Identify the Formula, Oxidation number and Charge (Positive/ Negative) of th three POLYATOMIC IONS below. Place answers on spaces trailing after the image. Name of lon Formula Oxidation Charge of No. lon Polyatomic ions Nitrate (77) (78) (79) Carbonate (80) (81) 82) Sulfate (83) (84) (85)
Identify the Formula, Oxidation number and Charge (Positive/ Negative) of th three POLYATOMIC IONS below. Place answers on spaces trailing after the image. Name of lon Formula Oxidation Charge of No. lon Polyatomic ions Nitrate (77) (78) (79) Carbonate (80) (81) 82) Sulfate (83) (84) (85)
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter11: Solutions
Section: Chapter Questions
Problem 40P
Related questions
Question
77
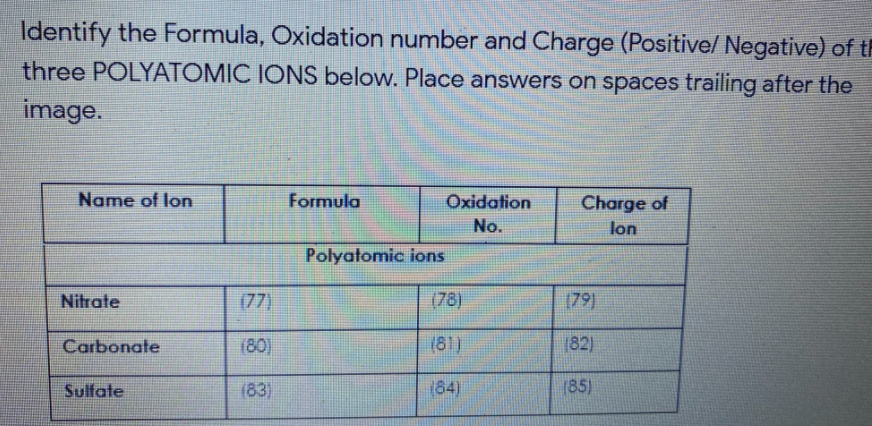
Transcribed Image Text:Identify the Formula, Oxidation number and Charge (Positive/ Negative) of th
three POLYATOMIC IONS below. Place answers on spaces trailing after the
image.
Name of lon
Formula
Oxidation
Charge of
No.
lon
Polyatomic ions
Nitrate
(77)
(78)
(79)
Carbonate
(80)
(81)
82)
Sulfate
(83)
(84)
(85)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning