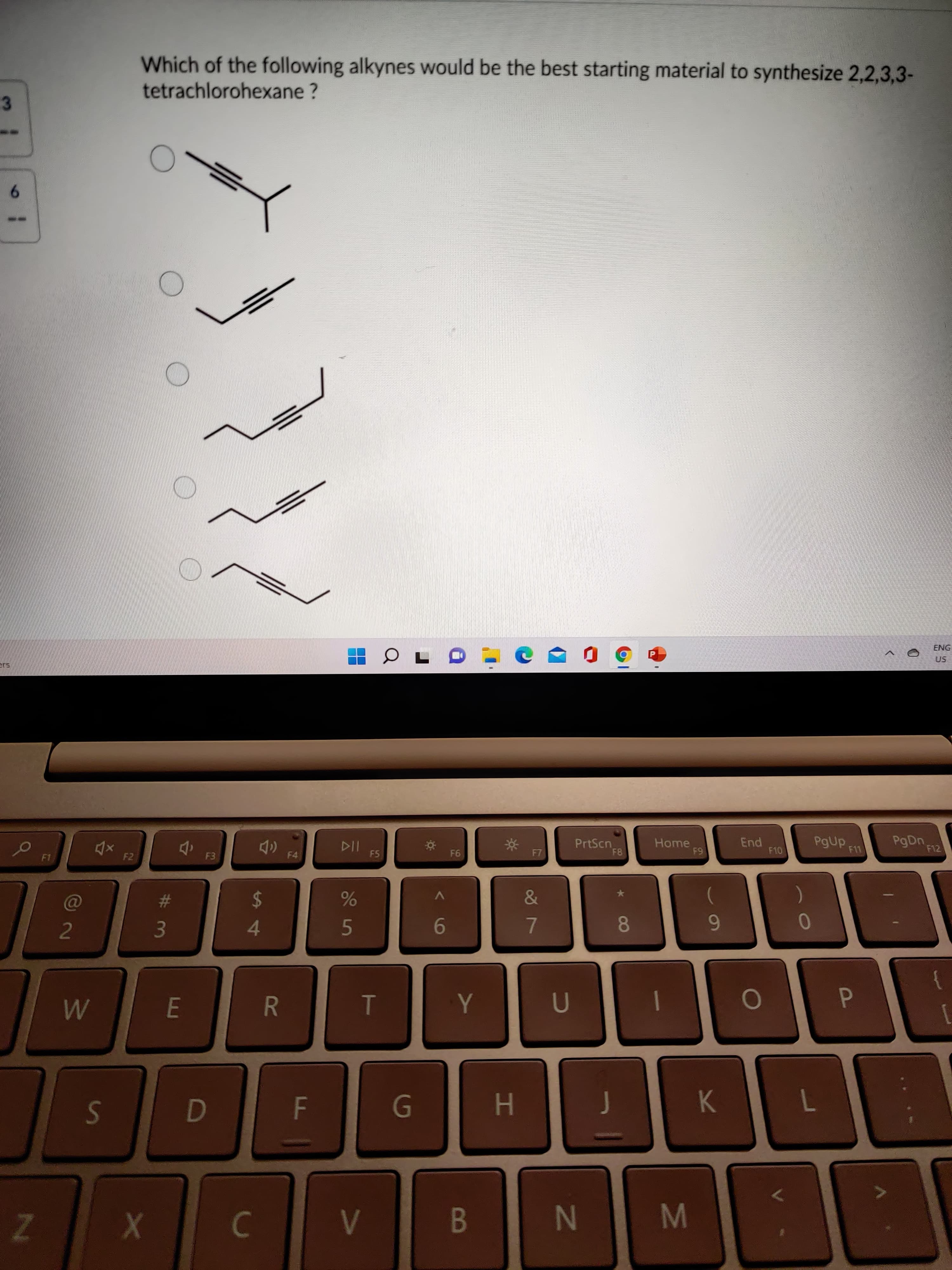
08 HI R A4 Z. M N K. P. B PgDn. 6 1 F10 PgUp ) End 64 7. F8 Home 9- 50 PrtScn 5. E. 94 F5 114 %24 3. F4 2. F2 F1 6. tetrachlorohexane ? Which of the following alkynes would be the best starting material to synthesize 2,2,3,3- 3.
Q: A penny has a mass of 2.50 g and the Moon has a mass of 7.35 × 10“ kg. Use this information to answe...
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to ...
Q: 7. The reaction for lime (CaO) production from limestone (CaCO3) is as follows CACO3(s) + heat 2 CaO...
A: Endothermic reaction can be defined as the reaction which involves absorption of heat. Given reactio...
Q: Lewis structure name of highlighted group H -C= Н — С — Н н —N— Н H Н —с — н 0=C-N - H H H H H H -N ...
A:
Q: Gaseous butane (CH,(CH,),CH;) reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide ...
A:
Q: Which salt below will produce the lowest [H 3O +] in an aqueous solution? 0.1170 M N2H5Br, Kb(N2H4)...
A: Salts dissociates into ions and these ions get solvated in the aqueous media and establish equilibri...
Q: Question 4: What is mixed melting point, and how can it be used to identify a compound. Provide two ...
A: In this experiment first we mix two compound in which one is known and another is unknown after that...
Q: Doubling a gas temperature from 100 K to 200 K causes a gas volume to double. Would you expect a sim...
A: Temperature in degree scale(oC) and kelvin scale(K) are related as following. Co+273.15=K
Q: Aqueous hydrobromic acid (HBr) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium b...
A:
Q: . What is the number of moles of nitrogen if the mass of morphine is ninety-two point four grams? 2....
A: 1. What is the number of moles of nitrogen if the mass of morphine is ninety-two point four grams? G...
Q: 2 Provide the structure(s) of the expected major organic product(s) (where appropriate) and provide ...
A: We have been asked to draw the major product along with mechanism.
Q: meo Et
A: The IUPAC name and other details of the molecule has been provided.
Q: p) Explain why the following reaction is endothermic based on pur knowledge of both bond enthalpy an...
A:
Q: An electrical engineer is analyzing an electroplating run and wants to calculate the charge that has...
A:
Q: Arrange the following from lowest to highest entropy: CS2 (l), CS2 (g), CC14 (g), S(s). Assume 1 mol...
A: Explanation:- Entropy is a measure of randomness or disorganized. So Higher entropy means more ran...
Q: Which of the following conditions characterizes a system in a state of chemical equilibrium? A. The ...
A:
Q: Question 5 What is the pH of 0.01 M acetic acid solution (Ka = 1.8x10-5) O 3.22 O 3.55 O 3.37 O 4.24
A: Concentration of acetic acid = 0.01 M Ka = 1.8 × 10-5
Q: Ető NaoH Hzi D R ーoR of (R)
A: The substrate molecule taken is aceto acetic ester. The methylene group between the two carbonyl gro...
Q: How many M-M bonds do you predict for each of the following molecules or ions? b. [(n°-CSH5)Mn(CO):]...
A: The number of metal-metal bonds of the above compounds can be calculated as follows
Q: Suppose a 250. mL flask is filled with 0.90 mol of Cl,, 1.5 mol of CHCI, and 1.9 mol of HCl. The fol...
A:
Q: A chemical engineer must calculate the maximum safe operating temperature of a high-pressure gas rea...
A: To calculate the value of maximum safe operating temperature , we would use ideal gas law.
Q: Liquid hexane (CH,(CH,) CH, reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide (C...
A: Given, Liquid hexane (C6H14) react with gaseous Oxygen (O2) gas to produce gaseous carbon dioxide (C...
Q: 1. а) Consider the reaction scheme below. [ ] PhLi HONH,, HCI A ELOH B (i) Deduce the structures of ...
A:
Q: lethane (CH,) gas and oxygen (0,) gas react to form carbon dioxide (CO,) gas and water (H,0) vapor. ...
A: Given Reaction:- CH4(g) + O2(g) -----> CO2(g) + H2O(g) Number of moles of methane = 7.0 mole...
Q: Reaction (AGº) (kJ) a. 2S(s, rhombic) + 302(g) → 2S03(g) 2SO3(9) b. S(s, rhombic) + O2(g) → 2SO2(g) ...
A:
Q: Wine goes bad soon after opening because the ethanol (CH,CH,OH) in it reacts with oxygen gas (0,) fr...
A: Mole can be defined as a standard unit for measuring large quantities of very small entities such as...
Q: Can you please answer some
A: Problem 1 :- Write the IUPAC Name for the given compounds .,
Q: A gas storage cylinder in an ordinary chemical laboratory measures 2.0 cm wide and 8.0 cm high. This...
A: We are given a cylinder of certain width and height containing Nitrogen gas at a certain pressure. W...
Q: An atom in which the number of protons is no longer equal to the number of electrons is ___. A. ...
A:
Q: How much energy is required to raise the temperature of 14.9 grams of solid tungsten from 22.5 °C to...
A: Given ;- Mass of tungsten (W) = 14.9 g Initial temperature = 22.5°C Final temperature = 35.7°C ...
Q: Consider the three statements below. Which statement(s) is(are) true? 1. Hydration is a special case...
A: Electrostatic force of attraction is defined as attractive force held between positive and negative ...
Q: Use the References to access important values if needed for this question. Arrange the following fro...
A:
Q: Solid PbBr2 (Ksp = 6.6 x 10-6) added to a solution of AgNO3, which results in the formation of solid...
A: Given : Ksp of PbBr2 = 6.6 x 10-6 Ksp of AgBr = 5.5 x 10-13
Q: In the laboratory a "coffee up" calorimeter, or constant pressure calorimeter, is frequently used to...
A: 66.29 g of zinc at 98.43 ◦C drops into 84.67 g of water at 23.26 ◦C. Final temperature of the solu...
Q: There is an overall 29-fold difference in reactivity of 1-chlorohexane, 2-chlorohexane, and 3-chloro...
A:
Q: The two molecules below are separately exposed under the given conditions. What will be the resultin...
A: Concept : Electrocyclic reaction 1) it allowed in both heat & light. 2) it is known to occur fo...
Q: .5. Anaerobic degradation of organic material can be represented by the reaction: CısH30O15 + a NH3 ...
A:
Q: 2. After a long winter quarter, you win the lottery and use your winnings to buy a tropical island i...
A: Final carbon content = 10-13% Initial carbon content = 10-10% Half life = 5745 years We have to cal...
Q: the following statements regarding Clemastine and its functional groups is NOT true? * Ether Tertiar...
A: Aromatic hydrocarbons are non-polar in nature.
Q: Use electronic factors to explain why methy carbanion is more stable than tertiary butyl carbanion
A: Answer is :
Q: try [Roferences] Use the References to access important values if needed for this question. Arrange ...
A: entropy is the amount of disorder in a system First consider the phase of each compound. Entropy inc...
Q: Ammonia (NH3) chemically reacts with oxygen gas (02) to produce nitric oxide (NO) and water (H,0). W...
A: The balanced chemical equation for the reaction of Ammonia ( NH3 ) and Oxygen gas ( O2 ) is, 4 N...
Q: intermolecular forces
A: In this question we have to explain with the knowledge of properties of matter in relation to interm...
Q: Suppose a 500. mL flask is filled with 0.70 mol of H, and 1.9 mol of I,. The following reaction beco...
A: Number of moles of H2 = 0.7 mol Number of moles of I2 = 1.9 mol Volume of rea...
