Q: Give the standard line notation for each cell. (a) 2 103¯¯(aq) + 10 Cr²+ (aq) + 10 H+ (aq) ⇒ 10 Cr³+…
A: Standard line notation : It is the representation of a electrochemical cell in a simplified way.…
Q: In the sulfite SO² ion, what is the formal charge on the sulfur atom? (a) -4 (b)-2 (c) 0 (d) +4 (e)…
A: We have to find the formal charge on the sulfur atom in the sulfite(SO32-) ion.
Q: Outline reactions used in the preparation of the following compounds CeBr3 Pm2(SO4)3.8H2O…
A: The objective of this question is to show the synthesis involves the controlled transformation of…
Q: What is the relationship between the keto and enol forms of acetone? 0 || HC CH3
A: Given compound is acetone which is in keto form.
Q: Using the stability (if necessary), sketch the following dependences, and explain ach plot T, (S, T)…
A: The aim of this question is to explain van der Waals gas, the equation of state includes corrections…
Q: Which of the following pairs can be used to illustrate the law of multiple proportions? O H₂O and…
A: The law of multiple proportions describe that two elements are connected to form more than one…
Q: 2. HO OH OH NaOH NH₂ CH₂ O₂CCH₂CH₂CH3-$ NH3 Biosynthesis of adreneline (epinephrine) 'OH NH₂ H₂O HO…
A: a - Proton Transfer:In chemical reactions, a proton transfer involves the transfer of a hydrogen ion…
Q: Which chair is more stable? о A. Chair 1 B. Chair 2
A:
Q: Br. Delta Draw all the possible monobromo - products for the following reaction? G Br₂
A: The given reaction is an example of a free radical reaction. In the presence of heat, halide…
Q: PQ-4. Predict the major product(s). (A) I Br X = X X X acetone (B) II (C) III (D) I and II
A: Product of following reaction is made by applying appropriate reaction mechanism.
Q: 3. For the following molecules determine the number of IR and Raman active vibrations: SiCla…
A: The main aim of this question is to determine the number of IR and raman active vibrations signals.…
Q: Draw the major organic product of treating y-butyrolactone with DIBALH (-78°C) in ether, then H₂O,…
A: Given structure of -butyrolactone and reagent and reagent is .Draw the product.
Q: Ground state 242 Cm undergoes alpha decay to the ground state as well as some low-lying excited…
A: The objective of this question is to show the ground state of California-252 (2 cm) undergoes alpha…
Q: Complete and balance the following half-reaction in acidic solution. Be sure to include the proper…
A: In an acidic solution, every excess oxygen atom is balanced by adding H2O molecules on the side…
Q: NO₂ OH NO₂ CO CO₂H OH CO₂H
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: Draw EI - MS for CH3CH2CH2CH 2BR Explaining your resonance for the peaks show clearly.
A: The objective of this question is to show the mass spectrum of CH3CH2CH2CH2Br which show to electron…
Q: Count electrons in Complexes: - what is the (i)oxidation state of the metal, (ii)what is the…
A: The main aim of this question is to show the complexes [Co(NH₃)₆]Cl₃ and K₃[FeCl₆] to determine…
Q: Draw the major organic product(s) of the following reaction. CI ● H₂O • You do not have to consider…
A:
Q: Please find the relative voltage.
A: The objective of the question is to calculate the relative voltage.
Q: [3] The following Figure is the electrocapillary curves (A) and differential capacitance curves (B)…
A: The objective of this question is to show the figure (A), electrocapillary curves represent the…
Q: When the Hg2+ concentration is 6.95×104 M, the observed cell potential at 298K for an…
A:
Q: Percent saturation 100 10 0 1 10 100 L [MM] 1000 1. Sketch a) A graded response b) A hypersensitive…
A: The objective of the question is to understand the different types of responses (graded,…
Q: 1. Solid Calcium carbonate (MgCO3) decomposes at high temperatures to form solid Magnesium oxide and…
A: The objective of the question is to understand the decomposition of Magnesium carbonate and…
Q: Compound W, C6H13Cl, undergoes base-promoted E2 elimination to give a single C6H12 alkene, Y.…
A:
Q: Draw a tetramer of this alternating copolymer. H₂N HO, O + OH NH₂
A: A polymer structure that is made up of two or more monomer species is called copolymer.The…
Q: The picture represents kinetic molecular theory of gases. Assuming that the Maxwell - Boltzmann…
A: The objective of this question is to explain the lower temperature and molecular mass of given…
Q: QUESTION 2 Once you have completed the first measurement, increase the temperature till it has…
A: The main aim of this question explain the relationship between pressure, volume, temperature and…
Q: When the Pb²+ concentration is 5.44×104 M, the observed cell potential at 298K for an…
A: The objective of the question is to find the concentration of Mn2+ in an electrochemical cell…
Q: What is the structure of a molecule with that has an integration of 6H, 2H, 1H, and 1H? O…
A: 1H-NMR spectra is used to identify the hydrogen environment of the unknown compound.The integration…
Q: Which set of reagents will best accomplish the following reaction? H OH LT A Br NaBr, acetone H₂SO4…
A: FOR ABOVE QUESTION NaOH ,DMSO IS REAGENT
Q: 2. Draw the most stable chair conformation of the following structures, also name them, use cis or…
A: The objective of this question is to explain the stability of two chair conformations:…
Q: Price the following prescription using Percentage Mark-Up plus Minimum Fee for the first…
A: The objective of this question is to analyze the pricing for the given prescriptions using these…
Q: Draw a structural formula for the substitution product of the reaction shown below. H CH3 NH3 CH3OH
A: "Dear student, as an expert we are only allowed to answer one question at a time and 3 sub parts at…
Q: iii) Find the number of molecules per cubic centimeter and effective diameter of Helium atom. Where…
A: The questions pertain to gas physics and involve the mean free path, number density, and kinetic…
Q: what is the product for the reaction- CH₂. Difficult concept - A topic I had difficulty…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: When the Pb²+ concentration is 5.35×10-4 M, the observed cell potential at 298K for an…
A: The objective of the question is to find the concentration of Al³⁺ in an electrochemical cell…
Q: how can i get value of I( nuclear spin number)? in some question proton has 1/2=I value and 11B is…
A: The objective of the question is to understand how to determine the nuclear spin number (I) for…
Q: The decomposition of N₂O5 can be described by the equation 2 N₂O5 (soln) →→ 4 NO₂ (soln) + O₂ (g)…
A:
Q: Choose a structural formula for an alkene with the molecular formula C5H10 that reacts with HCl to…
A: The objective of this question is to show the Chlorination in alkene and formed major product. The…
Q: For determining the %w/w of salicylamide and caffeine in an unknown sample using acetaminophen as an…
A: The objective of this question is to show the choice of wavelength in HPLC analysis should be based…
Q: Identify the monomers required to make each of the following condensation polymers: Arggastly (a) to…
A: The objective of this question is to show the monomer present in the structure of two polymers.
