Q: Gold-198 is used in diagnosis of liver problems. If the half-life of this is 2.69 days, 1. What is…
A:
Q: Identify the type of reaction represented by each equation. Explain your answers. MnSO4 Mn0 + S03 b.…
A: 1) a) MnSO4 → MnO + SO3 Type of reaction - Decomposition Reaction is a reaction in which the species…
Q: HO NH2
A: Detail mechanistic pathway is given below to carry out the following conversion
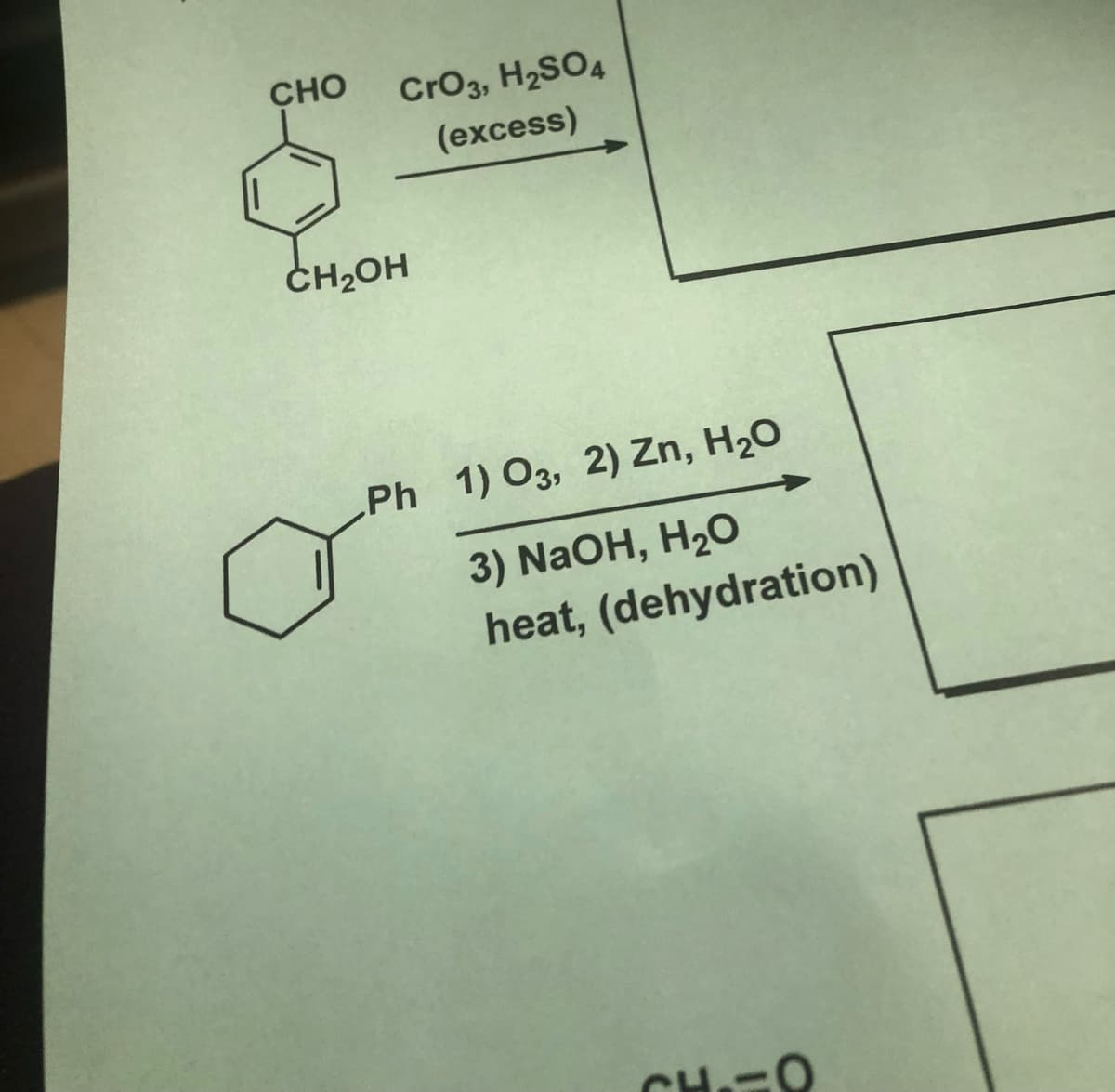
Q: 4. Write the product and give mechanism. (i) *CO (ii) =CH3 RCN.. Rh Me,p ..PMes
A: here we are required to predict the product of the organometallic reaction.
Q: Chemical Equilibrium 3. Kc=0.10 for 2ICl(g) → I2(g) + Cl2(g). What are the concentrations of each…
A:
Q: What is the molar solubility of Ag,co, in an aqueous solution that is buffered to a pH of 4.07?…
A: According to the problem, the aqueous solution of Ag2CO3 is buffered to a pH of 4.07. From the…
Q: Strontium-90 is one of the products of the fission of uranium-235. This strontium isotope is…
A:
Q: ) CH - 20CH; (2) CH CH4 - CH. -C,H (3) CaH -CHa +H. (4) He +CH, -C,Hs Họ (5) H+ C3Hg C2H
A: Free radical mechanism takes place by following steps- Initial step (generation of free radical)…
Q: NH3 (aq) + HzО (1) +—> NH +он-
A: NH3(aq) + H2O(l) ----> NH4+ + OH- According to Le-chatelier principle if equilibrium is disturbed…
Q: In the experiment of the cycle of copper reactions, discuss likely reasons for losing or not…
A: The law of conservation of mass was proposed by the scientist Antoine Lavoisier .The context of the…
Q: Identify the following reactions by type and predict the products (any 4): (i) (ii) (iii) (iv) (v)…
A:
Q: Arrange the following according to increasing boiling point.
A:
Q: 18 mL 0.10 M HClO (Ka= 3.5x10-8) was titrated with 0.06 M NaOH. Calculate the pH after the addition…
A:
Q: 1. Calculate the pH of a solution prepared by dissolving 2.13 g of picric acid, (NO2);C6H2OH (229.11…
A:
Q: Balance the equation and calculate the E∘cell of the given reaction Zn(s) + H+ (aq) ----> Zn2+(aq)…
A: Given- Zn(s) + H+(aq) -> Zn2+(aq)+H2(g)
Q: Given a 1.00 L solution containing 1.00x10-2 M each of Ca+2 and Ag+. A 1.00 M Na2CO3 solution is…
A: Substance that has lower value of Ksp , will precipitate first. Solubility product KSP is the…
Q: 32. Which bond will have the lowest bond energy: C=C, C-C, C-N, C-S? А. C=C В. С-С С. C-N C-S All of…
A: Answer: Bond energy is the specific characteristics of two atoms forming the bond.
Q: What quantity in moles of nitrogen gas at 130.0 °C and 13.3 atm would occupy a vessel of 15.8 L? mol
A:
Q: Why are polymers used in controlled drug ?delivery system? How effective are they
A: Answer - According to the question - A polymer is any of a class of natural or synthetic substances…
Q: Complete and balance the following equation: Cr2O2−7(aq)+CH3OH(aq)→HCO2H(aq)+Cr3+(aq) (acidic…
A: The given redox reaction is as follows: Cr2O72- + CH3OH → HCO2H + Cr3+ A redox reaction is balanced…
Q: 4. Write the product and give mechanism. (i) *CO Et (ii) =CH3 PMes Rh H, RCN/. MesP
A:
Q: 2. ond DrNuy the
A: I guess there is a mistake in the formula given. It should be Li4(Mo2(CH3)8.
