Q: Calculate the number of moles of C2H6 in 5.41×10^23 molecules of C2H6.
A: 1 mole of substance has 6.022x1023molecules or atoms This particular number is called Avogadro's num...
Q: Please give explanation
A: Hess's law states that in a chemical reaction change in enthalpy remains the same irrespective of th...
Q: How will you convert:(i) Propene to Propane-1-ol?(ii) Ehtanal to Propan-2-ol
A: A chemical reaction is symbolic representation of the conversion of substances to new substances. In...
Q: The copper(I) ion forms a complex ion with CN2 according to the following equation:Cu+(aq) + 3CN-(aq...
A: Chemical equation Given Cu+(aq) + 3CN-(aq) ↔ Cu(CN)32-(aq) K = 1.0 * 1011 ................. A Chem...
Q: How many formula units make up 32.4 g of magnesium chloride (MgCl2)?
A: Calculate the number of moles of magnesium chloride. Number of moles = Given massMolar massNumber of...
Q: How can I attach an amine group (-NH2) to a carboxyl group (-COOH)? What are the experimental condit...
A: The reaction of highly acidic carboxylic acid and highly basic amine group leads to a highly unreact...
Q: Would you help me this (5) Thank you
A: A chain reaction involves many sequential steps during a reaction, that is, product of a reaction wi...
Q: How many grams of water will be lost when 8.000 g of YBr3 .9H2O is heated?
A: The question is based on the concept of stoichiometry of reaction. first we write the balance dehydr...
Q: Which compounds exhibit geometric isomerism? Draw andname the two isomers in each case:(a) propene(b...
A: Geometrical isomers are the isomers having the same molecular formula but different orientation of t...
Q: Calculate the standard Gibbs free energy change for the following question
A:
Q: Using the given starting material and any necessary organic or inorganic reagents, indicate how the ...
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for you...
Q: The stereochemistry of bromine addition to a cycloalkene is described as 1) Markovnikov 2) anti-Mark...
A: The mechanism of addition of Br2 to cycloalkene, say cyclohexene takes place as follows:
Q: Show how norbornane can be prepared from cyclopentadiene.
A: Norbornane can be prepared from cyclopentadiene through pericyclic reaction.
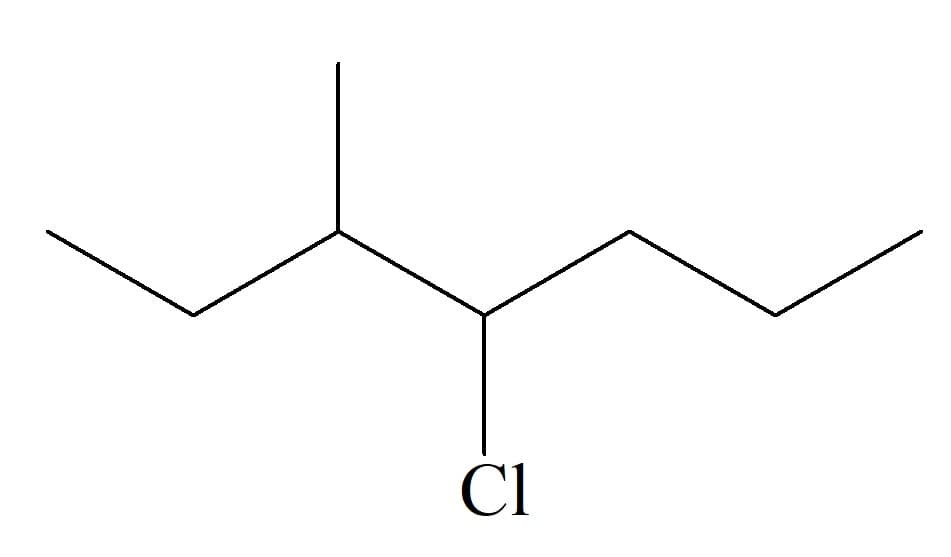
Q: what is the branch name & location? main chain name?
A: Select the longest chain Numbering PWS(Prefix + word root + suffix)
Q: Calculate the work (in kilojoules) done during a synthesis of ammonia in which the volume contracts ...
A: Work done of a system can be calculated by following formula: W=-PextV2-V1 where W is work done, Pex...
Q: calculate the de Broglie wavelength of electrons accelerated through 50kV. (b) Is the relativistic c...
A: Electrons have a rest mass, unlike photons, they have a de Broglie wavelength which is really shor...
Q: He gas at 490mmHg, N gas at 0.75atm, and neon at 520 torr. What is the total pressure?
A: Given, partial pressure of He = 490 mm Hg Partial pressure of N2 gas = 0.75 atm Partial pressure of ...
Q: HELP ASAP
A: Central Atom with One or More Lone Pairs. The molecular geometries of molecules change when the cent...
Q: Draw the enantiomer, if any, for each structure. refer to the photo below
A:
Q: Ethylene can be produced by the pyrolysis of ethane:C2 H6 ⟶ C2 H4 + H2How many kilograms of ethylene...
A:
Q: Choose the larger atom in pair. Sn or Se
A: Arrangement of elements in order of increasing atomic number with recurring chemical properties is g...
Q: An analysis of city drinking water for total hardness was done by two students in the laboratory and...
A: Given, two sets of data. Mean can be calculated as: x = mAX xA+nBX xBmA+nB x = mean of two sets mA =...
Q: Is the attached compound an aromatic? if the compound that not aromatic, state why this is so.
A: Aromatic compounds have the delocalization of pi electrons on a planar and cyclic ring. Aromatic com...
Q: Predict which member of each pair is more soluble in water. Explain your prediction.
A: According to our guidelines, I can answer only first three sub-parts. Because you have posted multip...
Q: Draw zwitterion forms of these amino acids. (a) Valine (b) Phenylalanine (c) Glutamine
A: (a) The Zvvitter ion form of amino acid Valine contains ionisable forms of acidic group (-COOH) as n...
Q: There are two isotopes of an unknown element, X-19 and X-21. The abundance of X-19 is 11.51%. A weig...
A:
Q: (a) Give a mechanism for this reaction, showing how the two products arise as aconsequence of the re...
A: a.
Q: Balance the following equations. a. CaF2 (s)+ H2SO4 (aq)→ CaSO4(s) + HF(g) b. MnO2(s) + NaCl(s) + ...
A:
Q: How do you name an ionic compound if it contains a polyatomic ion?
A: A metal cation will always be the first element in the binary ionic compound, while a nonmetal anion...
Q: Predict the product of the following reaction. PhCH2OH + PCC (pyridiniumchlorochromate) in methylene...
A: PCC i.e., pyridinium chlorochromate is a mild reagent which is used in oxidation, helps in convertin...
Q: Methane clathrate also called methane hydrate is a form of ice in which methane gas has been trapped...
A:
Q: For the reduction of MnO,¯ to Mn²* in aqueous acid, ɛ° = 1.51 v. For the reduction of I, to 21- in a...
A: Answer:- This question is answered by using the simple concept of electrochemistry which involves th...
Q: I am told that your previous answer on my question is wrong by my proffesor, can you review it pleas...
A: For case of dilution following formula is used M1V1 = M2V2 When m1 = Conc. of [H+] in previous solut...
Q: Explain the difference between heat capacity and specific heat capacity.
A: The heat capacity of any substance is the amount of heat that is required to increase the temperatur...
Q: A chemist determined by measurements that 0.080 moles of mercury participated in a chemical reaction...
A: Given that,moles of mercury = 0.080 molesMolar mass of mercury = 200.59 g/molNumber of moles = mass ...
Q: An analysis of city drinking water for total hardness was done by two students in the laboratory and...
A: We are given data : For student A: 228.3, 226.4, 226.9, 227.1, and 228.6. And For student B: 229....
Q: The conversion of cis-1,2-diphenylethene to trans-1,2-diphenylethene is catalyzed when I2 is added a...
A:
Q: Consider the following unbalanced particulate representation of a chemical equation: I= purple F= gr...
A: In the shown representation: purple color represents Iodine atom Green color represents fluorine ato...
Q: Synthesize attached compound from benzene, organic halides with < 5 C's, and any other organic or...
A: Electron withdrawing group are meta-directing. Electron donating group are para- and ortho- directin...
Q: Chemistry Question
A: The result of multiplication or division should be rounded to least number of significant figures pr...
Q: C(CH3)3X +H20 C(CH3)3OH where (X-F, Cl, Br, I) give reaction with each of halogen as well determine ...
A: An organic problem based on SN1 mechanism, which is to be accomplished.
Q: Justify why H2 and O2 left unreacted at room temperature even though they have large -ΔG?
A: This happen due to high bond energies of O2 & H2 molecules.
Q: Identify the circled hydrogens in each of the structures below as homotopic (H), enantiotopic (E), d...
A: Two hydrogen atoms are considered as homotopic if replacing one in turn by another group gives ident...
Q: leaks of hydrocarbon vapors from process equipment and evaporation of hydrocarbons from open areas, ...
A: Emissions that occur from open areas rather than from stacks or vents are called fugitive emission. ...
Q: a) Using the molecular formula shown, calculate the elements of unsaturation for this compound. Sug...
A: (a) The degree of unsaturation can be calculated as follows: Degree of unsaturation = C + 1 - H2 - X...
Q: Predict the output of energy as heat from the combustion of 1.0 dm3 of octane at 298 K and 1 bar. It...
A: The number of moles of octane in the system is calculated as shown below. m=density×volume1 dm3=1000...
Q: Rf value can be matched with to identify the solute Solvent front Solute itself None O Database
A: Given: Retension factor value(Rf value) To find: Identify the solute. Solution: Rf stands fir reten...
Q: As we drive an automobile, we don't think about the chemicals consumed and produced. Prepare a list ...
A: Fuels which are used in automobiles are Petrol, Diesel and CNG (gasoline). Fuels are mostly composed...
Q: Consider the sample of gas depicted here. What would thedrawing look like if the volume and temperat...
A: According to the ideal gas equation, when the volume and temperature of the gas is kept constant, th...
Q: Draw all stereoisomers formed in attached reaction.
A: Interpretation- To draw all the stereoisomers of the given reaction - Introduction- When this ...
what is the branch name & location?
main chain name?
Step by step
Solved in 2 steps with 2 images


