Q: Refer to the schematic diagram below, what is compound D? | Conc. H,SO, a-naphthol Conc. HS0, acetic…
A: The answer is as follows:
Q: Q 20 Complete the following reaction: NC. NaOCH3 ELOH Ph NC Ph Ph CN CN Ph Ph NC
A:
Q: What is the ratio of lactic acid (Ka = 1.37x10-4) to lactate in a solution with pH =4.09? Write…
A: Given Ka = 1.37 × 10-4 PH = 4.09 [ lactic Acid ][ lactate ] = ?
Q: What must oxygen atoms be in order to usually act as leaving groups? 1) Oxygen atoms generally must…
A: Oxygen an element of p-Block . Oxygen is an electronegative atom can stabiles negative charge on…
Q: What is the conjugate acid of H2PO4-1? Select one: a. H3PO4 b. HPO4-2 c. PO4-3 d. H4PO4+1
A: According to Bronsted- Lowry A conjugate acid is a substance that is formed when a base accepts a…
Q: Draw the structure of the predominant form of CF3CH2OH (pK, = 12.4) at pH = 14. Interactive 3D…
A: pH= 14 indicates highly basic conditions. Under basic conditions hydroxide proton is deprotected.…
Q: 1. C is basic and is also known as sulfonamide group. * TRUE FALSE 2. A is a an amide and imparts…
A:
Q: Complete the lecithin molecule by replacing each X to show myristic acid (CH₂(CH₂)2COOH) and the Z…
A: We have find out the complete structure of lecithin molecule.
Q: Calculate the concentration of C6H6O62- in an aqueous solution of 4.66×10-2 M ascorbic acid,…
A: Molarity is defined as total number of moles of solute present in 1000 ml of solution Molarity =…
Q: Complete the lecithin molecule by replacing each X to show myristic acid (CH₂(CH₂)2COOH) and the Z…
A: We have to complete the structure of the lecithin molecule A
Q: Determine the identity of compound B based on the tests performed. Compound A, B, C, D Chromic acid…
A: Correct Option : 1-hexanol
Q: Identify the predominant form of the molecule at pH 7. A C H2N, OH H2N H2N, он HNO HO OH HO. HO. HN…
A: We have to predict the predominant form of given molecule at pH of 7.
Q: Bromocresol green Yellow to blue during weak acid to strong base( 5.70 pH, 9.8 mL), blue to yellow…
A: Neutralization reaction of methyl amine and nitric acid: CH3NH3++H2O↔H3O++CH3NH2 kb of methylamine:…
Q: Heptane-1,7-dioic acid is commonly known as O A. pimelic acid O B. adipic acid C. malonic acid O D.…
A:
Q: OBr a) Heptanamide 1) (Me)>CuLi b) MeHC-CHCOMC 2) Н.О - OH (aq) c) 2 Acetaldehyde
A: Note: According to our guidelines we are supposed to answer only first three subpart
Q: Predict the product for the following reactions. a. CH3 HC/ZnCl2 -CH3 OH
A:
Q: How much 0.5 M KOH must be added to 200 mL of 50 mM glycine at ph 8.6 to bring its ph to exactly…
A: Henderson equation of buffer pH = pka + log salt/ acid
Q: Ка(НС-Нз О2) %3D 1.8 х 10 5 1. 0.10 M acetic acid/0.30 M sodium acetate pH : 2. 0.30 M acetic…
A:
Q: _____________________________________
A: Complete reaction is ; 5C2O4^2- + 2MnO4^- + 16H^+ ------> 2Mn^2+ + 8H2O + 10CO2 In this…
Q: Aşağıdaki bileşik çiftlerinin hangisi tepkimeye girdiğinde sonuç ürünü 2-metil-2-pentanol olur. Oa.…
A: Reaction of an aldehyde with Grignard's reagent yields primary alcohol whereas reaction of ketone…
Q: In 100 g/L 3-bromobutanamidesolution, Calculate concentration (g/L) of oxygen required to completely…
A: 3-bromobutanamide CH3CHBrCH2CONH2 when it is combustioned with excess of air or oxygen…
Q: (a) H,CO3 (carbonic acid) acidic nonacidic (b) H2C3H,O4 (glutaric acid) acidic nonacidic (c) HC;H3O2…
A: An acidic substance is the substance that can give H+ ions. They are corrosive and sour in nature.…
Q: CH3 1.BH3/THF a) 2. H2O2/-OH
A: The given reactions involve various organic reactions like hydroboration-oxidation, reduction,…
Q: Consider this scheme. Which species could be used to reverse the formation of cyanohydrin? A H20,…
A:
Q: HClag) NaOH (ag) NaHCOҳag) Sample Water ++ ++ ++ ++ ++ ++ Legend: ++ → completely soluble; +→…
A: The solubility of a compound can be given as the maximum amount of a substance that can be dissolved…
Q: Oxidation of Butanol -1 in KMnO4/H+ will produce-------------.
