Q: 3.5 Complete the following Grignard reactions. Which molecule serves as nucleophile and which one as…
A: The above complete Grignard reactions are given in attached image.
Q: 1. Identify whether the following substrates preferentially participate in the SN1 or SN2 reaction.…
A:
Q: 33. Which is a better nucleophile? Cl F or d. b. CH3NH2 or CH;OH СО a. H,S or HS C. CO or NH2 NH2…
A: 33. Better Nucleophile: a. H2S or HS- Answer: HS- Explanation: The conjugate base is always the…
Q: Which of the substrates below is the most reactive towards nucleophilic aromatic substitution…
A:
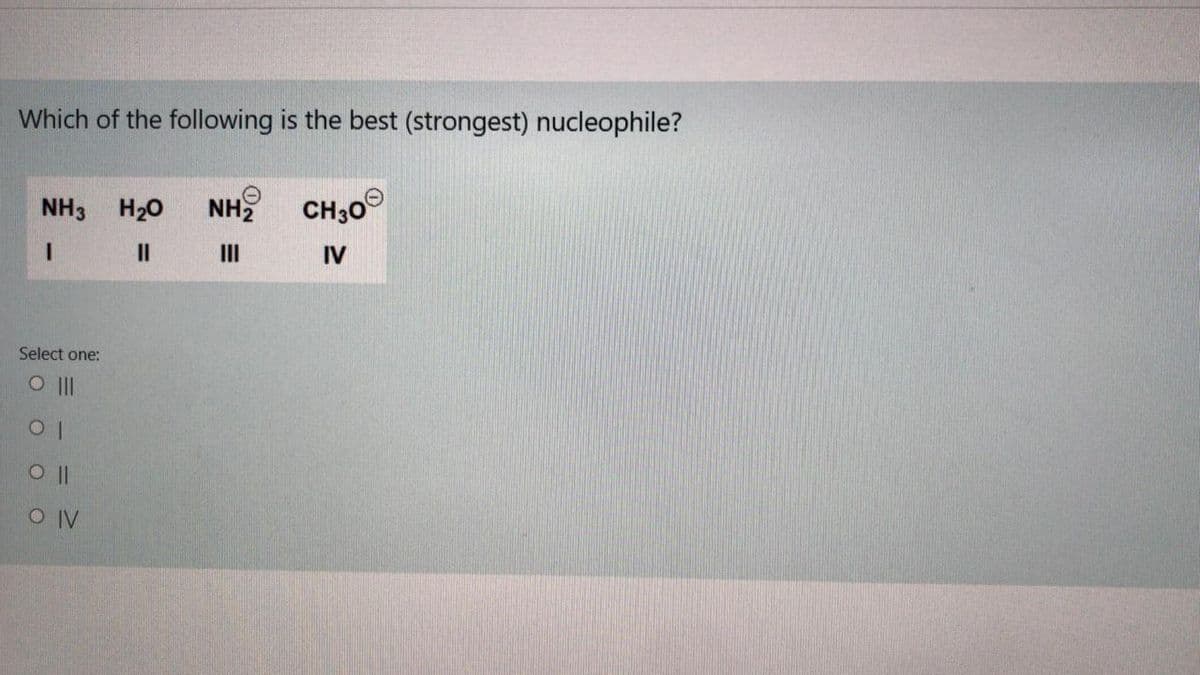
Q: ILe H;CCO H;CO H2O I II III IV
A: Nucleophilicity is the tendency of Nucleophile to donate electrons. Nucleophilicity is directly…
Q: 5:03 LTE O 9. Benzene is not a very reactive nucleophile and must be provided with a strong…
A:
Q: Which of the following anions is the most nucleophilic in polar protic solvents? а. F- b. Cl- С. Br-…
A:
Q: Identify the stronger nucleophile in the following pair ? CH3NH2, CH3OH
A: The CH3NH2 has a lone of electron over the amine which is unhindered and won’t not donate the…
Q: 9. Which of the reagents listed below will convert benzene to methyl benzoate? Methyl benzoate 1.…
A:
Q: Identify the stronger nucleophile in the following pair ? CH3CO2−, CH3CH2O−
A: A nucleophile has a negative charge on it which means it consist of a lone pair of electron for…
Q: CH, — 0— CH, + :0—н (b) СH, —0: + CH, —0— н CH; CH, -T ↑ I-Ö:
A:
Q: Select the reagents that would promote the following reaction most efficiently. A NaOH 1. H2SO4…
A: Applying concept of organic synthesis and application of reagents.
Q: Identify the stronger nucleophile in the following pair ? NH3, −NH2
A: Nucleophilicity is defined as the tendency to give the electrons. Species with negative charge will…
Q: When a single compound contains both a nucleophile and a leaving group, an intramolecular reaction…
A: A nucleophilic substitution reaction is defined as the reaction in which a nucleophile attacks on…
Q: Which of the following is a nucleophile Select the correct response: O2 O CC14 KMNO4 O H2 O NH3
A: We have 5 option , we have to choose correct nucleophile from them.
Q: Which one of the following is the best(faster reaction) base for the 2-methyl - 2-bromobutane E1…
A:
Q: 15) Which of the following species is the least nucleophilic? A) H20 B) BF3 C) (CH3)3N D) CH30- E)…
A: Given compounds: H2O BF3 (CH3)3N CH3O- CN- We have to find the least nucleophilic compound.
Q: In an SN2 reaction, which of the following nucleophiles will produce an ether? "C=CH "OH "O-C-CH3…
A:
Q: Which of the following are not examples of nucleophiles (1) F (2) H* (3) H2O (4) NH3 (5) Grignard…
A:
Q: Which of the following cannot react as a nucleophile? I. CH3NH2 II. (CH3)2N III. (CH3)3N IV.…
A: Nucleophiles: A nucleophile is defined as a substance that donates the electron pair to…
Q: What nucleophile could be used with a primary alkyl bromide to produce an ether? 0 A. Br OB. CH3NH…
A: Given, The nucleophile, which is used to produce an ether from primary alkyl bromide is:
Q: Select which molecule is the better nucleophile in the following pair: a). Br- or Cl- in H2O b).…
A: A better nucleophile is one which can easily donate lone pair and negative charge should be stable…
Q: Which of the following nucleophiles is most reactive in SN2 reactions? I. II. NH, NH II NH- Select…
A: Q1) option B Q2) option A
Q: What is the nucleophile in the following reaction? 2H20 + CH3I → CH;OH + F + H30* O CH3I O CH3OH O…
A: In an organic chemical reaction, there are nucleophiles and electrophiles. These are substances that…
Q: Which nucleophile would be best for converting a tertiary alkyl halide into an alcohol? CH3O CH3OH O…
A: Answer is explained below. Tertiary alkyl halide can be converted into alcohols via nucleophilic…
Q: Which of the following species is likely to be a nucleophile and which an electrophile? (a) CH3CI…
A: Electrophile is species which loves negative charge and nucleophilehile is species which loves…
Q: Which of the following is the strongest nucleophile? OH- H20 H2S HS-
A: Answer :- HS- -------------------------------------
Q: What product is formed when 1-bromopropane reacts with each of the following nucleophiles?
A:
Q: e. No Reaction Which nucleophile will produce the product for the following reaction Br OCH3 O a.…
A:
Q: 6. In the conversion in the box which of the following acts as a nucleophile? excess NH3 CI `NH2 +…
A:
Q: Can you match best nucleophile / conditions that will give a successful hydrolysis reaction for each…
A: For the given electrophile, acid chloride and acetic anhydride, water is used for the hydrolysis…
Q: 19) Azide anion is a very good nucleophile. Predict the major product from the following reaction.…
A:
Q: What product is formed when 1-bromopropane reacts with each of the following nucleophiles? a. HO-…
A: (a) When 1-bromopropane reacts HO-, they undergo substitution reaction.
Q: Arrange the following ketones in order of increasing reactivity towards nucleophiles with the least…
A: The carbonyl compounds give nucleophilic addition reactions. The carbon atom of the carbonyl carbon…
Q: Which of the following is the STRONGEST nucleophile? -H- -OH I II II IV а. I b. II с. Ш d. IV
A: a) The compound I is the weakest nucleophile because there is no heteroatom as well as it is…
Q: of the shown reagents is heeded to carry out the showIn reaction ??? H Br H OH HO НО Н + CH3 °CH3 CH…
A: The unimolecular nucleophilic substitution reaction, SN1 reaction involves the carbocation…
Q: Which of the following species is likely to be a nucleophile and which an electrophile? (a) CH3CI…
A: This question belong to reaction mechanism organic chemistry. Electrophile : all positively charged…
Q: If you wanted to perform a substitution reaction with fluoride (F') as your nucleophile, which would…
A: We know that, F- is favourable for SN2 reaction.
Q: 19. Rank the nucleophilicity for halides: a. F> Cl > Br >I b. Cl>F> Br >I c. I> Br> Cl>F d. I> Cl >…
A: Applying concept of reaction mechanism and reagent.
Q: Identify the nucleophile in the following reaction: 2R'OH + RX → ROR' + [R'OH2]* + X- a. X b. RX c.…
A: Since you asked multiple questions so as per Q&A guidelines of portal I solve first question…
Q: 5. In an organic reaction, which of the following is most likely to function as only a nucleophile?…
A: A nucleophile means a nucleus (proton) loving species. Hence, an species which has maximum tenency…
Q: Which of the following reactions take place more rapidly when the concentration of the nucleophile…
A: Given reactions:
Q: What is the nucleophile in the following reaction? SO IV OI Br I + Na CN II CN ! III 00 + Na Br IV
A:
Q: Which of the following species are likely to be nucleophiles and which electrophiles? OH CH3O CH3CI…
A:
Q: Which one of the following nucleophiles will give mainly direct (1,2) addition in its reaction with…
A: b) HBr : HBr is acid and it's released H+ ion this ion attack on oxygen lone pair and then Br- ion…
Q: 4) Which reactant in each of the following pairs is more nucleophilic? Explain. (i) (ii) (iii) (iv)…
A: A question based on molecules that is to be accomplished.
Q: 1) Identify the strongest nucleophile. Explain. -MgBr -ZnBr Me2CuLi А. В. С.
A: Nucleophile are those which can donate electron pair .So stronger the electron pair donor , stronger…
Q: What nucleophile would be required to effect the following reactions? (a) HO. Br (b) Br
A:
Q: 1c. What is the nucleophile in the following reaction? Br CH;COO Na L0OCCH3 + + NaBr 1d. What is the…
A:
Q: When a single compound contains both a nucleophile and a leaving group, an intramolecular reaction…
A: Intramolecular reaction is that reaction which takes place within a single molecule.
I need the end answer quickly
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- The following reaction has a ΔSsystem < 0 O2(g) → 2O(g) T or F can you explain why this is false?2. In the following reactions identify the nucleophile by writing Nu on the atom that does the attacking. Then circle the atom that is the leaving group in the electrophile. a. b. N Br F + + Na Me Iton MeOH olapidmolo-1 ydw niele, brinsw Insbrta staulis pach Maiy bookWhich of the following is the best nucleophile? A) -OH B) H2O C) MeOH D) All are equal Which of the following is the best nucleophile? A) NH3 B) H2O C) (CH3)3N D) All are equal Which of the following is the best leaving group? A) I B) Br C) Cl D) All are equal
- Rank the nucleophiles in following group in order of increasing nucleophilicity. H2O, −OH, CH3CO2-For problem 8.17, all of the reactions will be SN2. For each reaction, identify and evaluate each nucleophile (strong? weak? Strong or weak as a base?) Also, evaluate each solvent as polar protic or polar aprotic. I recommend drawing the structure of each solvent. (g) Sodium methanethiolate (NaSCH3) in ethanol10. Correct order of reactivity towards nucleophilic addition:- Formaldehyde 2. Acetaldehyde 3. Acetone a) 2 > 1 > 3 b) 1 > 2 > 3 c) 3 > 2 > 1 d) None of the above
- For problem 8.17, all of the reactions will be SN2. For each reaction, identify and evaluate each nucleophile (strong? weak? Strong or weak as a base?) Also, evaluate each solvent as polar protic or polar aprotic. I recommend drawing the structure of each solvent. (d) Sodium cyanide in dimethyl sulfoxide (e) Sodium azide in aqueous ethanol (f) Sodium hydrogen sulfide in ethanolWhich of the following is the best nucleophile in an SN2 reaction? Group of answer choices H2O (CH3)3CO- CH3O- CH3OHConsider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:
- Draw the products and show the mechanism for the reactions below. Between each reactionpair indicate which one will proceed faster and explain whyHello 9. Propose a mechanism for the following reaction. H₂CRO4, H₂SO4 OHIf anhydrides react like acid chlorides with the nucleophiles, draw the products formed when each of the following nucleophiles reacts with benzoic anhydride [(C6H5CO)2O]: (a) CH3MgBr (2 equiv), then H2O; (b) LiAlH4, then H2O; (c) LiAlH[OC(CH3)3]3, then H2O.