Q: 40 198 -1
A:
Q: For this lab, we assumed the final product was pure. This is generally a good assumption when the…
A: The amount of oxalate can be determined by titrating it with permanganate ions, such that the…
Q: A solution was prepared by dissolving 12.0 g of glucose (180 g/mol) in 150. mL solution. What is the…
A:
Q: Which of the following statements is/are TRUE about the Calorimetry experiment? I. The…
A: following statements are TRUE about the Calorimetry experiment - I. The reactions were kept…
Q: increases, the boiling points also increase. Which of the following factors is best associated to…
A: 1) As we know, alkanes are non-polar substances. As molar mass increases, surface area increases…
Q: Compute for the concentration of an alcoholic solution of Methylene blue with an absorbance reading…
A: It is an example of Lambert beer Law Here we are required to find the concentration of alcoholic…
Q: What is the temperature in K of 0.500 mole of neon in a 2.0O L vessel at 4.32 bar? R = 0.08314 L·…
A:
Q: pH of the solution that results when 20.0 mL of 0.175 45.0 mL with distilled water.
A: Givrn, Ka(Formic acid) = 3.74 M1 = 0.1750 M Volume (V1) = 20.0 mL V2 = 45.0 mL Using dilution law,…
Q: Write the formation equation for X-(aq), MX(s), and M+(aq). Assume all atoms used are monoatomic…
A: Here we have to write the formation equation for given atoms assuming they are monoatomic solids in…
Q: Group 7. must he add to the 150 mL mixture to get a 20% ethanol mixture? Bernard has a 150 mL of 5%…
A: Given data we have 150 mL of solution : V1=150 mLwhich contain 5 % ethanol : C1=5%and water…
Q: (1R,2R)-2-methylcyclohexanol same as (1R,2R)-2-methylcyclohexan-1-ol
A:
Q: 11. Account for the order of acidity of the following compounds. ÇOOH COOH COOH ÇOOH -NH2 CH3 NO2 K,…
A: ->Acidity is proportional to Ka value (or) inversely proportional to pKa . -> For Basicity we…
Q: H-O H-O H
A: ->Primary alcohol oxidize to carboxylic acid by cronic acid. ->Secondary alcohol oxidize to…
Q: 18 mL 0.10 M HClO (Ka= 3.5x10-8) was titrated with 0.06 M NaOH. Calculate the pH after the addition…
A:
Q: Consider the Table of Unknowns reacting with Group Precipitants provided. Which of the following…
A: For cation analysis, different group reagents are used, such as HCl, H2S, ammonium carbonate.
Q: Refer to the provided image True or False 1. Past equivalence point, all of the silver chloride has…
A: The given titration is performed with 0.12N AgNO3, NaC12 H25SO4,K2CrO4. This is example of…
Q: The pH of a solution prepared by mixing 50.0 mL of 0.125 M NaOH and 30.0 mL of 0.125 M HNO3 is…
A:
Q: A 30.0 g sample of chromium is heated to 100.0 °C in a boiling water bath. The sample is added to a…
A:
Q: Which statement best describes the reaction TiCl4(l) + 2H2O(l) --> TiO2(s) + 4HCl(aq) at a low…
A:
Q: What is the pressure in a 6.00L tank with 20.0 grams of nitrogen gas at 385 K? atm
A:
Q: Calculate decay constant and half-time
A: Here we have to determine the decay constant and half life time of the given radioactive…
Q: What is the expected pH of an aqueous solution consisting 150.0 mL 0.2500 M HBr04 mixed with 150.0…
A:
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A: Given table is :
Q: Question 5 What chemical reactions occurring in the ozone layer allow it to remain stable? Chlorine…
A: We have to tell about the chemical reactions that occuring in the ozone layer that allows it to…
Q: Calculate the cell potential for the galvanic cell in w
A:
Q: Solve the following half-life problems. Show your complete solution. 1. Gold-198 is used in the…
A:
Q: Calculate the mole fraction of oxygen in the air. If air contains 18.0 g oxygen gas at 285 K at 1.20…
