Chapter 17 Multiple Choice Question 39 eview | Con Part A A 100.0 ml sample of 0.10 M NH3 is titrated with 0.10 M HNO3. Determine the pH of the solution after the addition of 50.0 mL of HNO3. The K, of NH3 is 1.8 x 105. O 9.26 O 7.78 O 7.05 O 4.74 O 10.34
Chapter 17 Multiple Choice Question 39 eview | Con Part A A 100.0 ml sample of 0.10 M NH3 is titrated with 0.10 M HNO3. Determine the pH of the solution after the addition of 50.0 mL of HNO3. The K, of NH3 is 1.8 x 105. O 9.26 O 7.78 O 7.05 O 4.74 O 10.34
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter16: Solubility And Complex Ion Equilibria
Section: Chapter Questions
Problem 52E: The concentration of Mg2+ in seawater is 0.052 M. At what pH will 99% of the Mg2+ be precipitated as...
Related questions
Question
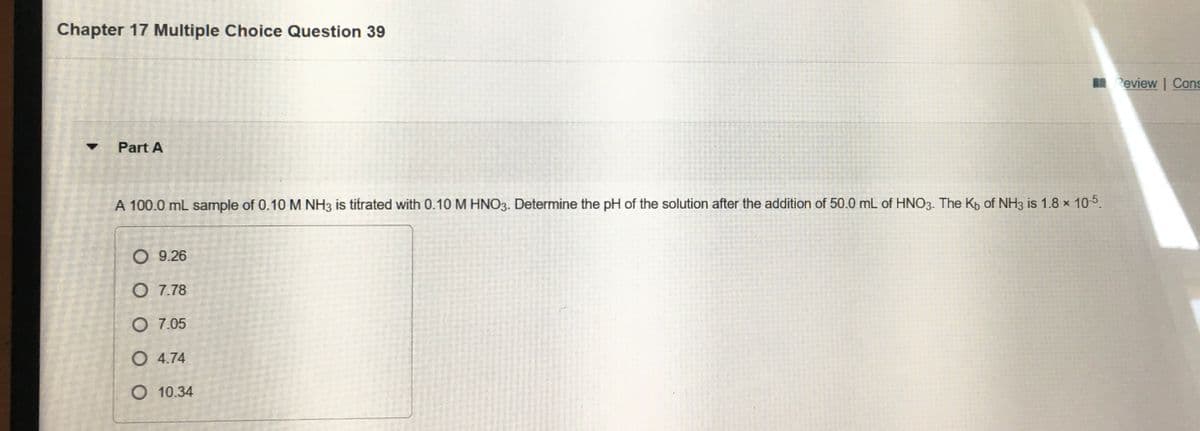
Transcribed Image Text:Chapter 17 Multiple Choice Question 39
I Review | Cons
Part A
A 100.0 mL sample of 0.10 M NH3 is titrated with 0.10 M HNO3. Determine the pH of the solution after the addition of 50.0 mL of HNO3. The Kp of NH3 is 1.8 × 105.
O 9.26
O 7.78
O 7.05
O 4.74
O 10.34
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning