Q: When an acid reacts with a base in aqueous solution, the products always include all that apply.) □…
A: Answer:In this problem we have to identify the characteristics of acid-base reaction, so that common…
Q: b. i. Provide mechanisms for the formation of A, B, and C NaOCH₂ CH30H A ii. Which of the products,…
A: Given An alkyl iodide and a strong base Na OCH3 in methanol solvent.Draw the mechanism
Q: 8.00 -NH₂ Mg(504) CH₂Cl₂ Reflux + H₂O
A:
Q: Calculate the pH and concentrations of CH, NH, and CH, NH; in a 0.0443 M methylamine (CH, NH,)…
A:
Q: Which alkene would you expect to have the smallest heat of hydrogenation? I IV Alkene V Alkene Il…
A: The stability of alkene depends on the number of alkyl groups attached to the carbon atom of the…
Q: Use the blank MO diagram (below) to aid in answering the question. Calculate the bond order for BN.…
A: Bond order from the molecular orbital diagram is calculated by using the following equation as
Q: lease don't provide handwritten solution ....
A: Dehydration of alcohol is acid catalyzed reaction which forms the alkenes.
Q: A buffer solution contains 0.350 M NH 4Br and 0.331 M NH3 (ammonia). Determine the pH change when…
A:
Q: Draw the Lewis structure for PO(OH)3
A:
Q: How many grams of glyoxylic acid and sodium glyoxylate are needed to prepare 1.50 L of a 1.40 M…
A: Given,Molarity of buffer = 1.40 Mvolume of buffer = 1.50 LpH = 4.00pKa = 3.34
Q: Draw the mechanism (curved arrows) of the following reaction that takes a dihalide and eliminates it…
A: Here an organic reaction of formation of alkyne from dihalide in the presence of base is given.The…
Q: 3. Draw the product to each of the following reactions. a. b. d. HO 12 "NH2 HCI, heat حملن جملة لز…
A: The given reactions are shown below.We have to predict the major product of the above…
Q: Use standard reduction potentials to calculate the equilibrium constant for the reaction:…
A:
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A: of is added to of a solution of is added to of a solution that is in both and we have to write…
Q: Rank the following molecules in order of increasing preference for an equatorial placement of the…
A: bulky group prefers to occupy equitorial position
Q: Draw a stepwise mechanism for the conversion of the starting enol to its keto form in acidic…
A: Given is organic reaction. This reaction is known as tautomerization reaction.This is keto-enol…
Q: byproducts. 1.03 2. Zn, HOAc
A: One of the important reactions of alkene is ozonolysis. In the ozonolysis reaction, the…
Q: Predict the main type of intermolecular force that would exist between molecules of the following…
A: The objective of the question is to give the main type of intermolecular force that exists in carbon…
Q: Limonene is present in the rind of lemons and oranges. Based on its structure given below, would you…
A: Limonene has two enantiomers -- one of them present in lemons having citrusy smell and other one is…
Q: Which of the following is the product of the below reaction? H₂O H*
A: A question based on reactions of alkenes. A reactant and its reaction condition are given for which…
Q: Provide the correct IUPAC name for the compound shown here. H Arx
A:
Q: 17. The following reaction fails because: OMe NaOMe DMF is (a) These are conditions for SN2…
A:
Q: Provide the correct systematic name for the compound shown here. NH₂ ů
A: IUPAC rules:- Main chain is the chain containing maximum number of carbon atomsNumbering start from…
Q: Calculate the lattice energy of TIF (s) using the following thermodynamic data (all data is in…
A:
Q: 1. Show the enolate and Predict the major product(s), if any. Account for all carbons. M Ph OCH 3 H…
A: For first case,in the reactant there are two carbonyl group and a methylene group present between…
Q: Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen…
A:
Q: A chemist fills a reaction vessel with 4.47 atm chlorine (C1₂) gas, 1.37 atm phosphorus (P₁) gas,…
A:
Q: Suppose you mix 100.0 g of water at 26.4 °C with 75.0 g of water at 76.5 °C. What will be the final…
A: mass of the cold water, m = 100 g initial temperature of the water, T1 = 26.3°C initial temperature…
Q: A concentration cell similar to the one shown is composed of two Al electrodes and solutions of…
A: Answer:A concentration cell is a type of cell that contains same electrolyte in both compartments…
Q: Using average bond enthalpies, estimate the enthalpy change for the following reaction: H₂(g) + F2…
A: The objective of the question is to determine the enthalpy change for the given reaction.
Q: When the following reaction was run, the resulting organic products B and C were obtained. oft- 1%…
A: Any cyclic secondary halide undergoes elimination reaction presence of a strong base.Here the -OEt…
Q: Draw the structural condensed formula of N,N diethylethanamine
A: The given compound is amine having general formula NR2 where R is Hydrogen or any other group . Main…
Q: Please don't provide the handwriting solution
A: Oximes are a class of imine compounds having functional group present. Generally, oximes are…
Q: organic OH OH diethyl ether aqueous Fill in each of the large boxes above with the exact structures…
A: The question is based on solvent extraction. It uses the principle of nernst distribution law.It…
Q: What is the purpose of washing the organic phase with brine solution? Select one: O a. O b. To…
A: The objective of the question is to give the purpose of washing the organic phase with the brine…
Q: Provide the correct IUPAC name for the compound shown here. CI H
A: IUPAC: Name of compounds follow the rule:Find longest carbon chainIdentify the main group in longest…
Q: What is the pH of an 0.30 M solution of potassium benzoate? Type your answer...
A: We have 0.30 M of potassium benzoate, we need to find the pH of this solution.Ka for benzoate ion =…
Q: what is the form of the integrated rate law which will produce a straight line if the following…
A: The integrated rate law for chemical reactions provides a mathematical expression that relates the…
Q: Provide IUPAC name: - assign cis/trans or E/Z if necessary
A: IUPAC name
Q: 3.459 gof plant food was dissolved in water and mixed with a solution ofMgSO4followed by slow…
A:
Q: 4 Products 6 Products 2 Products 5 Products HBr (1 mole)
A: The given reaction is called an organic synthesis reaction.The reactant carries an alkene function…
Q: Propose a reasonable synthesis for the molecule below. O O HO O
A:
Q: Check the box under each molecule in the table below that is an isomer of this molecule: ОН OH If…
A: Isomers have the same molecular formula but different structures.
Q: Which of the following is the major product of the reaction below taking place at -40 °C? A HBr 1…
A: The given diene is a conjugated diene and it reacts with one mole of hydrogen bromide and forms…
Q: Predict the electron region geometry around atom X for XY₂ that has 16 valence electrons. You will…
A: The total number of valence electrons (A) in XY2 = 16 given that X is the central atom. The total…
Q: 3. According to the Hammond Postulate, what does transition state 3 (TS3) most closely resemble?…
A: According to Hammond's postulate the transition state of a reaction resembles either the reactants…
Q: V 1,3-butadiene H-Br T HBr + H-Br →→ вк →→ •Br 3-bromo-1-butene B 3 Br 4 1-bromo-2-butene GM³ +0₂H
A: Diene give two addition products on reaction with hydrogen halide 1,2-addition product and…
Q: Provide the correct IUPAC name for the compound shown here. 6- 2- (Z)- (E)- 1- 5- 3- 4- iso tri di…
A:
Q: Provide the correct systematic name for the compound shown here. ZI
A:
Q: The product for the following reaction sequence is True False он NaH THF NaOCI 20 OH 0°C
A: Epoxide opening under basic conditionOxidation of secondary alcohol to ketone
Write the mechanism for this reactions. include resonance structures in the mechanism.
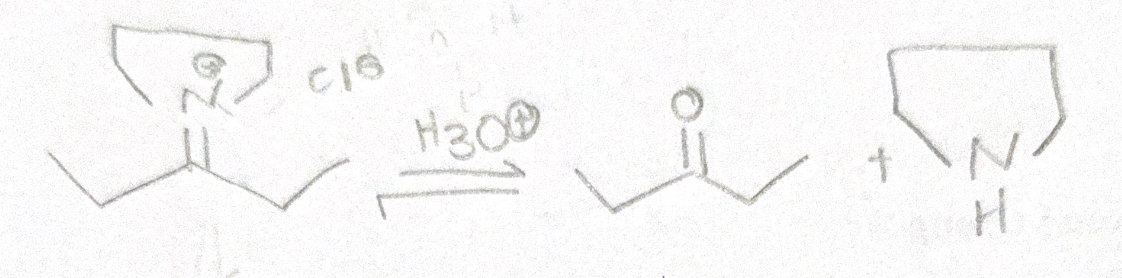
Step by step
Solved in 3 steps with 1 images


