Chemistry A student measured concentrations of potassium ions in the solutions using flame photometry and calibration curve was plotted. From the calibration curve shown below, answer the question. .. Calibration Curve of Student 120 y = 1.9943x + 0.8095 R - 0.999 100 80 60 40 20 20 40 60 Concentration of Potassium ions (ppm) The instrument response for student sample was 75, calculate the concentration of potassium ions in the solution based on the findings of the student. 72.20 ppm B 38.01 ppm 27 ppm 37 ppm Instrument Response
Chemistry A student measured concentrations of potassium ions in the solutions using flame photometry and calibration curve was plotted. From the calibration curve shown below, answer the question. .. Calibration Curve of Student 120 y = 1.9943x + 0.8095 R - 0.999 100 80 60 40 20 20 40 60 Concentration of Potassium ions (ppm) The instrument response for student sample was 75, calculate the concentration of potassium ions in the solution based on the findings of the student. 72.20 ppm B 38.01 ppm 27 ppm 37 ppm Instrument Response
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.13QAP
Related questions
Question
pls solve this problem with in 15 nto 30 min. I will give multiple ?. Explaination if necessary.
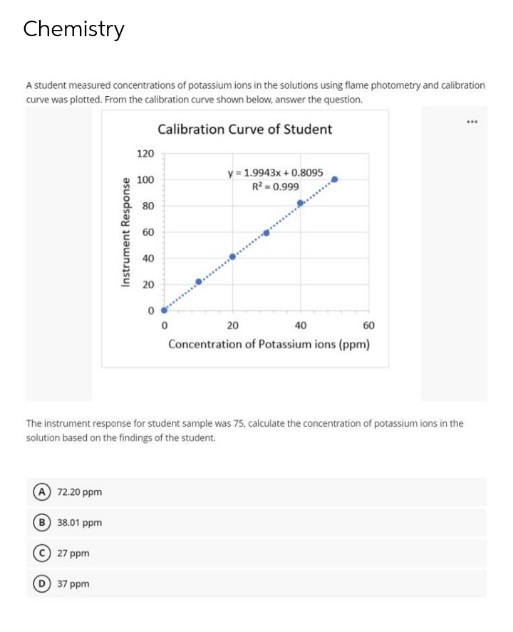
Transcribed Image Text:Chemistry
A student measured concentrations of potassium ions in the solutions using flame photometry and calibration
curve was plotted. From the calibration curve shown below, answer the question.
...
Calibration Curve of Student
120
y = 1.9943x + 0.8095
R - 0.999
100
80
60
40
20
20
40
60
Concentration of Potassium ions (ppm)
The instrument response for student sample was 75, calculate the concentration of potassium ions in the
solution based on the findings of the student.
72.20 ppm
B 38.01 ppm
27 ppm
37 ppm
Instrument Response
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning