OH-] = 1.0 x 10-7 Express the concentration to two significant figures and include the appropriate ...
A: Given-> [OH^-] = 1.0 × 10^-7 M If [H3O^+] is 1.0 × 10^-7 M => then it is neutral .
Q: Isotonic saline solution is 0.154 M NaCl(aq). What is the solubility of AgCl (Ksp = 1.8 × 10−10) in ...
A: Given data : Concentration of NaCl = 0.154 M Ksp of AgCl = 1.8 × 10-10
Q: 5. What masses of KOH and water are needed to make 1500g of 20% solution? MW- K:39, O:16, H:1
A: Weight of solution = 1500 gm Percentage = 20%
Q: There is a difference in acidity of the hydrogens labeled red below. Select all the distinguishing f...
A:
Q: 3. Compute the percentage adequacy of the CHO, CHON and Fats ? % Adequacy of CHO = Total CHO / Total...
A: Given: Total calories = 1552.2 kcal CHO = 232.83 g CHON= 58.20 g FATS= 43.11 g
Q: The following figure represents the emission spectrum of a hydrogenlike ion (Z is not 1 but has one ...
A:
Q: Let's spend some time with this reaction of NO2, an important compound in atmospheric chemistry (it'...
A: Ea (Activation Energy) is the amount of energy required by the reactant to produce an appropriate pr...
Q: 6. How much mL of Acetone is needed to make 100mL of an aqueous 30% (v/v) Acetone solution? How much...
A: Volume/volume percentage is a measure of concentration of the solution, which is the ratio of volume...
Q: If you have 72 g of sodium sulfate, calculate the sodium, sulfur, and oxygen atoms that are present ...
A: Formula of sodium sulfate - Na2SO4 Molar mass of sodium sulfate - 142.04 g/mol Avogadro's constant -...
Q: Consider a 1.00 L of 1.50x10-3 M hydrocyanic acid (HCN, Ka=4.9x10-10). Write the equilibrium expres...
A: Answer: In this question we have to perform the calculation for two types of solution: 1. solution o...
Q: Name the following isomer H.C CH3 2. 3. H.
A:
Q: Nitric acid (HNO3) is a strong acid that is one contributor to acid rain. The precursor e nitrogen d...
A: 1m3=1000L 1µg= 1×10^-6gm In the above question, HNO3 content is 6.8 µg/m3 it is in 0.30 g=0.30×1000 ...
Q: For the following reaction, 6.28 grams of iron are mixed with excess oxygen gas . The reaction yield...
A:
Q: Question 22 Match the correct response v NANO3 A. a basic salt NH4CI B. an acidic salt v NH4CIO4 C. ...
A:
Q: Write an equation to show how HC204 can act as a base with HS¯ acting as an acid. Express your answe...
A:
Q: 2. 1.00 mol of oxygen and trioxide gas is formed. At trioxide have been formed
A: Given Reaction:- SO2 + O2 -----> SO3(g) Initial concentration concentration of SO2 = 1.00 mole ...
Q: Individual solutions of Ba(OH)2 and H2SO4 both conduct electricity, but the conductivity disappears ...
A: The strong electrolytes are dissociated completely on all concentrations. The conductivity of an ele...
Q: [OH-] = 2.3 × 10-10 Express the concentration to two significant figures and include the appropriate...
A:
Q: Use standard reduction potentials to calculate the equilibrium constant for the reaction: Co2+(aq) +...
A: Standard electrode potential value of the given cells are as follows E°Cu2+/Cu =0.337 V and E°Co2+/C...
Q: Calculate the percentage concentration of a solution that contains 20 grams of sodium hydroxide, NaO...
A: The mass of solute in gram present in 100 mL of solution is known as the mass percent concentration ...
Q: Draw the major product from this reaction. Use wedge and dash bonds to indicate relative stereochemi...
A: We have to draw the major organic product of the given reaction.
Q: Calculate the p-value for each ion a. Na+, Cl-, and OH- in a solution that is 0.0345 M in NaCl and 0...
