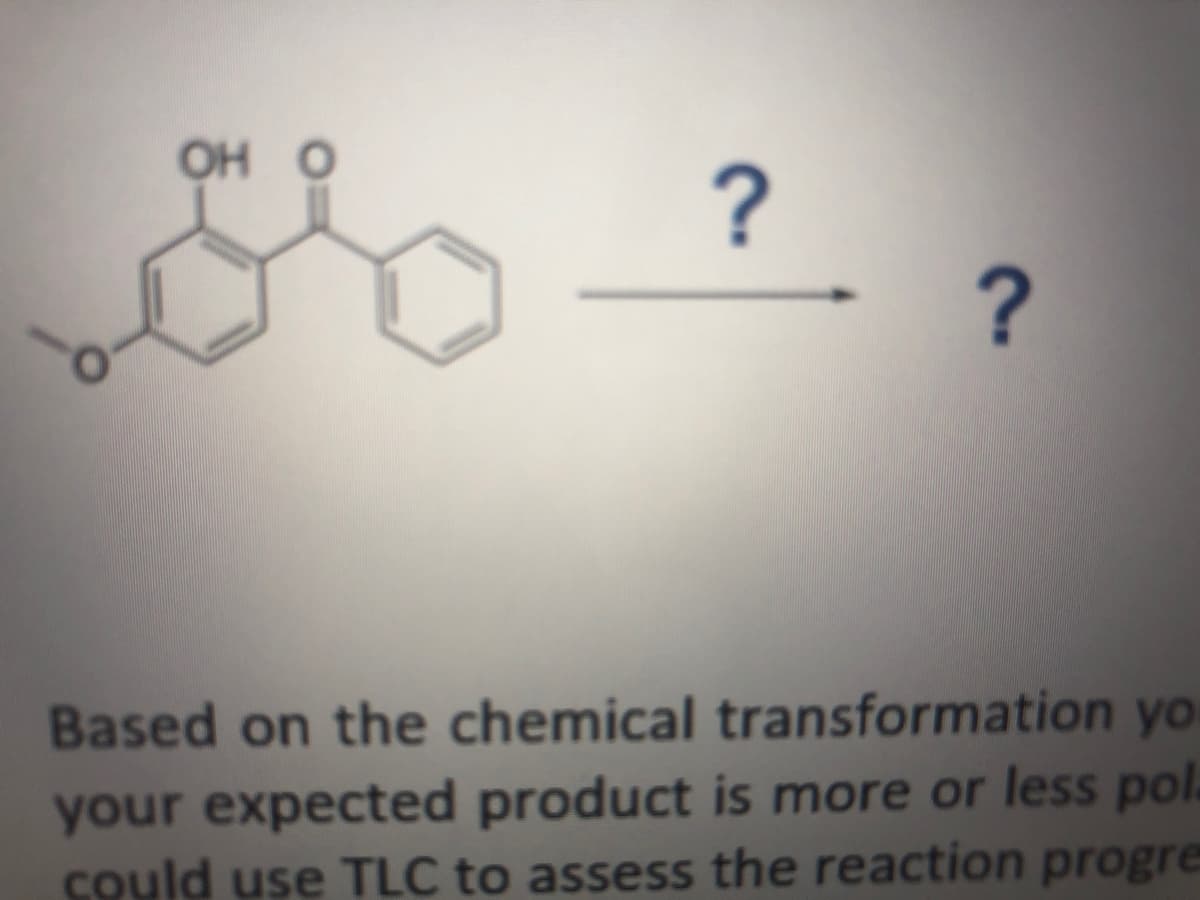
Q: Draw a stepwise mechanism for the following reduction. HO [1] LIAIH, HO [2] H20 HO.
A: LiAlH4 is lithium aluminum hydride which is a reducing agent which helps in reduction of the…
Q: What product result from nitration of benzonitrile ?
A: Reaction in which there in introduction of nitro group is called as nitration reaction. It is…
Q: prepare p nitrobenzyl bromide from benzen
A:
Q: What product result from nitration of benzaldehyde ?
A: The electrophilic aromatic substitution reaction is an organic reaction, in which an atom attached…
Q: Benzylamine synthesis from benzaldehyde follows the mechanism of reductive amination BECAUSE…
A: Statement 1: Benzylamine synthesis from benzaldehyde follows the mechanism of reductive amination…
Q: each molecule Write down the reduction reaction using appropriate reagents
A: Solution: We know lithium aluminum hydride (LiAlH4) is the strongest reducing agent and almost all…
Q: Devise a synthesis of each compound from the given starting material(s). Albuterol is a…
A: a. Synthesis of albuterol is given below, The first step involves the Friedel Crafts acylation…
Q: Show how you would synthesize octanal from each compound. You may use any necessary…
A: Aldehyde is oxidized with alkaline KMnO4 or CrO3/H2O to give acid.
