Q: SCH3 OCH3 Br N(CH3)2 1 2 3 A) 3>1> 2>4 B) 3> 2>1>4 C) 4 > 2 > 3 >1 D) 1>3> 2 > 4
A: Order the carboxylic acid derivatives from most reactive to least reactive towards nucleophilic…
Q: ?nucleophiles اختر واحدة أو أكثر a. CH4 Ob. Br c. H20 d. NH3 e. H2C=CH2 (-)f. OH (+)g. Br h. BH3
A: The electron rich species are termed as nucleophiles. Nucleophiles can be neutral or negatively…
Q: Which compound below is most likely to undergo SNAr reaction using NaOCH3 as the nucleophile?
A: In this question, as option c has 2 nitro groups present alongside F. It will be most likely to…
Q: of (a) vs. (b) vs. H
A:
Q: 7. Which nucleophile would you expect to add reversibly to an a,ß-unsaturated carbonyl? а. NaBHa b.…
A: Addition of a nucleophile to an α,β-unsaturated carbonyl compound Takes place in two ways; one is…
Q: Which molecule can loose a hydrogen and become a nucleophile? Select one: a. H3C– ECH b. H;C- -CH3…
A: The terminal alkynes or 1-alkynes are weak acids and loose a hydrogen when treated with strong base…
Q: Br is a better nucleophile than HSO4
A: Nucleophilic substitution reaction is the reaction in which one nucleophile substitutes the other…
Q: Identify the stronger nucleophile in the following pair ? CH3NH2, CH3OH
A: The CH3NH2 has a lone of electron over the amine which is unhindered and won’t not donate the…
Q: Solve this
A: A rearrangement reaction is a broad class of organic reactions where the carbon skeleton of a…
Q: B. In each of the following pairs, circle the species that is more reactive toward nucleophilic…
A: We know that greater electrophilicity of the carbonyl, then faster will be the nucleophilic attack
Q: H3C CH3 H3C CH2 H А. В.
A: Here both nucleophiles have one and two acetyl groups respectively and the lone pair (negative…
Q: Identify the stronger nucleophile in the following pair ? CH3CO2−, CH3CH2O−
A: A nucleophile has a negative charge on it which means it consist of a lone pair of electron for…
Q: Rank the nucleophiles in following group in order of increasing nucleophilicity. H2O, −OH, CH3CO2-
A: Water is a weak nucleophile because it is neutral. Since acetic acid is more acidic than water, the…
Q: а) c) 2 eq. H20 .CI Br d) Br + Br Which pair of nucleophile and electrophile will react fastest via…
A:
Q: ?Which of the following is not a nucleophile اخترأحد الخیارات a. NH3 -b. Br c. FeBr3 d. CH30CH3
A:
Q: Which structure is the best nucleophile for an SN² reaction? A. NH3 B. NH2 C. NH4* D. HCN O B
A: The question is based on the concept of reactive intermediates. We have to identify which is the…
Q: Which of the following is a strong nucleophile but a weak base? A CH3CH2SNa B) CH3CH2OH c) CH3CH2ONa…
A: -> CH3CH2SNa is strong nucleophile but weak base. ->CH3CH2OH is weak nucleophile and weak…
Q: a. nucleophile electrophile :0: eiectrophile nucleophile H. OH b. C.
A: Since you have posted a question with multiple sub-parts, I will solve the first three subparts for…
Q: 3. Circle the nucleophile in each pair that undergoes SN2 reaction most rapidly with Ph-CH2-Cl in…
A:
Q: Which of the following are good nucleophiles and strong bases? Choose all that apply. O1. HOC(CH3)3…
A: Nucleophiles are compounds having negatively charged atoms or lone pair tendency. And the compounds…
Q: Which of the following cannot react as a nucleophile? I. CH3NH2 II. (CH3)2N III. (CH3)3N IV.…
A: Nucleophiles: A nucleophile is defined as a substance that donates the electron pair to…
Q: Rate the below nucleophiles in terms of INCREASING nucleophilicity. Explain why. CH3OH CH;0 t-BuO-…
A: As the name suggests, nucleophiles are those reagents that possess attraction for the positively…
Q: Which of the following could act as a nucleophile?
A: Given:To give which of the following could act as a nucleophile
Q: Please answer this question
A: Nucleophiles are the molecules or substances that have tendency to donate electrons or react with…
Q: The reaction rate of which of the following alkylhalides can be enhanced by increasing the…
A: Alkyl halides react with the hydroxide ions to form alcohol. The reaction proceeds by the…
Q: Rank these nucleophiles from best to worst Na- I -I Best (II)>(1)>(III) worst Best (1)>(III)>(II)…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: ?Which of the following is not a nucleophile اخترأحد الخيارات a. CH30CH3 b. NH3 C. FeBr3
A: To determine which of the following is not a nucleophile. CH3OCH3 NH3 FeBr3 Br-
Q: Consider the following groups of compounds. In each series circke the weaker (poorer) nucleophile.…
A: The nucleophilicity of a substrate depends upon many factors such as basicity, nature of…
Q: Which of the following is the STRONGEST nucleophile? -H- -OH I II II IV а. I b. II с. Ш d. IV
A: a) The compound I is the weakest nucleophile because there is no heteroatom as well as it is…
Q: Which of the following is the best (strongest) nucleophile? NH3 H20 NH2 CH30 II II IV Select one:…
A:
Q: Which of the following is the strongest nucleophile ? О а. СНз O b. NH2 С. O C. HO O d. F O e. NH3
A: Nuclephile are species which are able to donate lone pair of electron
Q: Identify the stronger nucleophile in each pair. a.NH3, −NH2 b.CH3NH2, CH3OH c.CH3CO2−, CH3CH2O−
A: -NH2 is a conjugate base of NH3. The amine-based compound is a stronger base than the alcohol-based…
Q: Select which molecule is the better nucleophile in the following pair: a). Br− or Cl− in H2O b).…
A:
Q: Which of the following would be the best nucleophile when dissolved in ethanol? CI Br
A: Ethanol is a solvent in which hydrogen is bonded with Oxygen.
