Q: 2. A once-common unit of pressure (still used in the USA) is psi (pounds per square inch). One psi…
A: Gas constant R has defferent type of units and thus the value of this gas constant depends on the…
Q: Determine the oxidation state of the transition metal in each of these coordination compounds.…
A: Oxidation state of transition metal in coordination compound can be known by substituting oxidation…
Q: certain substance has a heat of vaporization of 52.85 kJ/mol. At what Kelvin temperature will the…
A: Given-> Heat of vaporization (∆Hvap) = 52.85 KJ/mole ∆Hvap = 52850 J/mole T1= 287 K P1=P P2 = 3…
Q: 2. Devise a synthesis of the following compound using 1-bromobutane as the only organic starting…
A:
Q: HH-
A: Answer:(a) an identity matrix is represented by HH-1 .
Q: a) b) d) fa Xy
A: Polymer: A polymer is any of a class of natural or synthetic chemical substances which are made of…
Q: Given the reaction shown , which of the following would cause an increase in the concentration of…
A: According to Le-chatelier's principle when any disturbance is made on equilibrium then it shifts in…
Q: Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free…
A:
Q: What is the pH of a solution that has an H concentration of 2.6 x 10 M? Enter your answer to the…
A: 4) Given, Concentration of the H+ ion = [H+] = 2.6 × 10-8 M pH of the solution = ? The formula used…
Q: A chemistry graduate student is given 500. mL of a 0.40M ammonia (NH,) solution. Ammonia is a weak…
A:
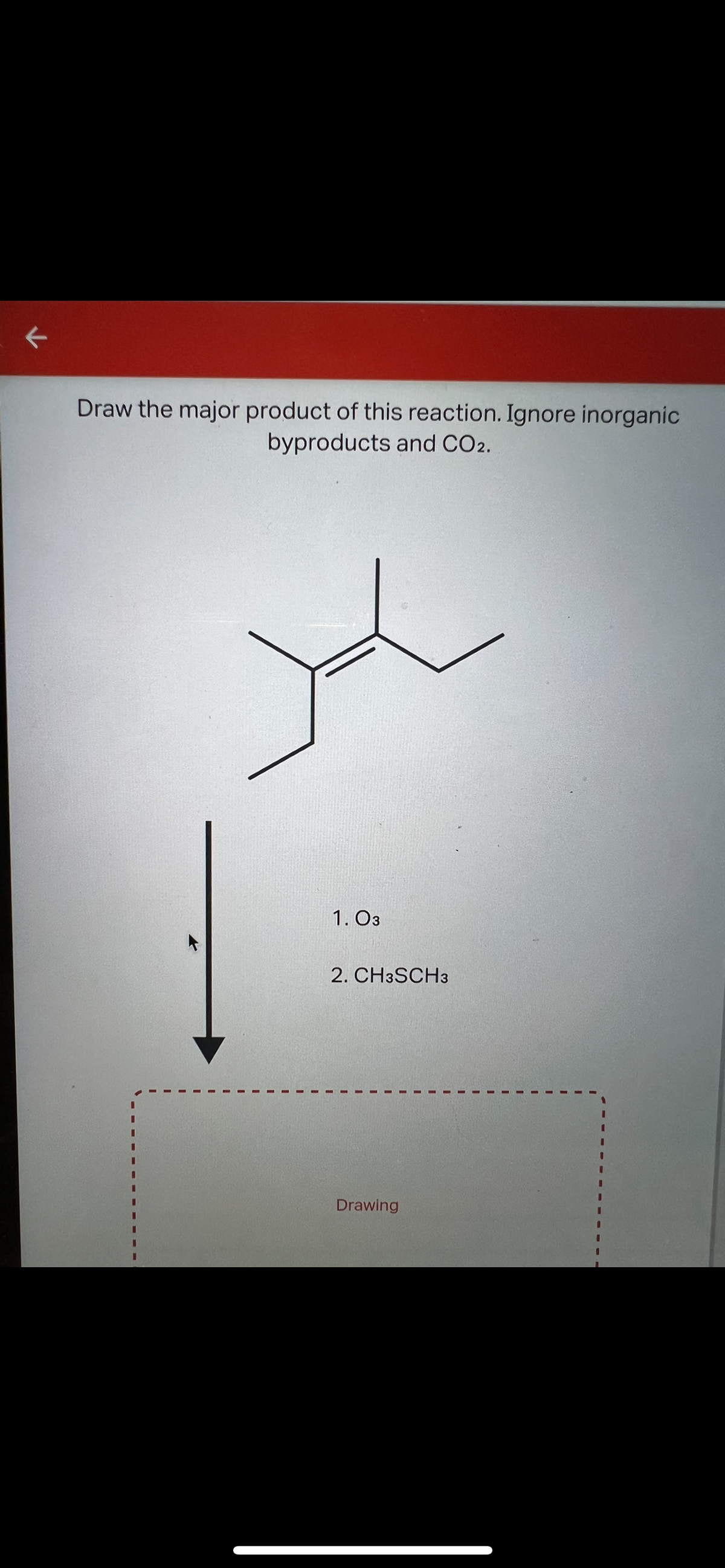
Q: Draw the major product of this reaction. Ignore inorganic byproducts and CO2. 1. O3 2. CH3SCH3…
A: First O3 acts as an oxidizing agent and do insertion of O3 in double bond and then finally converted…
Q: When the ionic compound Li2CrO4 is dissolved in water, which species will react with water to a…
A: Chemical formula of an ionic compound composed of two parts i.e. anionic part and cationic part…
Q: There is a missing reagent, provide a structure for it.
A: Dehydration means water get removed from molecule and alkenes are organic compounds containing…
Q: Consider the galvanic cell described by the following shorthand: Fe2* || Ag* | Ag. Select all true…
A: 1) for the given galvanic cell, Fe|Fe2+ || Ag+|Ag A) Iron metal is being oxidised to iron(III)
Q: Draw the major product of this reaction. Ignore inorganic byproducts and CO2. 1. O3 2. CH3SCH3…
A: ->Ozonolysis of alkene produce carbonyl compound.
Q: What is the predicted product for the reaction shown? H2SO4 HO OH II III IV
A:
Q: When a sample of an ideal gas is compressed at 25 °C from 52.0 L to 21.0 L at a constant pressure of…
A:
Q: Question 3 What is the correct order of operations related to NMR that yields a spectrum? 1 st Step…
A: Proton NMR is used to determine the structure of compound based on the number of adjacent proton.…
Q: Copper has a specific heat of 0.386 J/g°C. a) How much heat (in Joules) is required to increase 5.00…
A:
Q: When the ionic compound CuI2 is dissolved in water, which species will react with water to a…
A: Concept: when an ionic compound is dissolved in water then it form corresponding ion in the…
Q: The following figure is an example of band theory. How many down arrows, electrons, should be in the…
A: Band theory describes the electronic structure and electronic energy level in solids.
Q: Each of the molecules shown below generates two different HNMR signals. Match each molecule with…
A:
Q: If I have a 1% solution of T, how much (in %) will I need to add to a 750kg batch of solution to…
A: Given: 1% solution of T Overall concentration = 0.125% Mass of batch of solution = 750 kg
Q: Draw the major product of this reaction. Ignore inorganic byproducts and CO2. 1. O3 2. CH3SCH3…
A:
Q: 5. Write a Full synthesis of diclofenac sodium in details (the reagents and the name of the…
A: We have find out the mechanism for diclofenac.
Q: Chemistry The reactant concentration in a zero-order reaction was 9.00×10−2 MM after 115 ss and…
A:
Q: What is the chemical formula for the compound formed between vanadium(III) and iodine?
