Q: A galvanic cell is powered by the following redox reaction: 2+ 2 Zn²+ (aq) + N₂H₁(aq) + 4OH¯(aq) → 2…
A: Answer:Galvanic cell is a type of electrochemical cell that converts chemical energy into electrical…
Q: A chemist makes 400 ml. of sodium carbonate (Na, CO,) working solution by adding distilled water to…
A:
Q: [Review Topics] [References) Use the References to access important values if needed for this…
A:
Q: Please don't provide handwriting solution
A: Dienes shows the electrophilic addition reactions such as addition of halogens and addition of…
Q: One of the reactions that destroys ozone in the upper atmosphere is NO(g) + 03 (9) NO₂ (g) + O₂ (9)…
A: Standard gibbs free (Gibbs free energy at 298K) can be calculated using standard gibbs free energies…
Q: Propose a synthesis for this amine using only primary amines, aldehydes, ketones, carboxylic acid…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Draw the structure, then identify the hybridization for the indicated atom: a. SeF2 Se: b. HCN N: c.…
A:
Q: 7.) Draw a rough periodic table and indicate the position of the metals that make +1 ions as A, and…
A: Answer:During a chemical reaction, metal atoms generally have the tendency to lose the electrons and…
Q: Calculate the molar heat capacity at constant pressure (Cp) for a diatomic H2 gas (y = 1.41) with…
A: The relationship between the molar heat capacity at constant pressure (Cp) and the molar heat…
Q: A galvanic cell is powered by the following redox reaction: 2+ O₂(g) + 4H* (aq) + 4 Cu* (aq) →…
A:
Q: 1) 03 2) (CH3)₂S
A: The question can easily be solved if we know about reductive ozonolysis.In reductive ozonolysis the…
Q: a. Predict the sign of ASsurr for the following processes. H₂O(g) → H₂O(s) AS surr is negative AS…
A:
Q: For rubidium, the enthalpy of vaporization is 75.82 kJ/mol and the entropy of vaporization is 110.1…
A: Enthalpy of vaporization (∆H) = 75.82 kJ/mol = 75820 J/molEntropy of vaporization (∆S) = 110.1…
Q: What is the product or products when N₂O3 is dissolved in water? (reaction will not be balanced)…
A: We have to find the product when N2O3 is dissolved in water.
Q: which of the following is a reason electronic spectra of molecules typically contain broad peaks? a.…
A: The objective of the question is to identify the reason why electronic spectra of molecules…
Q: At a particular temperature, K = 3.9 × 10-6 for the following balanced reaction: 2Br2 (g) + O₂(g) →…
A:
Q: ) A truck driver carrying a load of lead nitrate (Pb(NO3)2) lost control of his semi- truck after…
A: The scenario described involves a truck carrying lead nitrate (Pb(NO3)2) crashing into Blue Lake,…
Q: A student proposes the following mechanism for a nucleophilic substitution reaction: Br: ? Notice…
A: The bimolecular nucleophilic substitution reaction is a concerted reaction that occurs in a single…
Q: A galvanic cell is powered by the following redox reaction: 2+ 2 Cro (aq) + 8 H₂O(1) + 3 Zn(s) → 2…
A:
Q: Design syntheses to produce the following compounds starting with any alcohol with 3 or less carbons…
A: Since you have asked multiple questions,we will answer first question for you. Please repost the…
Q: Provide the correct IUPAC name for the skeletal (bond-line) structure shown here.