Q: Which of the following is a stronger nucleophile in a polar, protic solvent? O HS
A: The reagent has a negative charge or lone pair of electrons acts as a nucleophile. As the…
Q: Identify the stronger nucleophile in each pair of anions. a.Br− or Cl− in a polar protic solvent…
A: A nucleophile is an electron rich species which consists of lone pair of electron pair or pi bond to…
Q: Which of the following is a weak nucleophile? OH- H2O CH3O-
A: We have to predict weak nucleophile .
Q: 9. Rank the following nucleophiles, in a solution of DMF, in order of weakest to strongest. НО CI…
A: DMF is a solvent known as dimethylformamide. It is a polar aprotic solvent. It doesn’t form hydrogen…
Q: (b) Which of the following will react most slowly with cyanide nucleophile (NC-) in an Sn1 reaction?…
A:
Q: Identify the stronger nucleophile in each pair. a. NH3,−NH2 b. CH3NH2, CH3OH c. CH3CO2−, CH3CH2O−
A: Nucleophile : Molecule which easily donates electron pair are called nuclephile.
Q: Which of the following is the strongest nucleophile? CH3S OH- H20 CH3O¯ NH3
A: Nucleophiles A Nucleophile is a chemical species that donates an electron pair to form a…
Q: Which of the following is not classified as a nucleophile? A acetylide (B) hydrosulfide c) alkoxide…
A: In this question, we will see that which can't act as a nucleophile means not a class of…
Q: Which molecule can loose a hydrogen and become a nucleophile? Select one: a. H3C- ECH b. H3C- -CH3…
A: Solution:
Q: Which is the strongest nucleophile among the following? CN- , OH-, CH3OH, NH3
A:
Q: Which of the following would react fastest as a nucleophile in an SN2 reaction? HOO D OF OSH
A:
Q: Rank the following compounds towards nucleophiles. (1-most reactive - 4- least reactive)
A: The most reactive nucleophile among the following is More the number of attached methyl / Ethyl…
Q: CH31 Na Rate=k[substrate][nucleophile] ОН Br "Br H20
A:
Q: Rank the nucleophiles in following group in order of increasing nucleophilicity. −OH, −NH2, H2O
A: The word 'nucleophile' comes from two words- nucleus and 'philos' means love. Nucleophiles are…
Q: What nucleophile is needed to convert (CH3)2CHCH2CH2Br to each product?
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
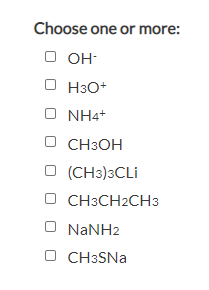
Choose all the nucleophiles
OH- H3O+ NH4+ CH3OH (CH3)3CLi CH3CH2CH3 NaNH2 CH3SNa
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Rank the following groups in order of decreasing priority. a. – COOH, – H, – NH2, – OH b. – H, – CH3, – Cl, – CH2CI c. -CH2CH3, -CH3, -H, -CH(CH3)2 d. – CH = CH2, – CH3, – C ≡ CH, – HRank the following groups in order of decreasing priority. a.−COOH, −H, −NH2, −OH b.−H, −CH3, −Cl, −CH2Cl c. −CH2CH3, −CH3, −H, −CH(CH3)2 d.−CH=CH2, −CH3, −C≡CH, −H2. Give each of the following compounds an appropriate name.a. (C2H5)2Ob. CH3(CH2)2C(OH)(CH3)C2H5c. CH2OH(CH2)3CH(OH)CH3d. CH3CH2OCH2(CH3)C3H7 (n)e. CH3OC6H9 (neo
- Order each of the following compounds from the most disordered (1) to the least disordered (4): H2O(l) NaCl(aq) CH4(g) NaCl(s)Name the following compounds: c. CH3CH2CHNH2CH3 e. CH3CHCH2BrCH3f. CH3CH2CHClCH3 a. CH3OCH2CH3b. CH3OCH2CH2CH3 d. CH3CH2CH2CH2OHRank the following groups in order of decreasing priority. −H, −CH3, −Cl, −CH2Cl
- Place the following compunds in order from lowest to highest boling points. Explain why you have ranked each compound C6H14 CH3OH H2O C3H8What is the empirocal formula for each compound? a. AL2CL6 b. K2Cr2O7 c. C2H4 d.(NH2)2CNH e. CH3COOHWhich of the following molecules has only single bonds. A. CHCHCH3 B. CH2CHCH3 C. CH3CH2CCH D. CH3CH3 E. CH2CH2 Which of the following molecules has a carbon-to-carbon double bond? A. CH3CCH B. CHCH C. CH3CH3 D. CH3CH2CH3 E. CH2CHCH3

