Q: Hexanes is a six member carbon chain. C-C-C-C-C-C, whereas Octane is an eight member carbon chain, ...
A: Since both hexane and octane have straight chain structure. Hence the packing efficiency of both the...
Q: Urea, CH4N20, is produced by the reaction between ammonia and carbon dioxide, as shown by the follow...
A: a.
Q: Methyl red has a pk, of 5.0 and is red in its acid form and yellow in its basic form. If several dro...
A:
Q: Aluminum oxide reacts with carbon to form aluminum metal and carbon dioxide. If a reaction uses 21.8...
A: Given, mass of aluminum oxide = 21.8 g Now, the moles of aluminum oxide can be calculated as, ...
Q: What product is expected from this reaction sequence? (A) -CH3 (В) -CH3 1) 2 N2OE 3) H30* EtO CH3 2)...
A:
Q: Explain Expressing the Equilibrium Constant in Terms of Pressure?
A: Equilibrium is the state at which rate of forward reaction is equal to rate of backward reaction. Eq...
Q: consider the equilibrium A(s) = B(s) + C(g) H > 0 predict and explain how or whether the followi...
A: The reaction given is A (s) ------> B (s) + C (g) ...
Q: Please answer #45 and #46
A: Given,
Q: 1. a. i. Write out all the isomers of the compound with molecular formula C4H10O. ii. Select the n...
A: We'll answer the first part of (a) since its not specified which questions answer is required Please...
Q: 1. 1.95 g H2 is allowed to react with 10.5 g N2, producing 2.29 g NH3, according to the unbalanced e...
A: The reaction given is H2 (g) + N2 (g) → NH3 (g) Balancing : since we have 2 N in LHS. Hence making i...
Q: 7. Element X has an electronegativity of 2.0, and element Y has an electronegativity of 3.0. In the ...
A: Given : electronegativity of X is 2 and Y is 3 Molecule is X3Y with central atom Y. Also the central...
Q: Perform the following retrosynthesis. (Provide the reactants and reagents required to convert the gi...
A: The hydroxylation of species alkene will occur in the sight of molecule osmium tetroxide OsO4 along ...
Q: 1. Which of the following can behave as Bronsted-Lowry bases in aqueous solution? H2PO4- HCN NaOH ...
A: Hey, since there are multiple questions posted, we will answer first question. If you want any speci...
Q: Draw a structural formula for the most stable carbocation with each molecular formula. Q.) C5H11+
A: Q.) Given carbocation C5H11+
Q: Which compound in set undergoes more rapid solvolysis when refluxed in ethanol? Show the major pro...
A: Solvolysis is a nucleophilic substitution reaction, in which intermediate is carbocation. Hence more...
Q: Suppose you are told that each reaction is a substitution reaction but are not told the mechanism. D...
A: SN2 reaction: It is the nucelophilic substitution bimolecular reaction. It is single step reaction....
Q: 6. In an organic chemistry lab, chemists were attempting to convert compound I into compound III via...
A: The leaving group attached to 3o carbon. Thus, nucleophilic substitution reaction must be occurred v...
Q: H2 + I2 ⇄ 2HI. Equilibrium constant is 46. Concentration of I2 is 1.0M concentration of H2 is 1.0M a...
A:
Q: What kinds of forces hold each of the three basic categories of atomic solids together?
A: SOLUTION: Step 1: The different types of atomic solids and the types of forces in them are as follow...
Q: An extra strength Tylenol caplet contains 500 mg of acetaminophen (C8H9NO2) as one of its active ing...
A: a) The molecular formula of acetaminophen is C8H9NO2 Hence molar mass of acetaminophen = Atomic mas...
Q: Have each group member explain to the group what happens if a system at equilibrium is subject to on...
A: Le Chatelier’s principle states that if there is a change in temperature, pressure or concentration ...
Q: Compare, with reasons second ionization energy, lithium or beryllium.
A: Energy required to remove loosely held electron present in atom is Ionization energy. Completely fil...
Q: By taking into account electronegativity differences, draw the products formed by heterolysis of the...
A: Cleavage of bond- In organic chemistry cleavage of bond can be classified as-homolytic and heteroly...
Q: Calculate the heat (in kJ) for a 2.80 kg system changing temperature from 85.0 oC to 10.0 oC? The s...
A: The heat is to be calculated
Q: A normal adult jawbone contains 200 mg of Carbon-14 in a living person. If scientists found a jawbon...