Q: Which pair of compounds would be synthetically useful when treated with NaOH? CHO
A:
Q: Draw the most stable enol form for this structure
A: In keto-enol tautomerism, equilibria lies towards keto form which is more stable than enol form.
Q: Draw two different ways to prepare each ketone from an acid chloride and an organocuprate reagent.
A: a) Please find below the two different ways to prepare the ketone from an acid chloride and an…
Q: Explain the general reaction of enolates—reaction with other carbonyl compounds ?
A: Applying concept of aldol reaction.
Q: Use Gabriel synthesis method to prepare 1-propanamine
A:
Q: n-acetylazoles go through hydrolysis more than regular amides. propose a reason for the reactivity…
A: Amide hydrolysis is not so easy because of the donating ability of lone pair of electrons on…
Q: Acetanilide is otherwise known as phenylacetamide. Acetanilide is formed from the reaction between…
A: Acetanilide is organic compounds and contains amide functional group
Q: Which of the following reagents will not undergo a substitution reaction with the benzenediazonium…
A: Given A. KIB. H3PO3 , H2OC. H3PO2 , H2OD. CuCNE. Cu2O, Cu2+, H2OReagents will not undergo a…
Q: Draw a stepwise mechanism for the following reaction. This reaction combines two processes together:…
A: A mechanism for the given reaction can be shown as
Q: Draw a reaction scheme for the reduction of benzil to hydrobenzoin. Draw the complete mechanism for…
A:
Q: Shown below is the tetrahedral intermediate in a nucleophilic acyl substitution reaction. Draw the…
A: In this equation, we want to draw starting material ( carbonyl reactant ) on left side box and also…
Q: Which of the following can be used for ring-opening reactions of epoxides? a Water b Grignard…
A: A multiple choice question based on introduction to organic chemistry that is to be accomplished.
Q: Devise a synthesis of each compound from the given starting material(s). Albuterol is a…
A: Given that,
Q: Draw the mechanism for nitration of phenylacetic acid. Include formation of the electrophile. The…
A: Given reaction, Nitration of phenylacetic acid
Q: When Compound A and Compound B are reacted with NBS both undergo substitution reactions to yield…
A:
Q: the carbonyl donor or precursor in the following reaction is: a. A b. B c. C d. NaOH
A: Precursor is the compound that participates in the chemical reaction to give another compound. We…
Q: Draw any Phase II metabolite of:
A: In phase II metabolism, coupling of drug or its metabolite to an another molecule like acetylation…
Q: The product of saponification is called salt. Explain why.
A: Reverse esterification that is the hydrolysis of ester compound is called saponification reaction .…
Q: The major reason that phenol is a more acidic than cyclohexanol is It is a better proton donor The…
A:
Q: Explain Claisen reaction ?
A: In organic chemistry, the Claisen reaction is a coupling reaction where the carbon-carbon bond…
Q: Draw the structure of both an acid chloride and an ester that can be used to prepare each compound…
A: Acid chloride is an functional group whose molecular formula is RCOCl, where R is an any alkyl or…
Q: What Grignard reagent and carbonyl compound can be used to prepare the antidepressant venlafaxine…
A: The crabonyl compound and the grignard reagent that can be used to form venlafaxine is as follows:…
Q: Match the reactions with the starting material or product. Match each item to a choice: Sandmeyer…
A:
Q: Draw out the overall balanced equation for the iodination of 4'-Hydroxyacetophenone to form the…
A: The iodo substituted product is formed by treating the 4-hydroxyacetophenone,by first the base,which…
Q: reagents
A: In Negishi cross coupling Reaction, Aryl halide or vinyl halide react with Organozinc to form…
Q: Draw and explain the mechanism for the ammoxidation of propene which leads to the formation of…
A:
Q: What product is formed when reacted with aqueous NaOH?
A: Carboxylic acid derivatives are hydrolyzed back into carboxylic acid or its conjugate base in the…
Q: What carbonyl compound and amine or alcohol are needed to prepare attached product?
