Q: 26. The osmotic pressure of a MgSO4 (120.4 g/mol) solution at 25°C is 0.489 atm. Calculate the…
A: Molarity is defined as number of mole of substance divide by volume of solution in liter
Q: For the following reaction: A(g) = 2 B (g) Given the following information, what is the equilibrium…
A:
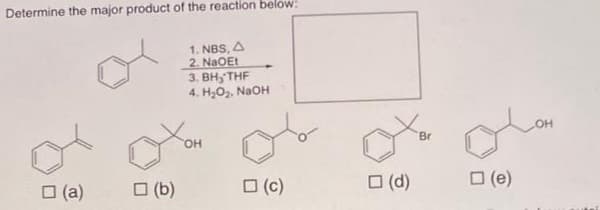
Q: 4. Propose a synthesis of the following compound using only benzene and propyne as sources of…
A: In this question, we will synthesized the final organic product by using starting material as only…
Q: The density of an aqueous solution containing 15.0 percent ethanol (C₂H5OH) by mass is 0.979- your…
A: Given : Mass percentage of ethanol = 15.0 ℅ Density of solution = 0.979 g/ml
Q: Station #3 = H2SO4 (aq) For this acid, what does the white bead represent?________ blue…
A: H2SO4 is strong acid, which is diprotic acid, i.e., it will dissociate two hydrogens, as follows,…
Q: S O app.101edu.co STARTING AMOUNT X How many grams of AgCl will be formed when 60.0 mL of 0.500 M…
A: To solve this problem we have to calculate the mass of AgCl .
Q: Mention and describe the groups involving the wavenumbers
A: The question is based on the concept of organic spectroscopy. we have to analyse the IR spectra…
Q: sorry i cannoted read step 2 clearly
A: We have to explain about the structure of the hydrated potassium ion and chloride ion. Introduction:…
Q: At a certain temperature, 0.88 mol N₂ and 2.654 mol H₂ are placed in a container. N₂(g) + 3H₂(g) =…
A: The question is based on the concept of chemical equilibrium. we have been given initial moles of…
Q: Indicate, by letter(s), the position(s) on the ring at which substitu occurs when the aromatic…
A: Bromination reaction to an aromatic ring is an example of aromatic electrophilic substitution…
Q: Be sure to answer all parts. Fermentation is a complex chemical process of wine making in which…
A: Chemical reaction C6H12O6 → 2C2H5OH + 2CO2 Amount of glucose = 840.9 g Molar mass of glucose =…
Q: For a 6.60x10-2 M solution of H₂Se, calculate both the pH and the Se2- ion concentration. H₂Se + H₂0…
A:
Q: Fermentation is a complex chemical process of wine making in which glucose is converted into ethanol…
A: Given, the chemical reaction is as follows,C6H12O6→2C2H5OH+2CO2amount of glucose for reaction =618.8…
Q: 2. Which statement is most accurate? * O Oxygen is more electronegative than carbon O Carbon is more…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: Dehydrohalogenation by E1 mechanism takes place in the following steps: Step 1 : Halide ion leaves…
Q: 1.) Indicate whether each of the molecules shown below is aromatic, anti-aro non-aromatic. You may…
A: First one, aromatic compound. Second one,anti aromatic compound. Third one, aromatic compound.…
Q: Problem: Answer the following about the acids shown below. (a) Draw curved arrows to show a proton…
A: The acid will release the hydrogen to base (H2O here), and will form the counter anion of the base…
Q: Determine percent yield for the reaction. Magnesium oxide can be made by heating magnesium…
A:
Q: The Solubility Product Constant for nickel(II) hydroxide is 2.8 x 10 ^-16 The maximum amount of…
A:
Q: A scientist measures the standard enthalpy change for this reaction to be -907.4 kJ/mol. 2NH3(g) + 3…
A:
Q: of 10 > O Macmillan Learni Step 1: How many moles of NaOH are present in 28.0 mL of 0.130 M NaOH?…
A: Molarity is defined as number of mole of substance divide by volume of solution in liter given…
Q: A hypothetical salt, MX, is studied and Ksp increases with temperature. (a) Write the chemical…
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: Will there be any reaction when bromine is added to toluene? What will happen when aluminum chloride…
A: In general when bromine is added to toluene, there will not a direct reaction. When AlCl3 will be…
Q: write the general equation to show the reaction between an ester and a grignard reagent. write the…
A: Grignard reagent (RMgX) is the source of nucleophile because it has carbon-magnesium bonding. Carbon…
Q: Use this information Al + 3/2 Cl(g) → AlCl ΔH = -352.82 kJ/mol Cl + H → 2HCl ΔH = -80.04 kJ/mol 2Al…
A: Given, a) Al + 3/2 Cl(g) → AlCl ΔH = -352.82 kJ/mol b) Cl + H → 2HCl ΔH = -80.04 kJ/mol c) 2Al +…
Q: 2) Draw the resonance structures for the following compounds? In some cases it is not possible to…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 0 0 0
A: Answer: Given diagram is the pictorial representation of acid HBr molecules in water and on the…
Q: "Synthesis gas" is a mixture of carbon monoxide and water vapor. At high temperature synthesis gas…
A: Answer: Value of equilibrium constant KP is always equal to the ratio of partial pressure of…
Q: Give the IUPAC name for the following compound. CN Multiple Choice O O O O…
A: According to the IUPAC nomenclature, First longest chain of carbon atom is identified. Numbering…
Q: Which product is formed in the following reaction? OC) 0.5 equiv of III OD) both I and II OB) II OA)…
A: The given reaction is addition reaction on alkyne by HCl.