A: Dear student since you have asked multiple questions but according to guidelines we will solve 1st…
Q: How many signals would you expect in the 'H NMR spectrum of the compound shown? 03 4 6 07 LO
A:
Q: What is the chemical formula and Lewis structure for copper (II) ammine complex.
A: Given-> Copper(2)ammine complex
Q: Identify the oxidation agent and reducing agent in the following reaction (equations may not be…
A: The correct answer is given below
Q: For a given chemical reaction, the value of AH is negative and the value of AS is negative. The…
A:
Q: vert 3 16 oF C2 H6 O to 9 mol
A:
Q: Sodium hypochlorite (NaClO) is the active ingredient in bleaching agents. What is the percentage…
A: To calculate percent composition of chlorine , we would use formula % Chlorine =Molar mass of…
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: Here we are required to balance the redox reaction
Q: ČH-OH HO-C-H H-C-OH H-C-OH O C-H CH,O- -0- H-C-OH O ČH;O- -0-
A: Lyases: These are the enzymes that catalyzes breaking of a covalent bond without hydrolisis.
Q: What monomers would you use to produce analogous polyester fibers?
A: Nylon is a synthetic polymer which consist of repeating units that are linked by amide linkages…
Q: draw the mechanism for all 3 steps Propose a plausible mechanism for the following transformation.…
A: Acid chloride reacts with ammonia to form an amide. The reaction completes in three steps.
Q: Calculate the amount of heat needed to boil 143. g of hexane (C,H14), beginning from a temperature…
A:
Q: Calculate the pH of an aqueous solution that contains 0.369 M potassium sulfite and 0.482 M…
A: Given- Concentration of potassium sulphite= 0.369M Concentration of potassium hydrogen sulphite=…
Q: Determine the pH of a 1.0M solution of sodium acetate (NaC2H3O2). Ka = 1.8x10-5 for acetic acid.
A: Dear student, As you have asked multiple question but according to guidelines we will solve first…
Q: Carbon is allowed to diffuse through a steel plate 15 mm thick. The concentrations of car- bon at…
A: We have find out the temperature at which the diffusion flux is 1.43 x 10-⁹ kg/m².s.
Q: 7. How many stereoisomers can you obtain from any type of bromination (not just the one we performed…
A: The number of stereo isomer obtained is given below
Q: Select all of the following reactions that would be expected to provide a negative change in…
A: if the value of ∆ng is positive then ∆S will also have positive value if the value of ∆ng is…
Q: Given the compound shown, draw the acid chloride and nucleophile that will synthesize this compound.…
A:
Q: From the change in internal energy, AE, given for 40 °C, calculate the change in enthalpy, AH, at…
A: Given reaction: N2O2(g) → N2(g) + O2(g) Given ∆E = -151.3 kJ at 40 ∘C We have to calculate the value…
Q: Molecule A is C6H9O2F and an aqueous solution of 0.0130 M of molecule A has a pH of 1.0 . Calculate…
A:
Q: CH3 SOH CH;CH,CCH3 SN1 reaction Br A, SOH = 100% CH3OH %3D B, SOH = 33% CH3OH, 67% H20 C, SOH = 67%…
A:
Q: Tritium is radioactive and decays by a first order process with a half-life of 12.5 yr. If an…
A:
Q: (a) (b)
A: a. 1,2-dimethyl cyclohexane is alkane molecule. 2900 cm-1 sp3 C-H stretch. b. m-xylene : 3050 cm-1…
Q: Find the density
A: As shown in the picture 2a The reading of volumetric flask is 400ml Picture 2b The reading of…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Arrange the following according to INCREASING reactivity towards E2: 1st ( least reactive)? 2nd? 3rd? 4th (most reactive)?Draw the major product of this reaction. Ignore inorganic byproducts. Assume thatthe water side product is continuously removed to drive the reaction towards products. Reagent= (CH2OH)2, TsOHWhat happens to the rate of an SN1 reaction under the following conditions? [RX] is halved, and [:Nu−] stays the same

