Q: NH он d) O,N- -NH2 -NH2 O,N- -NH2 -NH2 CH3
A: Bases are defined as the molecules which can donate it's electron pair. The more easily a molecule…
Q: What is the conjugate base of HSO4- ? O H2SO4 O SO4 O HSO4 O S042- O H2SO42-
A: HSO4- + H2O ⇔ SO42- + H3O+
Q: List the following alcohols in order from strongest acid to weakest acid. CH2=CHCH, OH, CH3 CH2 CH,…
A:
Q: H2 NH2 NH3 -NH2 -NH2 с. O,N CH d. CgH;NH, (CHs)2NH -NH2
A: In case of nitrogenous compound greater the availability of lone pair on nitrogen atom greater it's…
Q: please help?
A: Here we can decide by using electronegativity factor, Oxygen have highest electronegativity then…
Q: OH- + CH3COOH ==== CH3COO- + H2O . Which of the products is a conjugate base?
A: Given :- OH- + CH3COOH → CH3COO- + H2O To identify :- Conjugate base
Q: Which C-H bonds in the following molecules are acidic because the resulting conjugate base is…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Rank the following compounds from strongest acid to weakest acid
A: The presence of electron withdrawing group in parent acid increases its acidity. The reason is that…
Q: Which of the following is most weakly basic? O CH3CO2 О но- O CH3CH2O¯ O CH3O"
A: A substance which accepts a proton is known as base. The substance which is formed after the…
Q: Rank the compounds in each group in order of increasing acidity.
A: The given compounds are represented as follows:
Q: Rank the following compounds in order of increasing acidity of their most acidic hydrogen atom.…
A: Acidic character of an organic compound depends on the stability of its conjugate base. If an…
Q: Which of the following choices is a Brønsted acid/base conjugate pair? HClO3 / Cl- HCl /…
A: A bronsted acid is the species which can easily lose proton while the bronsted base is the species…
Q: Rank the following organic compounds below in terms of increasing acidity, with 1 being the least…
A: Here the acidity is dependent on the ease of donation of hydrogen ion.
Q: Which is the conjugate base? C6H5NH2+H2O ↔ C6H5NH3+OH-
A:
Q: What is the conjugate base of HSO4" ? O H₂SO4²- O HSO3²- O HSO4²- O H₂SO4 O SO4²-
A: What is conjugate base of HSO4- ?
Q: What indicated hydrogen atom is the most acidic? B HAA H₂ of C CH3 D
A: We have to select the most acidic H
Q: 5. Rank each of the five compounds by relative basicity. Use a "1" for the strongest base, a "2" for…
A: Given compounds: We have to rank each of these compounds by relative basicity.
Q: Rank the compounds in each group in order of increasing acidity. Br HOT OH OH a. b. СООН соон COOH…
A: Acidic strength is directly proportional to stability of conjugate base. Acidic strength is directly…
Q: In equilibrium, label the stronger acid, the stronger base, the weaker acid, and the weaker base.…
A: Given:
Q: What is the conjugate acid of HCO3 ? O A HCO3* в. Соз2- OC. CO3- D. H2CO3 O E. HCO32-
A: Bicarbonate anion is a conjugate base of carbonic acid. D option.
Q: он SO3H COOH SO3H ČH3 ČH3 NO2 ČH3 III IV I II
A:
Q: Please rank the following molecules in terms of their basicity. Assign 1 to the most basic molecule,…
A: The compounds given are,
Q: Rank the following compounds in order of increasing basicity. The first compound in your ranking…
A: The given compounds are the conjugate bases of the acid. Now, the more stable conjugate base would…
Q: Which of the following has significant acid properties in water? CH3Cl CH4 HF NH3
A: To determine which of the following is more acidic in water. 1. CH3Cl 2. CH43. HF 4. NH3
Q: Rank the following groups in order of decreasing priority: A.-COOH В.-Н C.-NH, D. -OH А В C D…
A: According to the CIP rule, the first attachment to chiral carbon will be assigned priority according…
Q: Which compound is considered as the conjugate base? * D. OH- B. C2H5NH+3 C. H2O A. C2H5NH2
A: Removal of H+ formed conjugate base. So,
Q: which of the following is a base? and why? - KCl -NH3 - HNO3 - CH3OH
A:
Q: Q4. The following compound can become protonated on any of the three nitrogen atoms. Which nitrogen…
A: The most basic nitrogen among 1, 2, 3 has to be identified
Q: 1. Which of the following molecules are polar? Which are nonpolar? (c) PFs (d) CFB13 (а) BFs (b)…
A: Dear student, I cannot answer other questions as it is not in consonace with bartleby guidelines. If…
Q: Rank the following compounds in order of increasing basicity. CH3 H3C CH3 H3C 00 KO 11 CH3 CH3 IV A)…
A:
Q: Name the following molecules, then rank their basicities in decreasing order (1 = most basic, 3 =…
A: Amines are the organic compounds with –NH2 as functional group. On the basis of number of R- groups,…
Q: Rank in order of increasing acidity: CI .CO2H .CO2H .CO2H CO2H CO2H .CH2OH CI
A: The acidity of any compound is the power to dissociate as more as protons in a free form in the…
Q: Rank the following compounds in order of increasing basicity: NH₂ NH₂ NH₂ NH₂ A) B) D) OCH3 NO₂ 2
A:
Q: Which of the following is the conjugate base of the compound shown below? H H c=C H H H A) H-c-co D)…
A: Answer 3: Introduction: The acid or base form after the losing or gaining a proton is called…
Q: CH3 -N 'H O,N- -NH2 NH2 CI - NH2 CH30- NH2
A: Basicties of aniline are because of donating property of lone pair of electrons of nitrogen
Q: What is the correct arrangement in increasing basicity of the given compounds? III < I < II III <…
A:
Q: H2SO4 + NH3 ==== HSO4- + NH4+ . Which of the products is a conjugate base?