A: Radioactivity is the phenomenon in which an unstable nucleus loses energy.
Q: how precipitation helps in extracting magnesium from sea water
A: Magnesium chloride is naturally extracted by seawater by precipitating it to magnesium hydroxide. Th...
Q: Write out all the isomers of the compound with molecular formula C4H10O . Select the normal/ primary...
A: Since the molecular formula of compound is C4H10O And the molecular formula of alkane with 4 carbon ...
Q: Draw structural formulas for the major organic product reaction
A: The reaction of given alcohol with DMP undergoes oxidation secondary alcohol to ketone as shown belo...
Q: If two reactions sum to an overall reaction, and the equilibrium constants for the two reactions are...
A: If two reactions sum to an overall reaction, then the equilibrium constant of the overall reaction i...
Q: annotate the IR Spectrum (including the C-Cl stretch and everything after finger-print region)
A:
Q: Use the mass spectrum of lead shown here to estimate 100% - the atomic mass of lead. Esti- mate the ...
A: INTRODUCTION: Mass spectroscopy is defined as the technique used to determine the atomic mass of an ...
Q: An unknown liquid has a heat of vaporization of 39.42 kJ/mole. If the normal boiling point is 89, wh...
A: Clausius Clapeyron equation-This gives relation between pressure and temperature if enthalpy of vapo...
Q: Questions 2 and 3
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: Complete these SN2 reactions, showing the configuration of each product.
A: SOLUTION: Step 1: The reaction of 1R-bromo-3R-methyl cyclopentane on reaction with sodium acetate on...
Q: Propose a mechanism for formation of product (1), and account for its configuration.
A:
Q: Indicate the order of reaction consistent with each observation.a. The half-life of the reaction get...
A: a) The given observation is the half-life of the reaction gets shorter as the initial concentration ...
Q: Bismuth-210 is an alpha emitter. Write an equation for this nuclear reaction and identify the produc...
A:
Q: Show how to convert 1-butyne to the compound
A: SOLUTION: Step 1: 1-butyne reacts with Na in liq. NH3 to form 1-butene. The alkene is then reacted w...
Q: Which of the following objects has the greatest kinetic energy?a 5000 kgtruck moving at 10 km/h (
A: Kinetic energy = ½ mv2 M = 5000 kg, V = velocity = 10 km /h
Q: What percent of water is in the compound calcium sulfate dihydrate?
A: The formula of calcium sulfate dihydrate is CaSO4 x 2H2O Hence the molar mass of calcium sulfate dih...
Q: Propose a pair of chain propagation steps and show that they add up to the observed stoichiometry.
A:
Q: Image
A: The reaction between MnO2 and Al –metal is shown as: Here, MnO2 and Al react in 3:4 ratio. Given th...
Q: A sample of hydrated aluminum sulfate wing 2.954 g was given to a student to be used in the water of...
A: Assuming the formula of hydrated aluminum sulfate is Al2(SO4)3 . yH2O where y = number of water mole...
Q: Show how alcohol or diol can be prepared from an alkene. Q.) 3-Pentanol
A: Acid catalyzed reaction: It is a reaction which involves breaking pi bonds in Carbon-carbon multiple...
Q: - Draw condensed formulas for the following compounds. a. 3,4-diethyll-2-hexene 5. 5-ethyl-3-heptyne...
A: a) Name : 3,4-diethyl-2-hexene Since the parent chain name is 2-hexene. Hence the parent chain is of...
Q: Samarium -151 is a beta emitter. Write an equation for this nuclear reaction and identify the produc...
A: The general beta decay reaction is AZX -----> AZ+1X + 0-1β
Q: Determining Density and Using Density to Determine Volume or Mass Calculate the density of mercury i...
A: Given, Mass of mercury = 1.00 * 102 g Volume = 7.36 cm3
Q: What number of atoms and what amount (moles) of atoms are in 800.0 g of zinc?
A: Given : mass of Zinc = 800 g
Q: Rank the indicated bonds in order of increasing bond dissociationenergy.
A: Order of increasing bond dissociation energy will be
Q: The compound P4S3 is used in matches. It reacts with oxygen to produce P4O10 and SO2. The unbalanced...
A:
Select the member of each pair that shows the greater rate of SN2 reaction with KI in acetone.
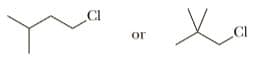
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images


