Q: The decomposition of ammonium carbamate (NH4CO2NH2) is shown below: NH,CO,NH2(s) =2 NH3(g) + 1 CO2(g...
A: The balanced reaction taking place is given by, => NH4CO2NH2 (s) ---------> 2 NH3 (g) + CO2 (g...
Q: 2NO(g) + O2(g) 2NO2(g); AH°= -125 kJ/mol; AS°= -150 J/mol-K Below a temperature of K, this reaction ...
A:
Q: Explain the different Types of compound under Hydrocarbons ?
A:
Q: Chemistry Question
A: NH3 + O2 -----> H2O + NO Firstly Balanced the Hydrogen in both side 4NH3 + O2 ----> 6H2O + N...
Q: The concentration of a certain sodium hydroxide solution was determined by using the solution to tit...
A:
Q: How do I find the mass and the percent of Cu with the data given?
A:
Q: The order of this reaction in N,O5 is a. b. 1. CO
A: Chemical kinetics is the study of the reaction that how fast or slow a reaction is taking place. The...
Q: The mass percent of C in a certain organic compound (C3H8X) is 29.28%. What is the identity of X?
A:
Q: The largest atom in a water molecule is _____________.
A: Water which is known as the universal solvent is a colorless, odorless and tasteless inorganic liqui...
Q: Give a complete reaction mechanism for the reaction of toluene in the presence of bromine and FeBr3.
A: Given Reactants : Toluene Bromine FeBr3 Products and Mechanism = To be determined
Q: A decomposition reaction has a rate constant of 0.175 1/ yr. How long (in years) does it take for th...
A:
Q: What is the purpose of the laboratory exercise about simple distillation and fractional distillation
A: To refining the liquid through a separation process is known as distillation. The main purpose ...
Q: Chemical formula of NH4+ and SCN-
A:
Q: An unknown solid object with a mass of 35.0 g was heated to 100.0 °C and transferred to a calorimete...
A: Solution: Mass of an unknown solid=35g Specific heat of water (CW)=4.184J/g∘C Mass of water (mw)=50g...
Q: Select the excess reagent in the following equation: P4+502=>P4O10 in open air P4 02 P4010 There is ...
A: The right option is O2 .
Q: Chemistry Question
A: The given chemical reaction is as follows: 2N2O5 g → 4NO2 g + O2 g The rate constant for this react...
Q: The initial contents of the reactor is 1.495 atm of SO3(g) and 5.000 moles of C(s). At equilibrium y...
A: To solve this problem first we have to calculate the equilibrium partial pressure of each reactants ...
Q: Does a reaction occur when aqueous solutions of cobalt(II) sulfate and ammonium phosphate are combin...
A:
Q: How many atoms of oxygen are in a 0.29 mol sample of CaCO3? O 5.2E23 5.90E+65 5.40E+56 6.40E+58 none
A: Moles of CaCO3 = 0.29 mol
Q: Given 15.0g HgO and the following unbalanced equation, complete a-f. ___HgO (s) → ___Hg (l) + ___O2 ...
A: Before doing the problem at first we should balance the chemical reaction 2HgO(s) = 2Hg (l) +O2 (g...
Q: A 0.10-M solution of formic acid (HCOOH) is prepared in various solvents: (a) pure water (b) 0.10 M ...
A:
Q: At the equivalence point, both the acid and the base are completely consumed by neutralization. Howe...
A: Please find the answer below.
Q: Good evening, Can I receive help with finishing this problem?
A: The question is based on the titration of weak base pyridine with strong acid hydrobromic acid. we h...
Q: convert 64gN to mol 4.4mol 4mol 4.6mol 6.4mol none of the above
A: Given values are, Mass of N= 64 g Molar mass of N= 14.007 g/mol
Q: Reaction rate is expressed in terms of changes in concentration of reactants and products. Write a b...
A: Rate of a chemical reaction is defined as decrease in concentration of reactants with time or increa...
Q: Consider the reaction X → Y + ZWhich of the following is a possible rate law?
A:
Q: CH3 HO H3C CH3 CH3 H3C
A: The question is based on the concept of chemical conversions. we have to convert the reactant into p...
Q: 5) A hydrate has the following percent composition: Pb -54.61%, C-12.66%, H=1.61%, O-16.88%, and H2O...
A: The formula of the compound in which the constituent elements are present in the simplest whole numb...
Q: Please help
A: Density is the ratio of mass to the volume of the substance . Density = mass/volume
Q: A hydrochloric acid solution consisted of a volume of 98.5mL with a concentration of 0.945M HCl is a...
A: Solution : Given, The volume of HCl = 98.5mL The concentration of HCl is 0.945 M The concentration o...
Q: The following acid-base reaction occurs spontaneously in the gas phase: NH3(g) + HCI(g)=NHẠCI(s) Dur...
A:
Q: What functional groups are present
A: Here we have to write the functional groups are present in the following molecule.
Q: What kind of intermolecular forces act between a hydrogen (H,) molecule and a neon atom? Note: If th...
A: In covalent compound various types of intermolecular force of attraction acts between molecules and ...
Q: Use the following information to determine the activation energy for the reaction shown here: 2NO →N...
A: The temperature v/s rate constant data given is,
Q: fīde and chromium(III) sulfate are combined? ne the solubility of compounds.
A: Yes This Reaction occur because you will obtained solid precipitated product Balanced molecular rea...
