Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter21: Benzene And The Concept Of Aromaticity
Section: Chapter Questions
Problem 21.43P: Following is an equation for iodination of toluene. This reaction does not take place. All that...
Related questions
Question
question 8
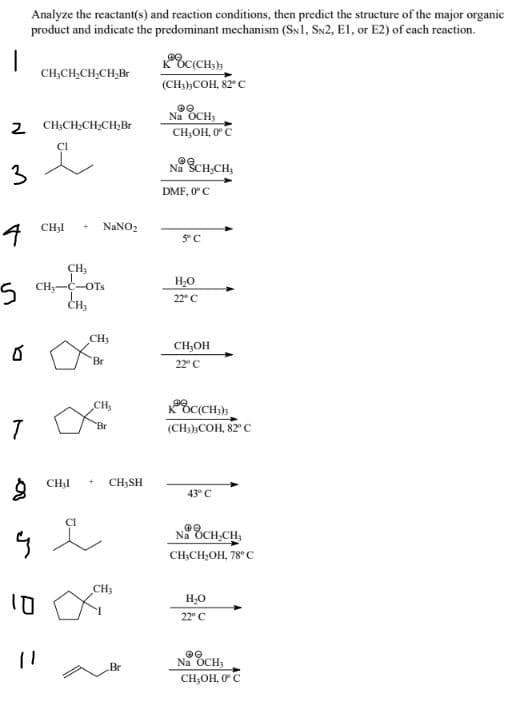
Transcribed Image Text:Analyze the reactant(s) and reaction conditions, then predict the structure of the major organic
product and indicate the predominant mechanism (SN1, SN2, El, or E2) of cach reaction.
CH,CH,CH,CH,Br
(CH;),COH, 82°C
2 CH;CH;CH;CH;Br
Na OCH;
CH,OH, 0°C
3
Na SCH,CH,
DMF, 0 C
CH3I
NANO2
CH3
5 CH;-C-OT.
CH,
H,0
22° C
CH3
CH,OH
Br
22" C
CH,
(CH:),COH, 82°
Br
CHạI
CH3SH
43° C
CI
Na OCH,CH,
CH;CH,OH, 78° C
CH3
H,0
22° C
Na OCH,
Br
CH;OH, 0C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning