Q: What is the effect of the following on the volume of 1 mol of an ideal gas?(a) The pressure is tripl...
A: (a) The pressure is tripled (at constant T) = By using the following formula, we can calculate the v...
Q: What is the effect on the solubility of silver chloride, AgCl, if sodium chloride, NaCl is added t...
A:
Q: Rank the following compounds in order of increasing reactivity in electrophilic aromatic substitutio...
A: Substitution reaction is defined as the reaction in which the displacement of one group by another g...
Q: 2.50 g of sodium acetate, CH3COONa was dissolved in 150.0 mL ofwater, H2O. A solution of 0.025 M eth...
A: Given: Moles of CH3COONa = given massmolar mass=2.5082mol = 0.03049 mol Moles of CH3COOH = molarity ...
Q: 19.
A: Given that:For first rowVolume, V = 43.0 mL= 0.043 L ( since 1 mL = 0.001 L)Number of moles, n = 0.4...
Q: Chemistry Question
A:
Q: A) For the reaction: 2CH4(g)⇌C2H2(g)+3H2(g) Kc= 0.130 at 1613 ∘C. -What is Kp for the reaction at th...
A: T = temperature in Kelvin R = gas Constant = 0.08206 L.atm/mol.K By using this we can calculate the ...
Q: Q4. Study the following reaction and answer the questions? H3C. A OH H,SO4 HO В H3C 1. What is the n...
A:
Q: What is a correct mechanistic step of this reaction?
A: The given reaction is SN2 reaction because the inversion of stereochemistry of reaction occurs . ...
Q: 23.
A:
Q: 5.
A: Mass of chlorobenzene = 1142 gm Molar mass of chlorobenzene = 112.5 gm/mole Number of moles of chl...
Q: 13.
A:
Q: What is/are the major product(s) of this dehydration reaction? OH H,SO, heat 身
A: Organic reaction mechanisms
Q: Europium-141 undergoes a radioactive decay according to the equation provided. a) Balanced the equat...
A: The above reaction is a radioactive decay where a positron emission occurs. In positron emission the...
Q: What is the concentration of a reactant after 28.5 s if the initial concentration is 0.150 M and the...
A: First-order reaction: The unit of rate constant s-1 states that the reaction is a first-order reacti...
Q: The reaction between nickel metal and hydrochloric acid is not a simple dissolution. Explain why?
A: Dissolution reaction means a solute whether in a form of solid, liquid or gas dissolves completely t...
Q: Draw both resonance structures of the anion formed by the reaction of the most acidic C-H bond of th...
A: Draw both resonance structures of the anion formed by the reaction of the most acidic C-H bond of th...
Q: a STIstruciure.com/cour ses/26/1/quZ zes/70679/take The following reaction takes place in a containe...
A: Given: N2(g) + 3H2(g) <----> 2NH3 (g) ∆H = -92kJ/mol According to Le- Chatelier's Principle, ...
Q: What is the major product of this reaction? H3C H CH, Ph t-BUOK t-BUOH Et H3C El Ph O H3C CHs Et Ph ...
A: t-BuOH is a bulky base, it abstracts the β- proton from the less-hindered side and causes the elimin...
Q: When you calculate the unknown concentration of an acid based upon your titration, you determine tha...
A: The concentration of an unknown solution can be calculated by titrating it with any solution of know...
Q: CIS-TRANS Isomers
A: Since you have posted a question with multiple subparts, we will solve only first three sub parts fo...
Q: The reaction 2A -> B is second order with a rate constant of 51/M-min at 24°C. (a) Starting with...
A: Consider the given reaction as; 2 A → B Given information is as follows; A0 = 0.0092 MAt = 3.7×10-3 ...
Q: What is the bond order of the He2+ ion?
A: The bond order of the He2+ has to be given.
Q: föllowing reaction Acetic acid 1 eq. Cl2
A: The given reactant is Benzaldehyde and 1 eq. Cl2.
Q: Alkanes are good organic solvents and miscible in non-polar solvent. Why alkanes are insoluble in w...
A: Given that a question of alkanes is a nonpolar solvent. We have to alkanes are insoluble in water. ...
Q: Identify reagents and catalyst used or major product obtained in each of the following reactions:
A: In this question, we will write a major product and Reagents.