Q: Good evenig, Earlier I submitted this problem to try and receive some help with understanding the co...
A: Not, as per Bartleby guidelines, we can answer only first 3 questions Given 50.0 mL of 0.200 M HCl...
Q: Chemical formula of Mg2+ and CO32-
A: Writing chemical formula of ionic compounds :- Electrical neutrality principle is used while writing...
Q: II. If 60.2 grams of Hg combines completely with 24 grams of Br to form a compound, what is the % co...
A: Given: Mass of mercury (Hg)=60.2 g Mass of Bromine (Br)=24 g The total mass of compound =(60.2 +24) ...
Q: what are the coefficients missing on the following equation?_KNO3+_H2CO3--->_K2CO3+_HNO3 (2,1,1,2) (...
A: In the given question we have to balance the chemical reaction with proper coefficient as shown miss...
Q: What is the % by mass of Cu in the ore malachite (Cu2(OH)2CO3) ? (Answer to the nearest 0.1 % , thre...
A: Interpretation: The % by mass of Cu in the ore malachite Cu2OH2CO3 is to be calculated.
Q: There are two different ways of making 2-ethoxyoctane from octan-2-ol using the Williamson ether syn...
A: The nucleophile alkoxide group strikes the compound alkyl halide through the SN2 mechanism followed ...
Q:
A: Given : To find the particles having highest average kinetic energy.
Q: OH + (0)
A: The product will be Cyclopentanone
Q: A study of the gas-phase oxidation of nitrogen monoxide at 25°C and 1.00 atm pressure gave the follo...
A: The reaction taking place is given as, And the reaction kinetics data given is,
Q: If I wanted to separate a mixture containing formic acid and naphthol that were dissolved in diethyl...
A: Both formic acid and naphthol are acidic in nature but pKa value of formic acid is 3.75 and pKa valu...
Q: How many grams of cadmium sulfide will be produced when 63.0 ml of a 3.00 Mof cadmium nitrate is mix...
A:
Q: The interhalogen compound BrF3 is a volatile, straw-colored liquid. The compound exhibits appreciabl...
A: The autoionization reaction of bromine trifluoride is given as: 2 BrF3 ⇌ BrF2+ + BrF4- Dissolution o...
Q: N2O + 2F2+ 6OH-2NO2- + 4F-+ 3H2O In the above redox reaction, use oxidation numbers to identify t...
A: N2O + 2F2+ 6OH- →2NO2- + 4F-+ 3H2O In the given redox reaction, the oxidation numbers of the elem...
Q: Five kilograms of ice at 0 degree Celsius is melted and converted to water at 0 degree celsius. Comp...
A: As this is a reversible isothermal process exchanging heat Q with the surroundings, its entropy is g...
Q: How many atoms of oxygen are in a 0.78 mol sample of CaCO3? 1.4E24 4.70E+25 1.50E+29 1.40E+20 none
A: Solution : NA-Avogadro's number =6.022×1023 In 1 mol of CaCO3, 3 oxygen atoms are present. In 0.78 m...
What is the name of this compound
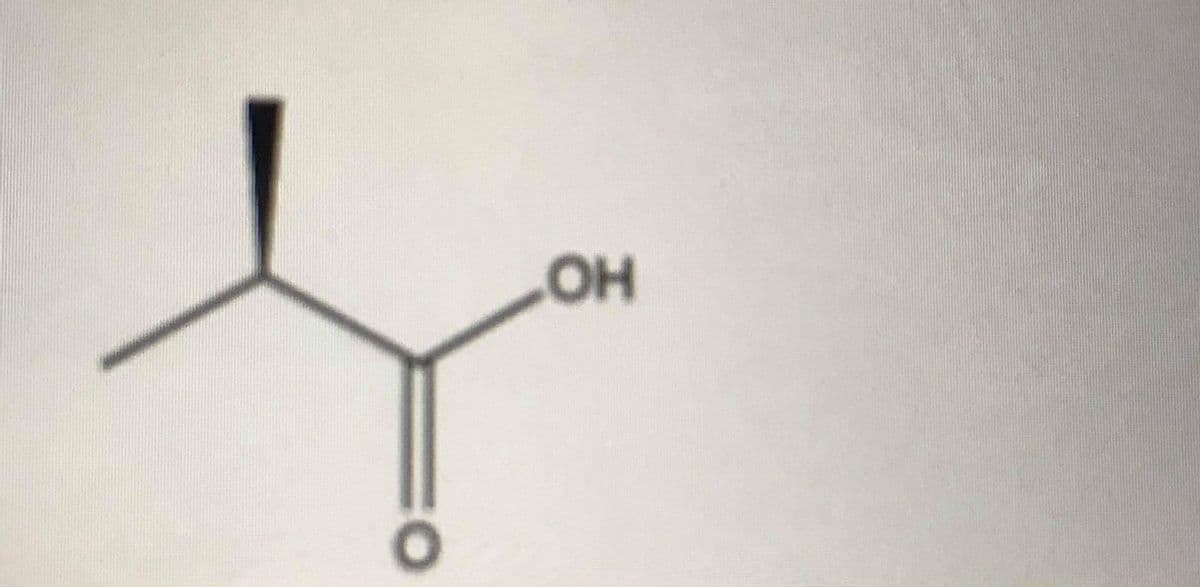
Step by step
Solved in 2 steps with 2 images


