Q: 1. Calculate AH for the following reactions, using AH°f values from your text or the internet. a. 2…
A: Enthalpy of reaction is equal to the difference between the enthalpy of product to the enthalpy of…
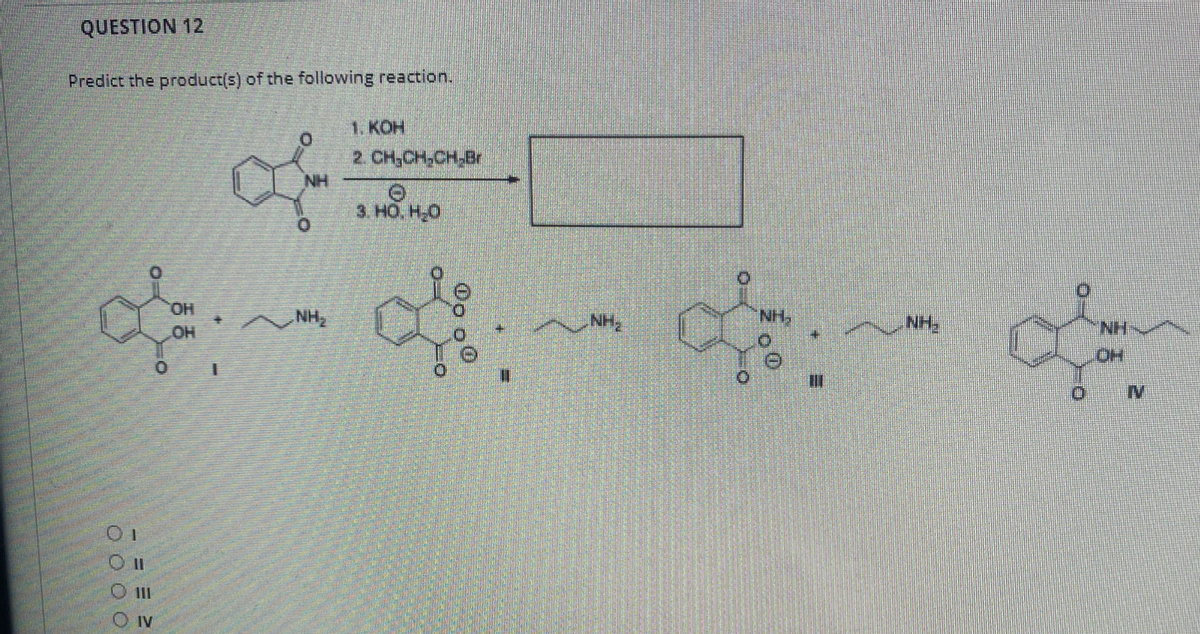
Q: Question 10 What is the product of the following reaction? 1. OsO4 2. NalO4 3. NaS203, H20 OH OH OH…
A:
Q: QUESTION 4 Predict the product for the following reaction: excess HOCH,CH,CH,OH H,SO, но OH
A: Given reaction is acetalisation reaction.
Q: O: H3C 1 mola CH3 H*
A:
Q: Give the major product(s) of the following reaction. 1) DIBAH (-78°C) (0.5 moles) 2) H;0*
A:
Q: For the reaction shown below, what is the value of n in the product? CEC-CH excess HCI CgH15Cln…
A: The given reaction is electrophilic addition across alkene and alkyne The details solution for this…
Q: Question 6 AUsing IR. how could you tell if the reactions below were complete? B) Using IR. Could…
A: A) In the first reaction, the reactant consists of C≡C, thus it will show a peak in the IR spectrum…
Q: 2. Write the name(s) of the product(s) for the following reactions. If no reaction can occur, write…
A: Here we are required to find the product of the reaction.
Q: 1. Which compound(s) represent the product(s) in the following equation?: C,H, + 50, H,0+ 3Co, A.…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: Calculate % yield: Based on amount of butylbromide used ____________
A: 1mole of butyl bromide gives 1 mole of product.
