Q: A carbon atom can absorb radiation of various wavelengths with resulting changes in its electron con...
A:
Q: What mass of glucose (C6H12O6) should be dissolved in 10.0 kg of water to obtain a solution with a f...
A:
Q: Which of these choices best describes the interpretation of the following peak that may be recorded ...
A: Given: 3.7 (2H, Doublet) the given compound which is suitable for given chemical shift value and sig...
Q: Draw the product of nucleophilic substitution with attached neutral nucleophile. When the initial su...
A:
Q: What will be the formula and electron dot structure of cyclopentane?
A: The formula of cyclopentane is C₅H₁₀. This indicates that cyclopentane has 10 hydrogen atoms combine...
Q: Classify each crystalline solid as molecular, ionic, or atomic.(a) Au(s) (b) CH3CH2OH(s) (c) CaF2(s)
A: Each crystalline solid a molecular, ionic, or atomic has to be classified.
Q: which compound is this? how to draw the structure of its oxidation product?
A: The given compound is a type of thiol. The name of the following compound can be given as:
Q: Chemistry Question
A: Hypochlorite ion reacts with water and form hypochlorous acid and OH-. By using Kb (base dissociatio...
Q: Write a general equation for the reaction of an alkali metal with each substance.a. a halogen b. wat...
A: Alkali metals are first group elements. They show a fixed valency of +1. Halides show -1 unit charge...
Q: Formic acid is responsible for the sting of ant bites. By mass, formic acid is 26.10% C, 4.38% H, an...
A:
Q: Is 2-sec-Butyl-1-butene is incorrect? explain why it is incorrect? and if it's incorrect what is the...
A: The name 2-sec-Butyl-1-butene is incorrect has to be determined The reason of its incorrectness has...
Q: Explain Fetal Hemoglobin and Equilibrium?
A: Red Blood cells contains the hemoglobin due to which RBC is red in colour. Hb(Hemoglobin) carries th...
Q: Define Organic Reactions?
A:
Q: Write the expressions for the half-lives of zero-order, first-order, and second-order reactions.
A: Half-life: The half-life of a chemical reaction is the time needed for half of an initial concentrat...
Q: Physiological saline solution has an osmolarity of 0.30 osmol/L. An aqueous solution of sodium chlor...
A: Isotonic solution : When two solutions are separated by semipermiable membrane and no flow of solut...
Q: 3. 25.00 mL of Saturated Solution of KHTar that already contained 0.10 M KCl: NaOH Volumes: Final 6....
A:
Q: Which of the transition elements in the first transition series have anomalous electron configuratio...
A: In order to attain stability some transition metals show anomalous electron configuration. The stabi...
Q: Define Homogenous?
A: A mixture is formed when two different substances are mixed. The homogenous and the heterogeneous ar...
Q: Which of these choices best describes the interpretation of the following peak that may be recorded ...
A:
Q: Give an approximate range of wavelengths for each type of electromagneticradiation and summarize the...
A: a. gamma rays:
Q: What is the volume of a gas in mL at STP if the gas changed conditions to 341 mL at 25.0 oC at 653 ...
A: Given: Initial Pressure(at STP) P1 = 760 torr = 1 atm Final Pressure P2 = 653 torr = 0.8592 atm In...
Q: 8. Draw the condensed structural formula for each of the following alkanes: a. 3,3-dimethylpentene b...
A: a) The name given is 3,3-dimethylpentene Hence the parent chain is of 5 carbon with alkene i.e doubl...
Q: Molybdenum crystallizes with the body-centered unit cell. The radius of a molybdenum atom is 136 pm....
A: Determine edge length of the unit cell:
Q: A substance has a band gap of 0.85 eV at 273 K. Is this substance best classified as an insulator, a...
A:
Q: Write the products of the following sequences of reactions. Refer to your reaction roadmap to see ho...
A:
Q: Explain why 1-propanol (CH3CH2CH2OH) is miscible in both water (H2O) and hexane (C6H6) when hexane a...
A: Propanol belongs to the class of alcohol. Here we ave two solvents water and hexane. Out of which wa...
Q: Consider the reaction:SO2Cl2( g) <-------->SO2( g) + Cl2( g) Kp = 2.91 * 103 at 298 KIn a reac...
A:
Q: The rate equation for the reaction of CH3CH2Br with −OH is: rate = k[CH3CH2Br][−OH]. What effect doe...
A: For a general reaction, A + B ----> C , rate law equation can be expressed as, ...
Q: Homolysis of the indicated C–H bond in propene forms a resonancestabilizedradical. Question: Draw a ...
A: Radical that is generated after homolysis of the indicated C-H bond in propene is shown below. The ...
Q: Why are X-rays used for crystallography? Why not use some other, more accessible type of electromagn...
A:
Q: What is an electron configuration? Give an example.
A: Electron configuration: The complete description of the atomic orbitals occupied by all the electron...
Q: 1. Write out all the isomers of the compound with molecular formula C4H10O
A: Molecular formula of the compound given is C4H10O Since the alkane with 4 carbon will have molecular...
Q: Explain why atomic radius decreases as we move to the right across a period for main-group elements ...
A: In the main-group elements, the atomic radius decreases on moving from left to right across a period...
Q: To what volume should you dilute 125 mL of an 8.00 MCuCl2 solution so that 50.0 mL of the diluted so...
A:
Q: Explain how doping can increase the conductivity of a semiconductor.
A: Conductivity is due to the mobility of electrons; therefore, conductivity can be increased by incr...
Q: The activation energy of a reaction is 56.8 kJ/mol and the frequency factor is 1.5 * 1011/ s. Calcul...
A: Given: The energy of activation= 56.8kJ/mol frequency factor(A) Temperature= 25+273.15K =298.15 K ...
Q: Chemistry Question
A: Given : Half life of first order reaction = 40 days
Q: A solution contains 0.115 mol H2O and an unknown number of moles of sodium chloride. The vapor press...
A: Given,
Q: Solid sodium carbonate is slowly added to 125 mL of a 0.106 M calcium acetate solution until the con...
A: Given : volume of calcium acetate solution = 125 mL = 0.125 L (...
Q: Describe how you would prepare a 250.0 mL buffer solution that has a pH of 7.40 and has a H2PO4 − co...
A:
Q: The azide ion, N3 - , is a symmetrical ion, all of whose contributing resonance structures have form...
A: Resonance structure : This are the equivalent structures that have nearly same energy. The azide ion...
Q: Explain Bond Dissociation Energy ?
A: Given: Bond dissociation energy.
Q: Question in image.
A: The appropriate Lewis structure for NO3- ion is need to be identified.
Q: 1) 12.8 g of NaCl is added to water to make 507.1 g of solution. Calculate the mass percent concentr...
A: Concept: Mass percent concentration = (mass of solute/ mass of solution ) * 100% Molarity = Moles of...
Q: What is the charge of : MoF6 NH3 CBr4 SO3
A: As we can assume net charges on all species are zero. 1.) MoF6 : All Fluorine is having -1 char...
Q: Use Coulomb’s law to calculate the ionization energy in kJ>mol of an atom composed of a proton an...
A: In order to calculate the ionization energy we first write down the given data: Distance of separati...
Q: How to draw any nucleophilic substitution product ?
A:
Q: 1. Write out all the isomers of the compound with molecular formula C,H19O a. Select the normal/ pri...
A:
Q: Aluminum reacts with chlorine gas to form aluminum chloride according to the reaction below. 2 Al (...
A: According to ideal gas law, PV = nRT where P = pressure V = volume n = moles R = gas constant = 0.08...
Q: How Concentration and temperature also affect reaction rate ?
A:
Predict the b-elimination product(s) formed when each bromoalkane is treated with sodium ethoxide in ethanol. If two or more products might be formed, predict which is the major product
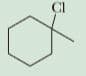
Step by step
Solved in 2 steps with 1 images