Q: a) b) + H₂ Ni CI OH
A: When benzene reacts with Cl2 in presence of Lewis acid then it forms chloro benzene
Q: = Consider the following equilibrium: H₂(g) + I₂ (s) ⇒ 2 HI(g) AH 298 +51.9 kJ AS 298 AS 298 +165.3…
A: Since we know, ∆Go = ∆Ho - T∆So And the relation between ∆Go and equilibrium constant K is ∆Go = -RT…
Q: Calculate the mass of disodium EDTA (FW 372.25) needed to make 500-mL 0.015 M solution of EDTA. What…
A: To calculate the mass of disodium EDTA required to prepare 500 mL of 0.015 M solution of EDTA.
Q: 1) Draw two different Lewis structures, corresponding two condensed structures, and corresponding…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 4:25 PM Thu Mar 2 Initial (M) Change (M) Equilibrium 2.70 0 0.70 At high temperature, SO₂ and NO₂…
A: We know that Molarity = moles/volume Since volume = 1 L Molarity = moles
Q: What diene and dienophile would react to give the following Diels-Alder product? Select one: I 0.0…
A:
Q: Consider the following chemical reaction at equilibrium: NH3(aq) + H₂O(1) ⇒ NH₁*(aq) + HO¯(aq) If…
A:
Q: To prepare 50 mL of a 5.0% aqueous m/v saline solution from a 10% aqueous m/v saline stock solution,…
A: Dilution of the solution: When we dilute a concentrated solution with pure water, the amount of…
Q: Rank the following aqueous solutions in order of decreasing freezing point. Select 1 (highest…
A: Freezing point depression is a colligative property which depends on the no. of solute particles…
Q: 6. You need to prepare 825. g of a 7.95% by mass calcium chloride solution. How many grams of…
A: Given, Mass % of the calcium chloride solution = 7.95 % Mass of the solution = 825. g Mass of…
Q: Fluoride ion selective electrodes obey the following equation: E(V) K-3(0.0592)log[F] where the…
A: Solutions- Here, we have to determine the value of K and β from plot.
Q: Carbon-15 has a half-life of 2.5 seconds. Suppose we have a sample containing 200 mg of carbon-15.…
A: In this question, we will determine the how much mass of C-15 will remain after 5second. Given that,…
Q: (D) Another repeating transformation is the reductive amination that shows up in the Schemes 3, 5…
A: Reductive amination involves conversion of carbonyl group to amine via imine intermediate.…
Q: Balance each disproportionation reaction occurring in acidic solution. a)Sn2+(aq)→Sn(s)+Sn4+(aq)…
A: Answer:- This question is answered by using the simple concept of balancing the disproportionation…
Q: BONUS: The H NMR spectrum for para-nitrotoluene is shown below is the signal for Ha or Hb more…
A: We have to know the concept of downfield and upfield, which is shown graphically below-
Q: What is the frequency of light (in Hz) of a photon emitted when an electron in a hydrogen atom…
A:
Q: Draw the most stable conformation of cis-1-tert-butyl-3-ethylcyclohexane. (a) (b)…
A:
Q: pH = pK₂ + log [HCO3] (0.030) (Pco₂) where [HCO3] is given in millimoles/litre and the arterial…
A:
Q: A 20 wt% Sn-80 wt% Pb alloy is heated to a temperature of 150°C. For this temperature, calculate the…
A: We have find out the mass fraction.