Q: The dissociation of molecular iodine into iodine atoms is represented as 1₂(g) = 21 (g) -5 At 1000.0…
A: The objective of this question is to find the equilibrium concentrations of I2 and I in a chemical…
Q: O2, initiator
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: For which of the following electron transitions in a hydrogen atom does the light emitted have the…
A: Given,The transitions:A. n=4 to n=3B. n=4 to n=2C. n=4 to n=1D. n=3 to n=2C. n=2 to n=1
Q: 8. The kinetic results of the following bimolecular reaction with two different types of catalysts…
A: When the reactant molecules convert into products, they require the minimum amount of excess energy.…
Q: b. HO OH -NH₂ Д
A: The objective of this question is to explain the mechanism of given starting material at heating…
Q: Calculate the vapor pressures of n-hexadecane at 15 °C (in bar and Pa) (1) by using the the boiling…
A: The main aim of this question is to show the vapor pressures of n-hexadecane at 15 °C (in bar and…
Q: The obtained organic extract (3.8 L/min) enters to a second extraction phase where is mixed with an…
A: The main aim of this question is to calculated the metabolite concentration in the spent and…
Q: 1. Compute the binding energy (mass defect) of the carbon atom with six protons and six neutrons…
A: The first part of the question is asking for the calculation of the binding energy (also known as…
Q: Using the above rules, give the IUPAC names for the following: a) b) c) d) t me co
A: Steps of nomenclature : 1) Choose the longest continuous carbon chain as parent chain. 2)The…
Please don't provide handwriting solution
Step by step
Solved in 3 steps with 1 images

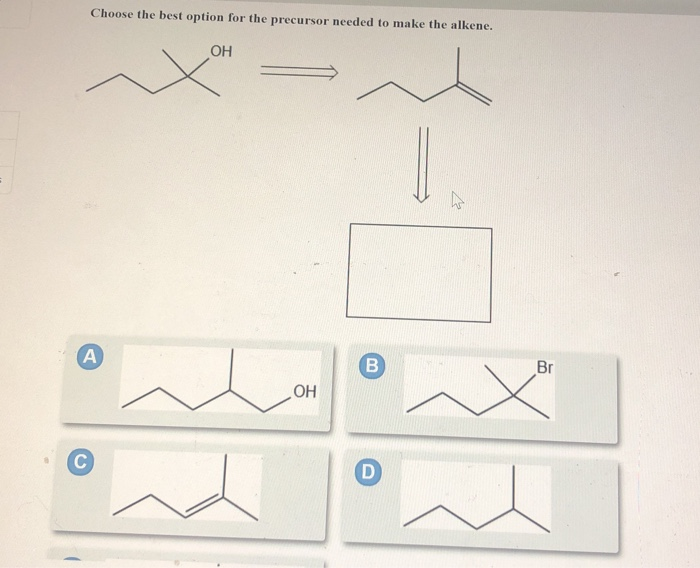
- Which of A to E shows the likely product obtained from the hydrogenation reaction shown below?Draw the products formed when attached compound is treated with HNO3 and H2SO4. State whether the reaction occurs faster or slower than a similar reaction with benzene.What steps are needed to convert Y to the alkene Z ?
- Which alkene choices and reaction conditon created this product?What starting alkene reacted with H2O and H2SO4 catalyst is required to produce 2-methylbutan-2-ol?For each of the steps in Problem 7.26, determine whether the product can eliminate a leaving group to produce a compound that is different from the reactants. For those that can, draw the appropriate curved arrows and the new product that forms.
- Illustrate the Electrophilic Addition of HX to an Alkene ?Explain why the reaction in Problem 18.48 proceeds dramatically more slowly under neutral conditions than under either acidic or basic conditions.Which of the following is the major organic product in the reaction sequence in Image 21? A. b B. c C. a D. d