A:
Q: Strong base is dissolved in 525 mL of 0.200 M weak acid (K₁ = 3.37 × 10-5) to make a buffer with a…
A: Answer:Buffer solution is a type of solution that resists the change in its pH on adding small…
Q: o mechanism needed just reagents and general conditions 3. Show how to synthesize alkene B from…
A: Given is organic synthesis reaction.Given is multi step reaction.The given reactant is alkene and…
Q: MISSED THIS? Watch KCV: The Change in Enthalpy for a Chemical Reaction, IWE: Stoichiometry Involving…
A: Enthalpy is used to indicate the heat changes occurring during a reaction. A negative enthalpy value…
Q: The standard cell potential for a redox reaction is 1.26 V. For the balanced equation involving a…
A: Equilibrium constant is defined as the ratio of concentration of products to the concentration of…
Q: b. CH3 CH3 NaBr DCM
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: A small amount of acetonitrile (CH,CN) is dissolved in a large amount of water. Imagine separating…
A: The enthalpy of a solution is defined as the quantity of heat absorbed or liberated when a solute is…
Q: Draw and upload a proposed synthesis for the compound below by building the heterocyclic ring. Show…
A: The details of the synthesis of the given molecule are provided below:
Q: The heat of vaporization of water is 40.65 kJ/mol. How much heat is required to vaporize 330 g of…
A: We know the amount of heat or energy required to vaporize water into the gas at its boiling point is…
Q: 8) Predict the product. OEt 1. NaOEt 2. H30*
A: The Claisen reaction or Claisen condensation is a carbon–carbon bond forming reaction. In this…
Q: What is the configuration of this alkene? CI X Trans E OZ Neither E nor Z
A: we assign priorities to different substituents present across double bonds based on CIP rules.When…
Q: ii. A buffer solution was prepared by mixing 34 cm³ of 0.40 M NH, solution and 21 cm³ of 0.50 M…
A: Henderson-Hasselbalch equation for buffer solution -pOH = pKb + log([salt]/[base])[Salt] =…
Q: Which one of the following represents an acceptable set of quantum numbers for an electron in an…
A: The objective of the question is to identify the correct set of quantum numbers for an electron in…
Q: Consider the reaction C₂H4(g) + H₂O(g) CH3CH₂OH(g) Using the standard thermodynamic data in the…
A: Answer:Here:
Q: Select the true statements from the following a. Alkali fuel cell stacks can give outputs in the…
A: Alkali fuel cell type of fuel cell where alkali metal hydroxide is used as electrolyte and water is…
Q: Predict the major product(s) for the following reaction. Excess HI Heat ? Modify the given structure…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Cl₂ H₂O P Atoms, Bonds and…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: How many chiral centers are present in this molecule? (a) (b) two (c) (d) one three none ملها
A: Chiral centre is a carbon atom which are attached with four different groups.
Q: 3. Propose an efficient synthesis to get the final product using malonic ester Hint: 6 steps to get…
A: Solution If effcient synthesis to get the final product using malonic Ester..
Q: The mechanism of this reaction involves two carbocation intermediates, A and B. Part 1 of 2 OH Draw…
A: Given that, the reaction is:
Q: An analytical chemist is titrating 77.9mL of a 0.7600M solution of methylamine CH3NH2 with a 0.4700M…
A: In this question, we want to determine the pH of the Solution after titration of weak base with…
Q: A 100.0 mL aliquot of a 0.100 M solution of benzoic acid (C6H5COOH, Ka = 1.2 x 10-4) is titrated…
A: The objective of this question is to calculate the pH at the equivalence point of a titration…
Q: What reaction(s) are the best method for performing the synthesis shown below? Select one: NH₂ a. 1)…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Without doing any calculations, match the following thermodynamic properties with their appropriate…
A:
Q: Given the following equation: 2 C4H10 + 13 028 CO₂ + 10 H₂O How many grams of C4H10 are needed to…
A: Given is combustion reaction of butane.Given data:Mass of H2O produced = 54.3 g
Q: Suppose a 500. mL. flask is filled with 0.10 mol of Cl₂ and 0.50 mol of HCI. The following reaction…
A:
Q: Draw a sigma complex which forms during the reaction shown below. CI FeCl3
A: Given,The reaction:
Q: Which metal show +8 oxidation state? (a) Te (b) Ru (c) Re (d) OS
A: Both Ruthenium (Ru) and Osmium (Os) can exhibit the +8 oxidation state in some compounds.
Q: Which of the following chair conformations is the most stable, and why? (1) CI C(CH₂) (ii) Check C…
A: The basic principle is; the bulky group should always come at equatorial position.The higher the…
Q: The concentrations of NaCl and KI are 0.150 M and 0.110 M, respectively, in the electrochemical cell…
A: This problem is based on electrochemistry where we have to use nernst equation and then find out the…
What reagants are needed to convert from first structure to second structure?