Q: HỒ 1) NaH (2 equiv) он OH 2) CH31 (2 equiv)
A: Solution : Williamson Ether synthesis is very important for the synthesis of symmetrical and…
Q: Molecular weight (g/mol) Literature MP ("C) Density (g/ml.) Amount Used (g or ml) Amount Used (mol)…
A: Amount used in moles is calculated using the equation Number of moles =Amount used (g)Molecular…
Q: What would be the best starting material for the following chemical conversion? 1. CH3COCl in…
A: Introduction :- O-nitroaniline is orange solid. It has melting point 72 °C…
Q: Consider the reactions below. In which cases is product formation favored by decreased temperature?…
A: When energy is released in an exothermic reaction, the temperature of the reaction mixture…
Q: You plan to synthesize your favorite ester Isoamyl acetate (banana smell) during your next lab…
A: A question based on equilibrium concept that is to be accomplished.
Q: What is the product of the reaction series shown below? 1. NaOEt 2. 3. H,0, H*, heat A. B. HO. он С.…
A: We have reaction of our reactant with base NaOEt, cyclohexenone and then acidic hydrolysis. We have…
Q: The product of the following reaction is : H. OH (Nat, OH-V ACETONE H. OH H H Na H. Br H. Он но H.…
A: SN2 is substitution nucleophilic biomolecular reaction.
Q: Question 21 Draw the product(s) for the following reaction. Upload your images as one file on the…
A:
Q: Study the reactions. 1. C2H2 (g) + 2H2 (g) → C2H6 (9) 2. C2H4 (9) + H2 (g) → C2H6 (9) 3. C2H2 (g) +…
A:
Q: PCC oxidation KMnO, H20 но CH,OH H" COOH SO, conc H2SO.
A: 1. The reaction of alcohol with PCC is an oxidation reaction in which milder oxidizing agent PCC…
Q: QUESTION 2 For which of the following reactions would the AH° for the reaction be equal to the AH°F…
A: Delta Hrxn = Hf (products ) - Hf (reactant ) For the condition given above : Hf (reactants) = 0…
Q: What is the main product of the reaction shown? 1) CH,MgBr 2) H,0 3) H,SO,, heat Br OH Br
A: When a ketone reacts with grignard reagent ot gives a secondary alcohol.
Q: What is the product? 1) Excess BH,:THF 2) OH',H,O, 3) РСС 4) CH;CH,CH,P(Ph);
A: Hydroboration-oxidation is the addition of borane across the double bond followed by oxidation using…
Q: What is (are) the product(s) of the following reaction? 1. LDA in -78°C 2. EtBr 1. Name the product…
A: In step-1, LDA deprotonates at beta-position of ketone to form an enolate ion. In step-2, -Et…
Q: 3. Alcohol Starting Materials 1. methanol 2. ethanol 3. 1-propanol 4. 2-propanol 5. cyclohexanol a.…
A: In the Jones oxidation, secondary alcohols are oxidized to ketones while primary alcohols undergo…
Q: How many moles of sodium metal are needed to make one mole of the final produc N OH 1 H 2 H OH 3 H…
A: Generally sodium metal is used in reduction reaction.
Q: Question 15 Select the correct product for the following reaction. OH OH 8.Y A. D.Y C. C O A
A: H2/Pt is used in hydrogenation. It reduces alkene to alkane
Q: Clz H;50, H;0 HgSO BH/HO H,, PtO, G CH;CH;Br NaNH, 2KOH, ethanol E KMNO., H
A:
Q: a) Water with 0=SeCl2 to form O=Se(OH)2 b) Water with O=SeCl2 to form SeO2 c) Methanol with POC13 to…
A: As you not specified so I am giving answer of first 3 subparts. First nucleophile attack on…
Q: 1) 9-BBN 2) H2О2, NaOН
A: When alkene or alkyne react with borane reagent and followed by treatment with oxidizing agents like…
Q: Hydrochloric acid (75.0 mL of 0.250 M) is added to 225.0 mL of 0.0550 M Ba(OH), solution. What is…
A: Very good the work you have done and all steps you followed are correct. The answer you have for…
Q: - Protected View. Saved to this PC - O Search (Alt+Q) Mailings Review View Help 6) Which of the…
A:
Q: Indicate the reagent/product for the reaction web below. 0, (CH),S H', H,0 Na, NH, A H", H,0 HgSo, D…
A:
Q: How is the preferred product gathered? * 6. 7 from 5 to 7 O from 7 to 5 O from 2 to 9 O from 9 to 2
A: Given process is distillation process
Q: QUESTION 3 Match each reaction with the appropriate type. (The same answer may be used more than…
A:
Q: What is the major product to the following reaction? 1. NaOEt 2. Br 3. H20, HCI,, heat
A:
Q: Question 44 Choose the best reagent(s) for carrying out the conversion of the reaction below: H3C-…
A: A. O3 in presence of Zn will converts alkene to aldehyde. B. Alkene in presence of KMnO4/H3O^+ gives…
Q: 1. For the reaction: H2 +Cl2 -----> 2HCl, 3.7 moles of H2 are mixed with excess HCl. How many moles…
A: 1. For the reaction: H2 +Cl2 -----> 2HCl, 3.7 moles of H2 are mixed with excess HCl. How many…
Q: What is the major product to the following reaction? 1. NaOEt 2. Br 3. НаО, НС., heat
A:
Q: QUESTION 13 1. Select the reaction that provides the highest yield of the follov CH3 H;C-Ċ-0-CH3 CH3…
A: The synthesis of ether takes place by Williamson ether synthesis method. In this method an alkoxide…
Q: HySO, a drops) Teat butanoic acd MW 8.106 gmel GO528 gm 2.00ml uned ethanoi MW 46 06 gmo d.0.78 m…
A:
Q: Predict the products (if any) that will be formed by the reaction below. If no reaction occurs,…
A:
Q: conc. HNO3 conc. H₂SO4 /25°C
A:
Q: What is the reactant in this reaction? 1. PPh3 2. BuLi ? 3. a. (CH3)2CHCH₂Br b. CH3CHBr2 C. CH3CH₂Br…
A: ->When phosphorus ylide reacts with carbonyl compound there is formation of Alkene.
Q: major product (from Figure #1 Sults fromn the following reaction (from Reaction #10)? Reaction #10…
A: When epoxide ring is react with alkyllithium in acidic medium their is formation of primary alcohol…
Q: Ost-lar 1. Write out the balanced equation for the reaction of 1 mole of NaOH with 1 mole of H;PO4:…
A: The question is based on the concept of acid base neutralization reaction. We have to write the…
Q: What is the product. (1.40 x 10^2) (2.16 x 10^-6)
A: The question is based on the concept of physical chemistry. we have to calculate the given quantity.
Q: excess H20 HCI 1. SOC, `OH 2. 2 CH3NH2 excess CH3CH2OH H3C HCI
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Help
Step by step
Solved in 2 steps with 2 images

- Give two reasons and explanations why cyclohexane, m.p=25 c and b.p= 161 c would not be a suitable solvent for recrystallization.C7H6O3 + C4H6O3 <-> C9H8O4 + C2H4O2 the reaction has 3 acids in it; one reactant and both both products are acids. Does this test confirm that your crude product is asprin?7. Pay attention to the formula of aloe vera extract lipbalm (Aloe vera.L): Glycerin 5% Cera flava 10% Nipagin 0.18% Lanolin 15% Oleum cacao ad 100% Aloe vera extract 10% The basic ingredients in the formula are.... A. lanolinB. aloe vera extractC. glycerinD. oleum cacao
- For the reaction shown below, 2.00 mL of a 2.00 M solution of bromine, 562 mg of alkene reactant, and excess acetic acid are used to obtain 322 mg of brominated product. What is the percent yield of brominated product? Bromine MW = 159.81 g/mol; Alkene reactant MW = 134.22 g/mol;Acetic acid MW = 60.05 g/mol; Brominated product MW = 328.04 g/molif you used 0.78 g of p-ethoxyphenol (MW=138.16 g/mol) and 0.78 g of benzylbromide (MW=171.03 g/mol) for Williamson ether synthesis, which one is thelimiting reagent? What is the mole ratio between two compounds?Show your calculation with significant figure rulesGive the correct formulas of the final products formed in the following reaction. Al(OH)3 (s) + HNO3(g) ⟶ ? Group of answer choices A.) AlN3 + HO2 B.) Al3NO3 + H2O C.) Al(NO3)3 + H2O D.) Al(NO)3 + H2
- 1. Calculate the percent by volume of acetic acid by fermentation. The volume of 6% C2H5OH is 3L. Assumptions: No volume changed during fermentation and conversion efficiency is 85%. Its overall chemical reaction facilitated by Acetobacter aceti: C2H5OH + O2 = CH3COOH + H2OThe equation that represents 2% ethanolic PBP soln. With 4-methylcyclohexene.What is the second product? There should be 2.