A:
Q: (1) Describe the difference of hybrid orbitals such as sp, sp? and sp3. (2) Why diamond and the lead…
A: DETAILS GIVEN BELOW
Step by step
Solved in 2 steps with 2 images

- Magnesium +Acetic Acid --> C2HG5+ O2 ---> Iron (III) carbonate -->The maximum dose over 24 hours for OTC (over the counter use) of acetaminophen (medication in Tyleonol) is 4,000 mg. Many OTC and prescription medications contain acetaminophen along with other ingredients. Acetaminophen can be found in many cold medicines and in prescription painkillers such as Norco, which also contains the addictive opiod hydrocodone. Keeping track of sources and amount of acetaminophen is very important for your health. Abuse of Norco due to the opiod can also lead to toxic/lethal doses of acetaminophen. There are now strict regulations on the number of tablets of Norco that can be prescribed (7 day maximum per prescription, not refillable without another prescription). A tablet of Norco contains 325 mg of acetaminophen. Thirty tablets are in a week-long prescription post major-surgery to aid in recovery. How many grams of acetaminophen are in the 30 tablets?An aqueous solution of sucrose (C12H22O11) is prepared by dissolving 7.4045 g in sufficient deionized water to form a 100.00 mL solution. Calculate the molarity of the solution. Do you mind showing work? I would assume that (given) gram weight (times moles present/divided by mole(s) of compound) then divided - again by compound weight will give a percentage, then divded by the .....that's where I'm a little lost. thanks
- In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The resulting crystals are filtered, giving a yield of 23.4– 24.0 g. (75–77%) of 4,4'-dibromobiphenyl, m.p. 162–163° (Note 4). How large of a scale can this reaction be run safely? Is it expensive?a mixture of 20 cm³ of a gaseous hydrocarbon and 80 cm³ O2 is burned. Combustion products after cooling pass into KOH solution, it is noticed that a volume reduction of 40 cm³ appears. Eventually 30 cm³ gas remains. If all volumes are measured under the same conditions, find the molecular formula of hydrocarbon.Dodecane C12H26 is burned at constant pressure with 150% excess air. What is the air-fuel for this process.
- Determine the percentage Fe in a sample of limonite from the following data:Sample : 0.5000g ; KMnO4 used = 50.00 ml ; 1.000ml of KMnO4 is equivalent 0.005317 g Fe,FeSO4 used = 6.00 ml; 1.000ml FeSO4 is equivalent 0.009200 g FeO ( ans 44.59 %)1) Commercial fuming Sulphuric acid (Oleum-H2S2O6) is 99.9% solution. Please convert it into molarity.2) Find out the Volume (dm3) of product (gas) at RTP when 0.58 M, 150 mL NaOH (aq.) reacts with 350 mL, 0.25 NH4Cl.What is the simplest CGS unit for "poise"?
- Your body deals with excess nitrogen by excreting it in the form of urea, NH2CONH2. The reaction producing it is the combination of arginine (C6H14N4O2) with water to give urea and ornithine (C5H12N2O2). C6H14N4O2 + H2O ? NH2CONH2 + C5H12N2O2 [Molar masses: 174.2 18.02 60.06 132.2] If you excrete 95 mg of urea, what quantity of arginine must have been used? What quantity of ornithine must have been produced?In an Orsat analysis the % CO2 of the product of the combustion is 20% . If percent excess air is 30% . find the maximum percentage of CO2 attainable.Na2CO3(aq) + CaCl2⋅2H2O → CaCO3(s) + 2NaCl(aq) + 2H2O(aq) It has been previously determined that : there are 1.50 grams of CaCl2⋅2H2O there are .0102 moles of pure CaCl2 and 1.081g of Na2CO3 is need to reach stochiometric quantities What is the maximum (theoretical) amount of CaCO3 in grams that can be produced from the precipitation reaction?