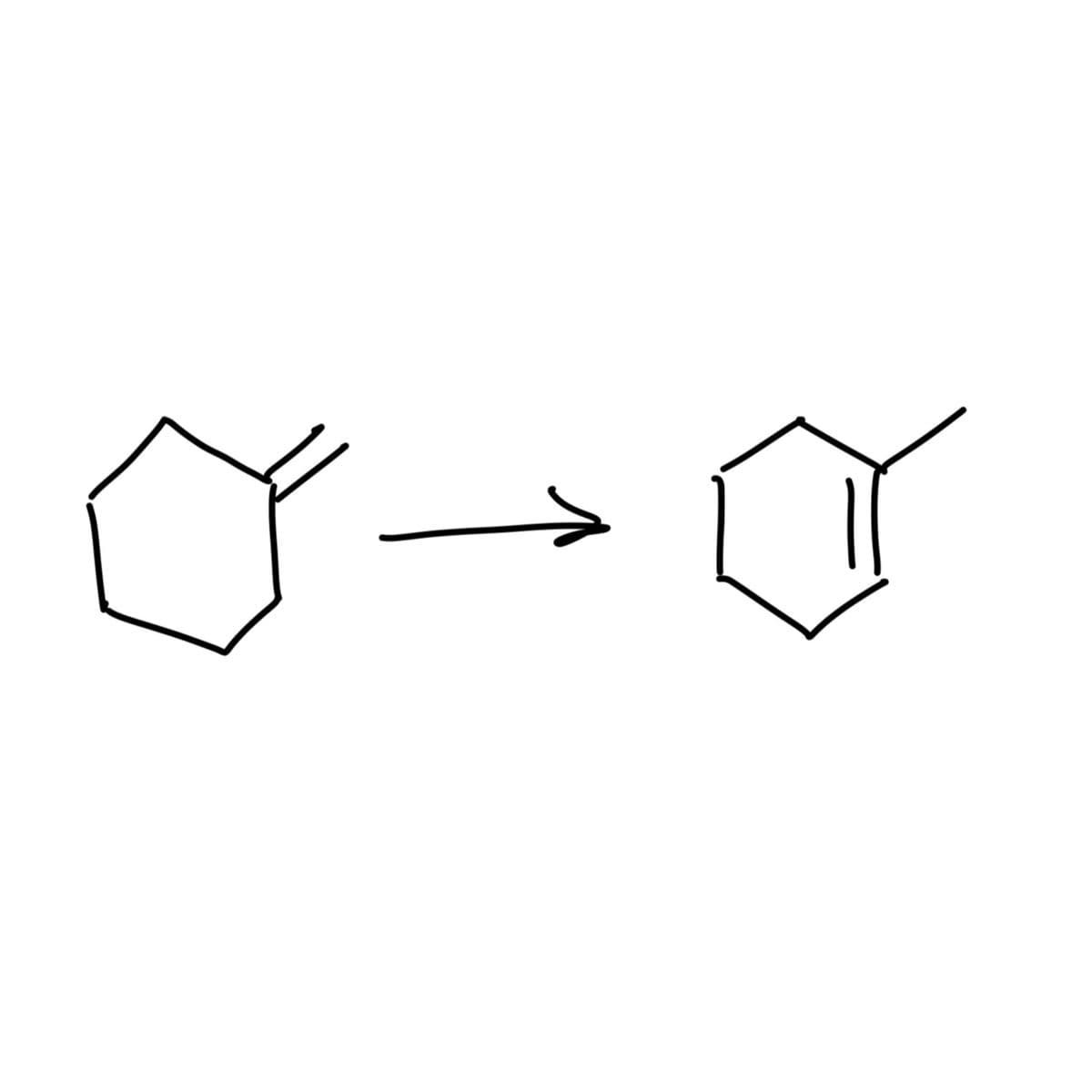
Step by step
Solved in 3 steps with 2 images


