liters of a 5% w/v boric acid so should be used to obtain the boric acid needed in preparing 1 liter of the buffer solution? sf bluone obrioirdaori. 0.1% 3. Menthol Hexachlorophene oiiqh Glycerin Alcohol 70%, to make bre alginoggasloR ow rg oni 0.1% 10.0% 500 mL Label: Menthol and Hexachlorophene Lo- tion. How many milliliters of a 5% w/v.stock solution of menthol in alcohol should be used to obtain the amount of menthol needed in preparing the lotion?
liters of a 5% w/v boric acid so should be used to obtain the boric acid needed in preparing 1 liter of the buffer solution? sf bluone obrioirdaori. 0.1% 3. Menthol Hexachlorophene oiiqh Glycerin Alcohol 70%, to make bre alginoggasloR ow rg oni 0.1% 10.0% 500 mL Label: Menthol and Hexachlorophene Lo- tion. How many milliliters of a 5% w/v.stock solution of menthol in alcohol should be used to obtain the amount of menthol needed in preparing the lotion?
Chapter16: Applications Of Neutralization Titrations
Section: Chapter Questions
Problem 16.43QAP
Related questions
Question
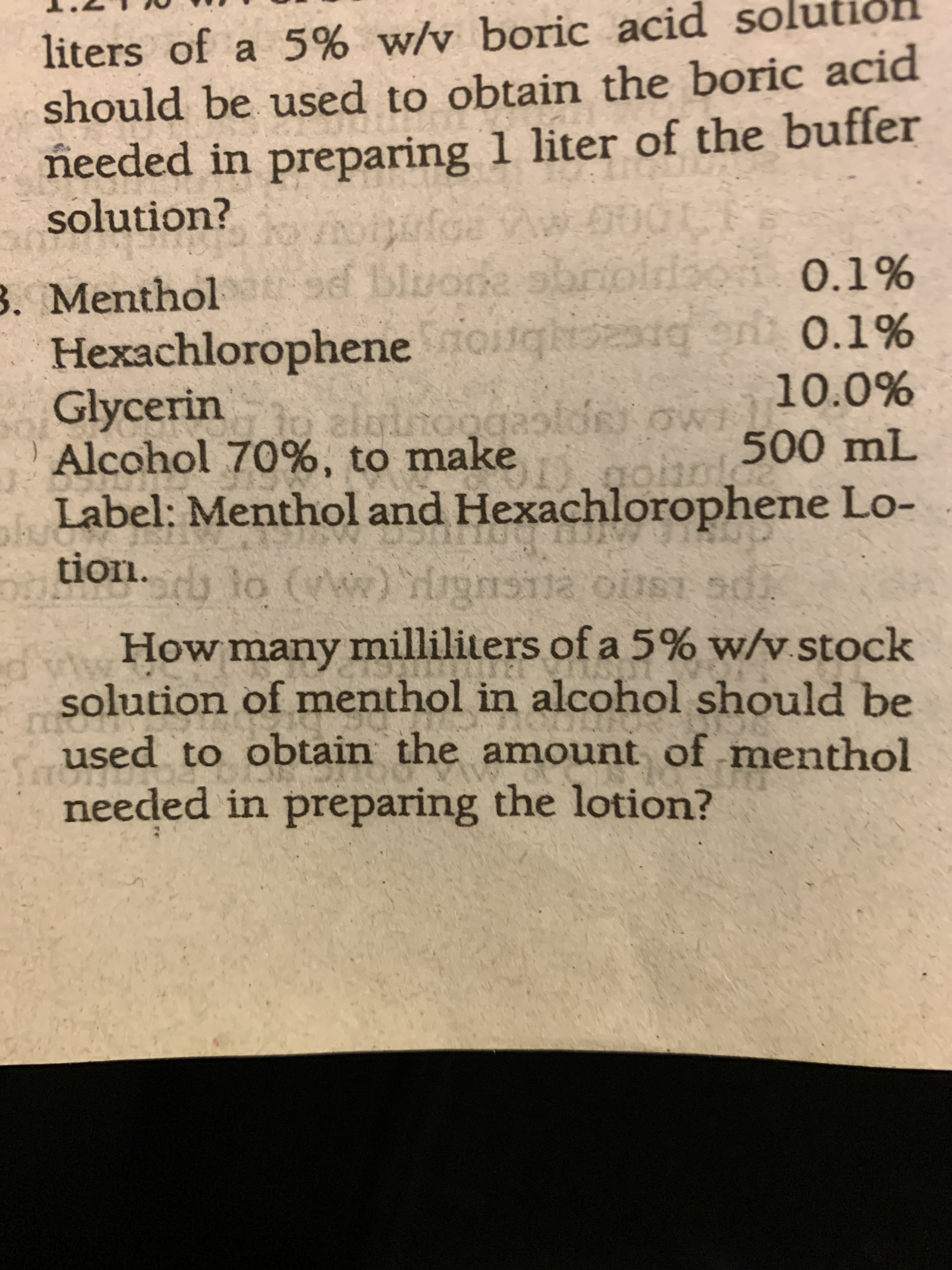
Transcribed Image Text:liters of a 5% w/v boric acid so
should be used to obtain the boric acid
needed in preparing 1 liter of the buffer
solution?
sf bluone obrioirdaori. 0.1%
3. Menthol
Hexachlorophene oiiqh
Glycerin
Alcohol 70%, to make
bre
alginoggasloR ow
rg oni 0.1%
10.0%
500 mL
Label: Menthol and Hexachlorophene Lo-
tion.
How many milliliters of a 5% w/v.stock
solution of menthol in alcohol should be
used to obtain the amount of menthol
needed in preparing the lotion?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole



Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning