Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter11: Intermolecular Forces And Liquids
Section11.6: Properties Of Liquids
Problem 2.2ACP
Related questions
Question
Using Table 2.1, calculate the required amount (in g) of ammonium sulfate to bring the solution from 40% to 60% saturation.
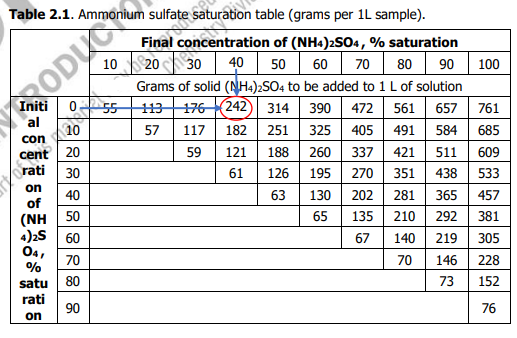
Transcribed Image Text:Table 2.1. Ammonium
table (grams per 1L sample).
40 50 60 70 80 90
Grams of solid (NH4)2SO4 to be added to 1L of solution
176(242 314 390 472 561 657 761
117 182| 251 325| 405 491 584 | 685
10
30
100
ODUO
55
57
20
59
121
188 260
337 421
511
609
30
61
126 195
270 | 351
438
533
40
63
130
202
281
365
457
(NH 50
4)2s 60
65
135 210
292
381
67
140
219
305
70
70
146 228
73
152
90
76
8 888g88
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning