Q: 1. Which of the following formulae represent the same compound? (1) CH,CH,CH-CHCH, (2) CH, CHCH CH;…
A: Rules for IUPAC nomenclature :- Select longest possible continuous chain with maximum carbon atoms.…
Q: 2) On the graph below, draw a potential energy diagram for the following reaction: Q +R + S + T…
A: The reaction given is, => And the data given is,
Q: Write TRUE if the BOLD word/phrase makes the statement correct. Otherwise, write the correct…
A: KMnO4 is strong oxidizing agent and it is purple in color. It can oxidize alcohols to carboxylic…
Q: Draw a mechanism and predict the major product for the following reaction: 1) xs LIAIH, 2) H,O" ?
A:
Q: Explain why chlorous acid (HClO2) has a higher Ka value (lower pKa value) than hydrocyanic acid…
A: Given: Chlorous acid (HClO2) has a higher Ka value (lower pKa value) than hydrocyanic acid (HCN).
Q: these two reactions: Reaction 1: 2OF2(g)⟶O2(g)+2 F2(g)ΔH=−49.4 kJ Reaction 2:…
A: ->A reaction is spontaneous if ∆G<0 ->A reaction is non spontaneous if ∆G>0
Q: 4. Which molecule has a plane of symmetry? CI H CI H. H CH3 H. ČH3 II а. I b. II с. III d. More than…
A:
Q: Calculate the percentage of the reactant that remained after 150 seconds for the following first…
A:
Q: pdf is needed, but algebra & For an instantaneous rate of change, is used to denote geometry can be…
A: Average rate of change is measured over a time interval, whereas instantaneous rate is measured at a…
Q: A group was given the responsibility of performing a recrystallization experiment in their…
A: Solution- Data given- compounds A and B. Solubility in g/100 mL solvent.
Q: Calculate the pH of a solution that is (This problem requires values in your textbook's specific use…
A:
Q: Compute the ionization constant
A:
Q: Which of the following will differentiate one isomer from another? A chemical formula elemental…
A: Which one of the following is correct
Q: ΔfusionH of H2O at 0oC is 6008.2 J mol-1. The values of the heat capacity, Cp,m for the solid and…
A:
Q: Use the Nernst equation to calculate the cell potentials (in V) of the following cells at 298.15 K.…
A:
Q: 3. CH3 CH3OH 4. H20 CI
A: Given reactions are : Determine the mechanism of elimination of each reaction. Draw the products…
Q: CH3 'Br CH3COO'Na* 4. CH3COOH CH3OH 3. 5.
A: We have to give the products, and also the mechanism of the reaction.
Q: QUESTION 7 Products that are synthesized by endergonic reactions tend to O Share electron in bonds…
A: an endergonic reaction is a chemical reaction in which the standard change in free energy is…
Q: Arrange the following energies in order of increasing magnitude: a) the typical energy of a…
A: The bond energy of a covalent single bond, the mean molecular translational energy for a gas at…
Q: 1I. Which of the following is the structural formula for methyl methanoate? A. но В. HH H-C-C-o-C-H…
A: Given structures are : Which of the following is the structural formula of methyl methanoate = ?…
Q: Question 2 1. Given the following equation: AIN + 3H2O --> Al(OH)3 + NH3 If 10.5 mol of AIN and 10.5…
A: Identify the limiting reactant....
Q: Mass Spectrum 100- 1H NMR Spectrum 6H M (74) = 100% M+1 (75) = 4.52 M+2 (76) = 0.28 doublet 80- 2H…
A:
Q: A voltaic cell is constructed in which the anode is a Cr2+|Cr3+ half cell and the cathode is a…
A:
Q: 21083Bi→42He +20681Tl + ________ Complete the nuclear equation above. alpha particle beta particle…
A: The reaction given is, => 21083Bi→42He +20681Tl + AZX
Q: Identify one amino acid from each of the following categories:
A:
Q: Exercise 26. Supply the appropriate reagent or substrate in each of the following reactions. 1. 2.…
A: The given reactions are examples of nucleophilic substitution reactions. In these reactions, the…
Q: 2) On the graph below, draw a potential energy diagram for the following reaction: Q + R → S + T…
A: Given reaction is : Q + R ----------> S + T PE of Q + R (reactants) = HR = 150 KJ PE of S + T…
Q: Determine spontaneity of each reactions. Calculate Gibbs free energy. Sn+4 + Fe+2 → Sn+2 + Fe…
A: Spontaneity of a reaction depends on the value of dG whether it is positive or negative
Q: Two solutions, 100.0 mL of 1.00 M AgNO3 (aq) and 100.0 mL of 1.00 M NaCl (aq), both initially at…
A: Answer: This is a question based on energy conservation where heat lost during the reaction will be…
Q: What is the major organic product of the following reaction?
A:
Q: What will be the complete hydrolysis products of the given structure below? CH2–0– -C17H37…
A: The given molecule is a phosphatidylcholine and it is the source of chlorine in the body. It…
Q: V hen) नीकट मठ्ूभ लाट् ल ।S S.25K10- the abseved the cett potentiat at 298 k +6ज वक्त घहकानकल्लात्वा…
A: Given : We have to calculate the concentration of Cr3+.
Q: The pH of a 0.55 M aqueous solution of hypobromous acid, HB1O, at 25.0 °C is 4.48. What is the…
A: The percentage of ionization of HBrO can be calculated as follows
Q: A gas weighing 23.6 g occupies a 10.0 L flask at 27.0 °C and 740 torr. Calculate the molar mass of…
A: Mass of a gas = 23.6 g Volume = 10.0 L Temperature = 27.0 °C Pressure = 740 torr Molar mass of…
Q: At 25°C the rate constant for the first-order decomposition of a pesticide solution is 6.40 x 10-3…
A:
Q: a. Mix 1 mL of glacial acetic acid, 1 mL of ethyl alcohol and 3 drops of concentrated sulfuric acid…
A:
Q: 3.53 Using the values for the heat of fusion, specific heat of water, and/or heat of vaporization,…
A:
Q: st heat of formation
A:
Q: III. Without reference to any data tables, choose which member of the following pairs has the…
A: Entropy is the disorderness or randommess of the molecules. It is maximum for gaseous molecules and…
Q: Exercise 25. Determine the mechanism of nucleophilic substitution of each reaction. Draw the…
A:
Q: by what percentage does the calculated racah paramater (B) value of 760 differ from the free ion…
A: Calculated value of racah parameter: Bcalculated=760
Q: Draw the protein: SRDR
A: We have to draw the protein: SRDR. Here, S is serine, R is Arginine, D is Aspartic acid.
Q: Will the following amino acid migrate in an electric field? Explain. + H3N- CH-C- CH-CH3 CH2 CH3
A: Given structure is : Will the following amino acid migrate in the electric field = ?
Q: 20.) Industrial production of methanol uses a synthesis gas which is a mixture of carbon monoxide…
A: This problem is based on Hess's law Hess's law states that the enthalpy change or heat evolved or…
Q: 1. Steam distillation results in increased atmospheric pressure. 2. The -CHO group stabilizes the…
A: 1. Steam distillation results in increased atmospheric pressure. TRUE
Q: Calculate the Constant Weight (in grams) of the Empty Crucibles. Show your solution. Crucible No.…
A: In gravimetric calculations, constant weight the minimum attained weight after multiple drying up of…
Q: Which of the following sugars and amino acids are more abundant in nature?
A: More abundant form of sugar and amino acids in nature
Q: The pH of a solution is 1.3. The hydrogen ion concentration of the solution is answer with 4 decimal…
A:
Q: pairs on both nucleophile and leaving group. Exercise 23. Give the products of the following…
A:
Q: what sources occur in a experimental error in a reaction of sodium bicarbonate with hydrochloric…
A: the solution is shown below.
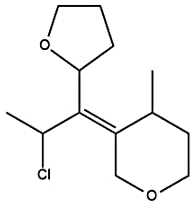
- Identify the configuration of the double bond of the given compound.
- Identify the chiral centers using an asterisk.
Step by step
Solved in 2 steps with 2 images