Q: + 0 A. I B. ॥ & || heat > www x x ||| ? IV US -
A: When a diene reacts with dienophile they undergo 4+2 cyclo addition reaction to form six member ring…
Give detailed Solution with explanation needed
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- Complete the balanced molecular chemicahal equation for the reaction below. If no reaction occurs, write NR after the reaction arrow. RB2CO(aq)+Fe(C2H3)2)2(aq)> Please explain Thanks.3. For medicinal substances: sodium chloride, formaldehyde solution, ascorbic acid - the general reaction of authenticity is the reaction with?A) Iron (III) chlorideB) Silver nitrateC) ChloramineD) Sodium nitriteE) Potassium permanganateFor 20.7 the solutions for part c and d dont seem right. Shouldn't reacting with 1 eq of H2 just take away the C=C bond and not the C=O bond. (H2 reduces C=C selectively to form a ketone according to page 738. Also the both the C=C and C=O bond should be reduced if excess H2 is used
- (C₈H₁₀) according to the following reaction: C₈H₁₀(l) + O₂(g) + NH₃(g) → C₈H₄N₂(s) + H₂O(l) How many grams of water would be produced by the complete ammoxidation of 16.50 moles of o-xylene?1.Which of the following is a possible product for the unbalanced chemical reaction: NaCl + K22SO44--> KCl K22Cl NaSO44 NaK22 no reaction occursCromolyn sodium, developed in the 1960s, has been used to prevent allergic reactions primarily affecting the lungs, as, for example, exercise-induced emphysema. It is thought to block the release of histamine, which prevents the sequence of events leading to swelling, itching, and constriction of bronchial tubes. Cromolyn sodium is synthesized in the following series of steps. Treatment of one mole of epichlorohydrin with two moles of 2,6-dihydroxyacetophenone in the presence of base gives I. Treatment of I with two moles of diethyl oxalate in the presence of sodium ethoxide gives a diester II. Saponification of the diester with aqueous NaOH gives cromolyn sodium. Q. Is cromolyn sodium chiral? If so, which of the possible stereoisomers are formed in this synthesis?
- Cromolyn sodium, developed in the 1960s, has been used to prevent allergic reactions primarily affecting the lungs, as, for example, exercise-induced emphysema. It is thought to block the release of histamine, which prevents the sequence of events leading to swelling, itching, and constriction of bronchial tubes. Cromolyn sodium is synthesized in the following series of steps. Treatment of one mole of epichlorohydrin with two moles of 2,6-dihydroxyacetophenone in the presence of base gives I. Treatment of I with two moles of diethyl oxalate in the presence of sodium ethoxide gives a diester II. Saponification of the diester with aqueous NaOH gives cromolyn sodium. Q.Propose a mechanism for the formation of compound I.Cromolyn sodium, developed in the 1960s, has been used to prevent allergic reactions primarily affecting the lungs, as, for example, exercise-induced emphysema. It is thought to block the release of histamine, which prevents the sequence of events leading to swelling, itching, and constriction of bronchial tubes. Cromolyn sodium is synthesized in the following series of steps. Treatment of one mole of epichlorohydrin with two moles of 2,6-dihydroxyacetophenone in the presence of base gives I. Treatment of I with two moles of diethyl oxalate in the presence of sodium ethoxide gives a diester II. Saponification of the diester with aqueous NaOH gives cromolyn sodium. Q.Propose a structural formula for compound II and a mechanism for its formation.If the G for a reaction is 4.5 kcal/mol at 298 K, what is the Keq for this reaction? What is the change in entropy of this reaction if H = 3.2 kcal/mol?
- What is the limiting reactant and theoretical yeild of this Chemiluminescent Reaction? The reaction uses 4ml of a 0.6M phenol (4-methylphenol), 1.2 mL of oxalyl chloride and 0.35 mL of triethylamine(i) Arrange the following compounds in an increasing order of basic strength :C6H5NH2, C6H5N(CH3)2, (C2H5)2NH and CH3NH2(ii) Arrange the following compounds in a decreasing order of pKb values :C2H5NH2, C6H5NHCH3, (C2H5)2NH and C6H5NH3(a) Morphine is known to be a very powerful pain-killer, and it is commonly used in medical treatments. Briefly describe how morphine is obtained, and how it works as a pain-killer. Please answer very soon will give rating surely