A: The organic compounds which contain -C-O-C- bond are classified as ethers. The oxygen atom bonded to…
Q: Identify the acetal in oleandrin, and draw the products formed by acidcatalyzed hydrolysis of the…
A: The net reaction for acetal formation is given below-…
Q: The least reactive carboxylic acid derivative is carboxylate itself. In biological systems, the…
A: The acyl phosphate ester acts as a highly reactive acyl derivative like acyl chloride and its…
Q: Phenylacetone can form two different enols.(a) Show the structures of these enols
A: Enol is a structure that has a double bond with a hydroxyl group attached to one of the olefinic…
Q: What will happen if a carboxylic acid reacts with a Grignard reagent? Draw and explain the…
A: The mechanism is as follows:
Q: i. Synthetize nicotine from nicotinic acid
A: Synthesis of nicotine from nicotinic acid
Q: Explain nucleophilic addition to the aldehyde to form analcohol ?
A: Given: Explain nucleophilic addition to the aldehyde to form an alcohol.
Q: Which of the following reaction yields a carbonyl-containing product? a Ozonolysis b Mild…
A:
Q: The starting material ethyl acetoacetate contains two carbonyl groups. Explain why only one reacts…
A: 1,2-diol is used to block the carbonyl group. The more reactive carbonyl group reacts first .Thus…
Q: How to synthesize phenyl methyl bromide
A: Interpretation - To tell about how to synthesize phenyl methyl bromide - Introduction - Phenyl…
Q: What product result from nitration of benzenesulfonic acid ?
A: SO3H group in benzenesulfonic acid is deactivating group. So, it activates meta position for…
Q: Draw a stepwise mechanism for the following reaction. This reaction combines two processes together:…
A: Given reaction is,
Step by step
Solved in 2 steps with 2 images

- A student planned to utilize HPLC to separate her chemically synthesized product (1) from th reactants (2 and 3). Q1 - Which mode of separation would be reasonable to separate these compounds? Justify your answer Q2 - Predict the elution sequence for compounds 1, 2 and 3 based on the above information. Justify youranswer.What is the 2,4-DNP test and how it help to determine if the reaction was successful? please type the answerA student planned to utilize HPLC to separate her chemically synthesized product (1) from th reactants (2 and 3). Predict the elution sequence for compounds 1, 2 and 3 based on the above information. Justify youranswer.
- According to the following TLC, choose all compounds that are present in the crude sample, before recrystallization? 1 methyl benzoate 2 triphenylmethanol 3 a non-polar impurity such as biphenylSeperation of Amino Acids by thin layer chromotography Lab. Please answer #12 and #13 as I am very confused. Thank you for your help!Table 1: Selected phytocomponents identified in the plant extract. Retention time, min Name of Compound 10.767 Benzaldehyde 15.790 Azulene 16.002 1-Piperazinecarboxaldehyde 17.935 Phthalan 20.731 2-methoxy-1,3,4-trimethybenzene 24.028 Methylesterpentanoic acid 26.993 Tetradecamethylcycloheptasiloxane 31.753 Hexadecamethyl Cyclooctasiloxane If GC is combined with MS, what data you will add as the 3rd column in Table 1 above. Fill in that data for each of the select identified phytocomponents.
- the % retention of the unnatural base pair drops from 100% to ~50% over the course of 75 hours after an initial pulse of unnatural dNTPs. Suggest at least two mechanistic explanations for the lack of retention – four sentences max.If TLC is performed with 1:4 v/v ethyl acetate:hexane in a TLC chamber and the spots are 4-bromobenzaldehyde, diethyl benzylphosphonate, reaction mixture if wittig reation( 4-bromobenzaldehyde , diethyl benzylphosphonate and potassium etoxide), crude product of this Wittig reaction and purified product of wittig reaction. How will you predict the movement of the spots and thier Rf values. Include a brief explanation for your predictions.What is the rate law expression for the reaction in sodium hydroxide and ethyl acetate? Trial NaOH CH3COOC2H5 Initial Condcutivity of NaOH Solution (uS/cm) Initial Rate (uS/cm)/s) 1 21 ml 2.1ml 1285 24.72 2 21 ml 2.1ml 2399 50.32 3 21 ml 2.1ml 1262 23.72
- What is the 2,4-DNP test and how it help to determine if the reaction was successful?For the reaction below; estimate how a TLC-analysis would look like (assume you have asolvent mixture where A-C separates.a) At the start of the reactionb) After 65 % conversionc) After full conversion of A.d) What is the functional group of each compoundSo I completing a lab report and one of the sections is on TLC analysis. In the experiment, the TLC plate was submerged in toluene ethylacetate and had spots of cinnamon leaf oil, cinnamon aldehyde, and cinnamon bark oil. 9 spots total were seen under the UV light, but different spots appeared following a vanillin stain. Why is this so and what does this mean?