A:
Q: The illustration to the left represents a mixture of nitrogen ( blue ) and oxygen ( red ) molecules....
A: The balanced chemical equation fot the reaction of Nitrogen gas and oxygen gas is, N2 + 2 ...
Q: ELEMENT X HAS AN ATOMIC NUMBER OF 9 1. How many atomic orbitabls does atom X have? 2. How many she...
A: Given, Atomic no. = 9 then,
Q: Use standard reduction potentials to calculate the equilibrium constant for the reaction: Hg2+(aq) +...
A: From Nernst equation Ecell = Eocell - 0.0591/n (logQ) at equilibrium Ecell = 0 Q = Kc Q = react...
Q: 57.3 g of Y(g) (molar mass = 26.92 g/mol) is placed in a 0.688 L flask for the following reaction to...
A:
Q: 1. Dmitri Mendeleev was regarded as the Father of the Modern Periodic Table; did you think it is Hen...
A: Since you have asked multiple question, we will solve the first question for you. If you want any sp...
Q: Which of the following rate laws would be classified as 2nd order? Rate = [A]^2 Rate = k[NO]^2 [O2} ...
A:
Q: Use the References to access important values if needed for this question. When the Cu2+ concentrati...
A:
Q: Suppose a 250. mL flask is filled with 0.70 mol of H, and 0.50 mol of HCI. The following reaction be...
A:
Q: 3.) Alkanes Alkenes Alkynes Alcohol Ether Aldehyde Ketone Carboxylic acid Ester Aromatic Hydrocarbon...
A:
Q: Salt bridge A concentration cell similar to the one shown is composed of two Ni electrodes and solut...
A:
Kindly check if my table is correct. Thanks.
Step by step
Solved in 2 steps with 2 images

- Dr Kootsookos was cooking breakfast one day and the saucepan handle got hot and crumbled in her hand. The saucepan handle was probably: a.) made of a thermoplastic b.) made of a thermoset c.) made of an elastomer d.) made of a ceramicWhat is the meaning of the safety symbol in Figure 10? * A- It indicates that a substance is flammable. B- It indicates that a substance is explosive. C- It indicates that a substance is an oxidiser. D- It indicates that a substance is shock sensitive. E- None of the options are correct.Explain the packaging listed below:- Compact cubic structure;- compact packaging(HCP)- Cubic- Octahedral- Tetrahedral
- What are the differences between crazes vs. cracks in polymer? Can you still use polymeric materials if there are crazes or cracks occurs?In the production of printed circuit boards for the electronics industry, a 0.320 mm layer of copper is laminated onto an insulating plastic board. Next a circuit pattern made of a chemically resistant polymer is printed on the board. The unwanted copper is removed by chemical etching and the protective polymer is finally removed by solvents. One etching reaction isCu(NH3)4Cl2(aq) + 4NH3(aq) + Cu(s) → 2Cu(NH3)4Cl(aq)A plant needs to manufacture 12000 printed circuit boards, each 7.75cm x 16.5cm in area. An average of 78.0% of the copper is removed from each board (density of copper=8.96g/cm3.) What mass of Cu(NH3)4Cl2 reagent is required?Most plastics are made using chemicals extracted from oil. Currently, only a small fraction of plastic waste is recycled. Describe at least two ways in which an improvement in plastics recycling benefits the environment.
- Nanostructures are from the breaking up of bulk materials a. The top down approach b. The bottom up approach c. The bottom down approach d. The top up approachWHICH FORM OF IRON WILL YIELD A HIGHER DENSITY : FCC OR BCC? IRON HAS AN ATOMIC RADIUS OF 0.124 NMExplain why Styrofoam a good is insulating material. Select one: a. Styrofoam is made of a polymer with high molecular weight. b. Styrofoam is plastic. c. Styrofoam is a polystyrene polymer that has trapped bubbles of air inside its macroscopic structure, and air is a good insulator. d. All options are correct.