A: Which of the products is a conjugate base?
Q: Rank the following compounds from strongest acid to weakest acid
A: pKa value for the given compounds are,
Q: What is the conjugate acid of HСОЗ ? O A. HCO32- O B. H2CO3 В. OC. CO3- O D. CO32- O E. HCO3+
A: conjugated acid is substance that is form when a base receive a proton ( H+ )
Q: What is the order of the compounds from most acidic to the least acidic? O CHCHCOH:CHCHC-CDH:…
A: Acidity is the ease with which hydrogen ion is released
Q: Arrange the following compounds in increasing order in terms of acidity. CH3CH2SH, PhSH, RSO3H
A: Acidity is defined as the ease with which a molecule can donate protons. if it can donate PROTONS…
Q: Draw the conjugate base of the following compounds e. NH3 f. CH3CH2OH g. CH;COOH h. CH3CH2NH2 а. НBr…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: е. NH2 a. N- 'N d. N. b. H C.
A: Basicity of Nitrogen depends upon the delocalisation or movement of lone pair of Nitrogen as…
Q: Arrange the four compounds in increasing order according to the acidity of conjugate acid. (CH3)2NH…
A: Given Acidic order
Q: Q3. Circle the stronger acid in the following pairs, and explain your answer. H.C-C-OH CF,CH,-OH…
A: ->We can identify stronger acid if the conjugate base is more stablized. And conjugate base is…
Q: Which of the circled hydrogen atoms is the most acidic? H (H) H-¢O H,C-¢O H,C-¢ H,C=¢ H,C-C=c<H II…
A: When hydrogen atom leave the compound then compound convert to anion and more stable is the anion…
Q: Rank the following species in order of increasing acidity. Explain your reasons for ordering them as…
A: Species in order of increasing acidity : NH3 < H2O < CH3OH < CH3COOH < HF <…
Q: Circle strongest acid and cross out the weakest. HCOOH HSO4- CH3NH3+ Circle…
A: The equilibrium favors the reaction that generally moves the proton (H+) to the stronger base in an…
Q: N H2NNH2 C N NH3 CEN CI C ΝΗ NH₂OH CH3 Q N CH3
A: Given, Rank each of the following sets of nitrogen bases in terms of basicity and explain your…
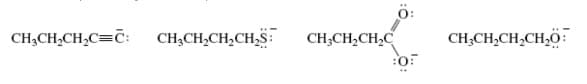
Step by step
Solved in 2 steps with 2 images

- What reagent is necessary to complete the reaction? CH3-CH2-CH-C-OH ? CHỊCH,CH C-0- Na* CH3 CH3 NaO O NaCl O Na O NaOH + H₂ODraw the product obtained when cis-2-butene is treated first with Br2 in CH2Cl2, second with NaNH2 in NH3, and then finally with Li in NH3.reactions and products for C9H8O4 C9H8O4 + O2 --> C9H8O4 + H2O --> C9H8O4 + HCl --> C9H8O4 + OH- --> C9H8O4 + Na --> C9H8O4 + F -->
- How many carbons and hydrogens does 2-Cyclohexyl-2-methyl-butan-1-ol contain?Account for the regioselectivity and stereoselectivity observed when 1-methylcyclopentene is treated with reagent Q.Hg(OAc)2 in H2O(a) What product(s) are formed when the E isomer of C6H5CH = CHC6H5 is treated with Br2, followed by one equivalent of KOH? Label the resulting alkene(s) as E or Z. (b) What product(s) are formed when the Z isomer of C6H5CH = CHC6H5 is subjected to the same reaction sequence? (c) How are the compounds in parts (a) and (b) related to each other?
- Give a common name (when possible) and a systematic name for each compound.(a) CH3OCH“CH2Oximene and myrcene, two hydrocarbons isolated from alfalfa that have the molecular formula C10H16, both yield 2,6- dimethyloctane when treated with H2 and a Pd catalyst. Ozonolysis of oximene forms (CH3)2C = O, CH2 = O, CH2(CHO)2, and CH3COCHO. Ozonolysis of myrcene yields (CH3)2C = O, CH2 = O, (two equiv), and HCOCH2CH2COCHO. Identify the structures of oximene and myrcene.Draw the products, including their configurations, obtained from the reaction of 1-ethylcyclohexene with the following reagents:a. HBr b. H2, Pd/C c. R2BH/THF, followed by HO– , H2O2, H2O d. Br2/CH2Cl2
- Rank the following groups in order of decreasing priority. −C≡CH, −CH(CH3)2, −CH2CH3, −CH=CH2Account for the regioselectivity and stereoselectivity observed when 1-methylcyclopentene is treated with reagent. Q) Br2 in H2OIndicate what to do to obtain the compund (cyclohexylidenemethyl)cyclohexane from (C6H5)3P=CHCH2CH3