A: Potassium permanganate is one of the most important oxidizing agents which is widely used in the…
Q: The correct product of the following reaction is -Br FC- -CIC-Ph -CC-Ph nBuLi -Br -CC-Ph Fam -Ph
A: n-BuLi can abstract an acidic proton or can undergo halogen exchange reaction. n-BuLi undergoes…
Q: Identify the strongest acid. Ka 1.29 x10^-3 (tartaric acid, a component of grapes) O pka = 3.86…
A:
Q: Give 2 different one-step methods to convert propanoic acid to methyl propanoate.
A: Here we are required to show two different method to synthesize methyl propanoate
Q: 16. Acetylsalic ylic acid (ASA), C9 H804(s), is used as
A: 1. Molecular mass of ASA (C9H8O4) : = 9 (12) + 8 (1) + 4 (16) g/mol = 108 + 8 + 64 g/mol…
Q: H Но. Ph pyridine || PHCCH2OH PHCHCH=0 heat Ph H OH ОН +
A: Pyridine acts as a base. Abstraction of proton by pyridine produces two alkoxide ions. Protonation…
Q: HO но- H H- OH OH +Resorcinol/dilute HCI 1.) 2.) Sucrose + Copper (II) Acetate/Acetic Acid но- H- но…
A: Carbohydrates are certain types of macronutrient present in foods and drinks. Sugars , starches and…
Q: 1.OML 3.4ML furan maleic anhydride
A:
Q: Evaluate the following IUPAC names. Type Right if the name is correct and put INC if incorrect…
A: Name of the compound is written with the substituents in alphabetical order followed by the base…
Q: Hello, how do I approach this question? Calculate the OH- concentration in a 0.000425 M solution of…
A:
Q: C7H6O3 + C4H6O3 C9H8O4 + C2H4O2 the reaction has 3 acids in it; one reactant and both both…
A: An acid is the one which contains ionisable proton. Aspirin has molecular formula C9H8O4.
Q: Which of the equalities is true when the pH is 7.76? O [H₂A] = [H₂A¯] [HA²-] = [A³-] [H₂A¯] = [HA²-]…
A: Answer: This question is based on the understanding of dissociation of polyprotic acid where as the…
Q: Draw the products formed in attached reaction.
A: The direct bonding of the halogens to the benzene ring produces the halobenzene molecules. For…
Q: 16) 100. 80. 8 Rel. Abundance Rel. Abundance 40. 100. 80.- 60.- 40. 15 (5.4%) A) 2-propenal propanal…
A:
Q: Fill in the box to complete the reactions: 1. ВН3:THF LOH 2. H2O2, NABH4 H2. Pd HO, + en HO
A: Hydroboration–oxidation reaction: The hydroboration–oxidation reaction is a stereospecific two-step…
Q: Biodiesel, a fuel typically derived from plant oils, can be manufactured by treating the oil with a…
A: Biodiesel fuels are an alternative to the diesel fuels, which are much eco-friendly form of fuel.…
Q: Calculate the pH and the equilibrium concentration of C6H6O62- in a 6.39×10-2 M ascorbic acid…
A:
Q: Draw the products formed in attached reaction.
A: The Heck reaction is the reaction of an unsaturated halide and an alkene in the presence of a base…
Q: can function as a Léwis acid. O CH2CH2 O CN O Cr(NO3)3 O CH3NH2
A: Lewis acid which gain lone pair of electron
Q: What is the net charge on Phe-Asp at each pH? a. pH 1? b. at pH 7? c. at pH 14
A:
Q: Draw the organic products formed in attached reaction
A:
Q: Place these in order of most likely to remain reactants (and not proceed to products), where 1 is…
A:
Q: Place the marker to indicate the ether functional group. Check O Check HO alcohol Use the marker to…
A: Given, The ether and an imine functional groups present in the molecules at:
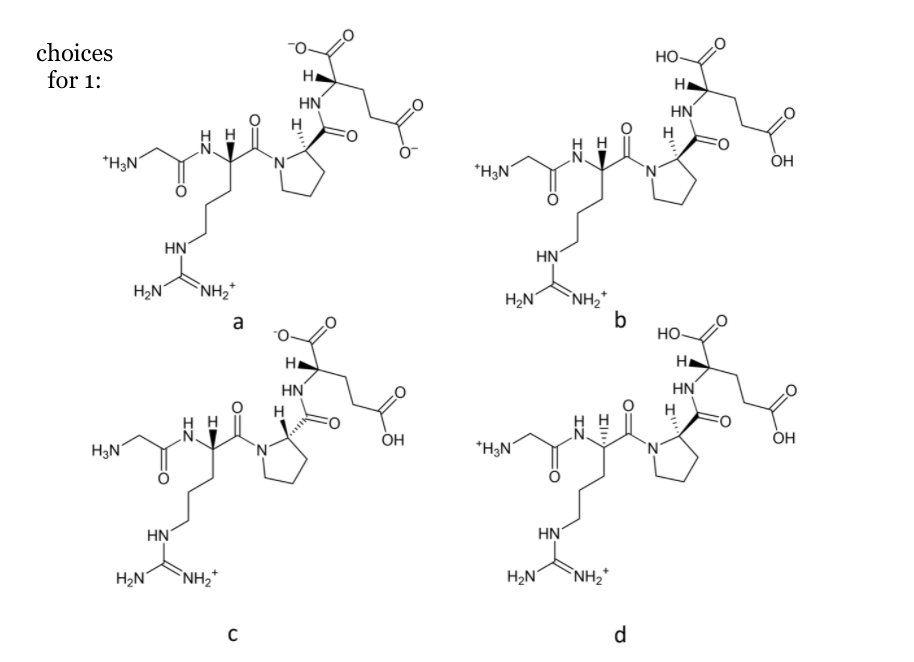
1. Choose the proper structure of the predominant form of G-L-R-L-P-L-E at pH 2. Provide a short explanation.
Step by step
Solved in 4 steps with 2 images

- Hi! I'd just like to confirm if I understood it correctly. :) K = [CS2] [H2]4 / [CH4] [H2S]2HOCI HF HCN H₂SO4 HOBr 2.29 x 10-8 O 2.30 K₂= 3.510-8 O 1.17 x 108 O 1.45 x 10-7 K₂ = 7.2 10-4 K₂ = 4.010-10 K₁ = very large K₂ = 1.2 10-2 Ka = 2.5210-⁹ (COOH)2 CH3COOH C6H5NH2 NH3 K₁ = 5.9 x 10-² K₂ = 6.4 x 10-5 K₂ = 1.8 x 10-5 Refer to Equilibrium Constants. What is the [H3O+] of a solution that is 0.0100 M in HOCland 0.0300 M in NaOCI? Kb = 4.2 x 10-10 Kb = 1.8 x 10-5C9H8O4 +NaHCO3 ------------->???
- still learning how to name correctly. Is this right?Name the compound Cl Cl CH3 - CH - CH2 - CH - CH2 - CH2 / C2H5the context is "Cosmone is a molecule used by fragrance manufacturers to provide a rich and elegant musky essence to many perfumes. Cosmone has the molecular formula C15H26O." I need help on part (e) and (f).
- the context is "Cosmone is a molecule used by fragrance manufacturers to provide a rich and elegant musky essence to many perfumes. Cosmone has the molecular formula C15H26O." I need help on part (h).NaOH (aq) +KHC8H4O4 =aq)NaKC8H4O4 (aq) + H2O (l) balncewhwn naming a compound do ignore the di,tertri and the iso and juat look at the alphebets of the names, like dimethyl and isopropyl;