Q: What is the major organic product? 1) LIAID, Et,0 2) H20 OH and MeOH O. OH and MeOH OD and MeOD OD a...
A: This is reduction of esters to give 1°alcohol . This reduction occurs generally in Et2O or THF solve...
Q: (OH] = 3.1x10-10 M Express your answer using two significant figures. ? [H3O*] = M %3D
A: Given [ OH- ] = 3.1 × 10-10 M [ H3O+ ] = ?
Q: What is the ionization energy of a Cl- ion? Express youranswer as a molar energy in units of kJ mol-...
A: The ionization energy pf Cl- ion has to be given,
Q: Write iupac names
A:
Q: 3. Methane (CH) or natural gas is a colorless, odorless gas. Chemical companies add a chemical that ...
A: Q.3: Methane (CH4) is a highly reactive compound and ignites immediately with a small amount of fir...
Q: The MOE results and a discussion pertaining to the conformation of Thioflavin T at low potential ene...
A: Thioflavin T is a type of Biphenyl compound. In Biphenyl compounds, two phenyl groups are connected ...
Q: Describe the standard hydrogen electrode by drawing the relevant figure. Why is this electrode is...
A: The standard hydrogen electrode is also known as a redox electrode and has a platinum electrode at w...
Q: Write the K, expression for the sparingly soluble compound calcium phosphate, Ca3(PO,);. If either t...
A: Sparingly soluble compounds are those compounds which have less solubility in a solvent. Solubility ...
Q: Write a solution plan (without actual numbers, but including the bond energies you would use and how...
A: Enthalpy is the sum of the internal energy and the product of the pressure and volume of a thermodyn...
Q: The following reaction shows the electrophilic addition reaction between analkene compound with hydr...
A:
Q: I need help wirrting and balancing the redox reaction please.
A:
Q: A virus has a diameter of 5.2 * 10-8 m. What is the most appropriate prefix for reporting the diamet...
A: There are names used as the prefixes for the different values which are in the form of 10n and 10-n....
Q: (ii) 2.50 g of sodium acetate, CH3COONA was dissolved in 150.0 mL of water, H20. A solution of 0.025...
A:
Q: 7.35 Arrange the following atoms in order of increasing atomic radius: Na, Al, P, Cl, Mg.
A: Elements are arranged in the periodic table according to the increasing order of their atomic number...
Q: Write a balanced net ionic equation to show why the solubility of PbF,(s) increases in the presence ...
A: Solubility of PbF2 in aqueous solution of a strong acid :
Q: Each of the insoluble salts below are put into 0.10 M hydrobromic acid solution. Do you expect their...
A: We are given compounds and asked for the solubility in 0.10 M HBr and the solubility in a 0.10 M HBr...
Q: The reaction occurring at the cathode in the electrolysis process of the AGNO3 solution is Ag + _ e-...
A: Current I = 12 A Time t = 1.5 hours Ag+ + 1e --> Ag We need to find Ag deposited on electrode...
Q: structure(s) yor organic product(s) of the following reaction. of 1 eq. Cl2 Acetic acid
A: Given reaction: To find: The major product.
Q: A solution contains 9.63×10-3 M sodium sulfide and 1.48×10-2 M ammonium iodide. Solid silver acetate...
A:
Q: 2.60 g of sodium acetate, CH3COONa was dissolved in 150.0 mL ofwater, H2O. A solution of 0.025 M eth...
A:
Q: Choose 1
A: The details solution for this reaction is provided below in attach image.
Q: What is the IUPAC name of this structure? Br O 4-bromo-5-ethyl-2-methylpentan-1-ol O 2-bromo-1-ethyl...
A:
Q: Q3. Draw the Mechanism (stepwise) of any ONE following reactions: Ag(I) 1. NO OH N' H,SO4 NH 2.
A: Mechanism of Beckmann rearrangement reaction. :
Q: 10.
A:
Name the structure.
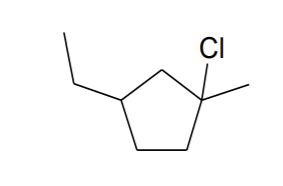
Step by step
Solved in 2 steps with 1 images