Q: chemical formula of NaOH denotes
A: Given: chemical compound =NaOH To determine =chemical formula
Q: 2H зн 3H 2H 2H IS7.44 2.85-
A:
Q: the precipitate to be obtained will be more likely colloidal if the computed relative supersaturatio...
A: Precipitation can be defined as the process of conversion of a dissolved substance into an insoluble...
Q: 16. Select the correct statement below: Phosphorous contains 10 core electrons and 5 valence electro...
A: Filling of electrons in orbital obeying hund’s rule and Pauli principle.
Q: Describe/explain the following : 1.) van der Waals forces A. dipole-dipole interaction B. dipole-...
A: Internolecular forces can be defined as the forces of interactions existing between molecules, atoms...
Q: The concept that exactly 2 electrons are shared between two nuclei means that this is a ___ bond. ...
A:
Q: A positively charged particle in the nucleus of an atom is A. an electron B. a neutron ...
A: A positively charged particle in the nucleus of an atom is given below
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- A haloalkane reacts with a strong base to yield only one alkene product. Select the possible structures that can give this result from Figure 9. * A B C DDoes the data provide evidence for the claim that the reaction of Br2 with alkenes proceeds via anti‐addition? The mleting point of the product was found to be 234.5-235.5C chemical reaction: trans-stilbene+pyrimindine tribromide --->acetic acid 1,2-di bromo-1,2-diphenylethaneWhich choice best describes the alkene below?
- Does the data provide evidence for the claim that the reaction of Br2 with alkenes proceeds via anti‐addition? The mleting point of the product was found to be 234.5-235.5C chemical reaction: trans-stilbene+pyrimindine tribromide --->acetic acid 1,2-di bromo-1,2-diphenylethane product was trans-1,2-dibromo-1,2-diphenylethaneFunctional groups such as alkynes react the same in complex molecules as they do in simpler structures. The following example of alkyne reaction were taken from syntheses carried out in the research group of E. J. Corey at Harvard University. You can assume that the reactions listed involve only the alkyne, not any of the functional groups present in the molecules. Draw the expected products for the following reaction .In SN2 reactions of haloalkanes, the order of reactivity is RI>RBr>RCl>RF. Alkyl iodides are considerably more reactive than alkyl fluorides, often by factors as great as 106. All 1-halo-2,4-dinitrobenzenes, however, react at approximately the same rate in nucleophilic aromatic substitutions. Account for this difference in relative reactivities.
- best condition for the reaction (1r,2s)-1 bromo-2methylcyclohexane->(s)3-methylcyclohex-1enePredict the major products formed when 2- methyl-1-butene reacts with: H2, Pt/25°C. Show the reaction mechanism the given alkene reactionsRank the following according to INCREASING reactivity towards SN2: 1st (least reactive)? 2ND? 3RD? 4TH (most reactive)?
- Free radical halogenation is used to replace one or more hydrogens on an alkane with one or more halogens (either chlorine or bromine). It is a substitution reaction. A student performs a free radical bromination on 3-methyl-pentane (5.88 g) to create the desired product of 3-bromo-3-methyl-pentane. The product mix, which hopefully contains almost all 3-bromo-3-methyl-pentane, is cleaned up by passing it through a gas chromoatograph, which separates out the 3-bromo-3-methyl-pentane from any undesired products made during the reaction (undesired products are created frequently in free radical halogenation). The initial product mix weighed 9.45 g; the purified 3-bromo-3-methyl-pentane weighed 7.21 g. What is the student's percent recovery?1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. What is the theoretical yield of 2,4-pentanediol expressed in grams? Show calculations. What is the percentage yield of pure 2,4-pentanediol?1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. a. Provide a balanced chemical equation to show the reaction between 1,4-pentadiene and sulfuric acid. Do not use molecular formulas in the chemical equation except for sulfuric acid. b. What reactant is the limiting reagent in this chemical equation? Show calculations to support your answer.

