Calculating and using the van't Hoff factor for electrolytes When 119. g of glycine (C₂H5NO₂) are dissolved in 1500. g of a certain mystery liquid X, the freezing point of the solution is 6.1 °C lower than the freezing point of pure X. On the other hand, when 119. g of potassium bromide are dissolved in the same mass of X, the freezing point of the solution is 7.3 °C lower than the freezing point of pure X. Calculate the van't Hoff factor for potassium bromide in X. Be sure your answer has a unit symbol, if necessary, and round your answer to 2 significant digits. i = [ x10 0/5 X
Calculating and using the van't Hoff factor for electrolytes When 119. g of glycine (C₂H5NO₂) are dissolved in 1500. g of a certain mystery liquid X, the freezing point of the solution is 6.1 °C lower than the freezing point of pure X. On the other hand, when 119. g of potassium bromide are dissolved in the same mass of X, the freezing point of the solution is 7.3 °C lower than the freezing point of pure X. Calculate the van't Hoff factor for potassium bromide in X. Be sure your answer has a unit symbol, if necessary, and round your answer to 2 significant digits. i = [ x10 0/5 X
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter12: Solutions
Section: Chapter Questions
Problem 12.95QE
Related questions
Question
Please keep in mind sig figs as well if needs scientific notation. TYSM :)
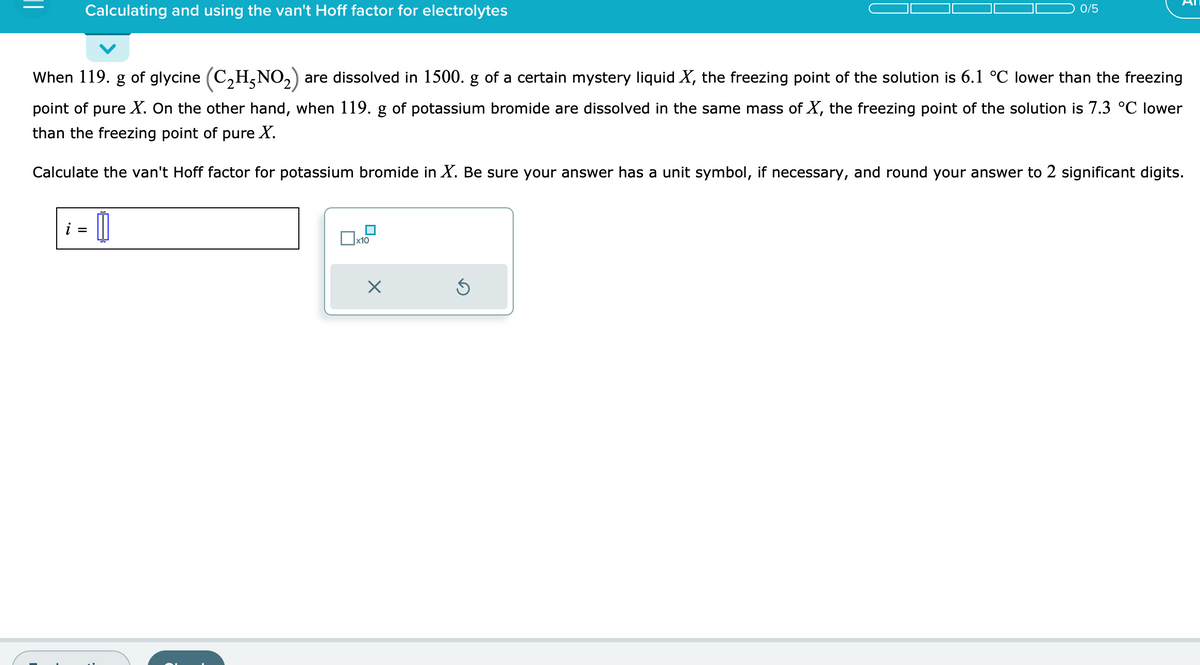
Transcribed Image Text:Calculating and using the van't Hoff factor for electrolytes
When 119. g of glycine (C₂H5NO₂) are dissolved in 1500. g of a certain mystery liquid X, the freezing point of the solution is 6.1 °C lower than the freezing
point of pure X. On the other hand, when 119. g of potassium bromide are dissolved in the same mass of X, the freezing point of the solution is 7.3 °C lower
than the freezing point of pure X.
Calculate the van't Hoff factor for potassium bromide in X. Be sure your answer has a unit symbol, if necessary, and round your answer to 2 significant digits.
i =
[
x10
0/5
X
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning