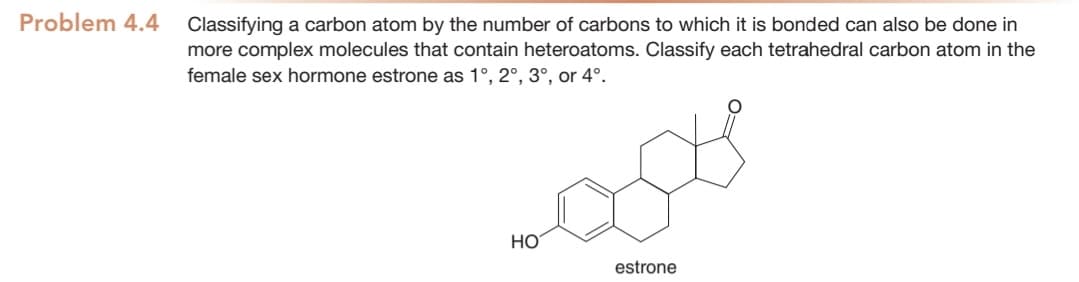
Classifying a carbon atom by the number of carbons to which it is bonded can also be done in more complex molecules that contain heteroatoms. Classify each tetrahedral carbon atom in the female sex hormone estrone as 1°, 2°, 3°, or 4°. HO estrone
Q: Draw a stepwise mechanism for the following reaction.
A:
Q: 2. Identify THREE functional groups presents in each compound A and FOUR functional groups presents…
A: In the above natural products, several functional groups are present and these are listed below. The…
Q: 5. Which of the following molecules are constitutional isomers? CH3CH2CH2OH CH;CH(OH)CH3 CH;CH2OCH3…
A: Isomers are the compounds having same molecular formula but having different properties i.e.…
Q: Draw the molecular frameworks for each molecule and tell how many valence electrons are required to…
A: A covalent bond is formed by the sharing of electrons. If the sharing atoms have the same…
Q: From the following structure locate these examples of types of atoms or bonds: 1. a highly polarized…
A: In given structure we have to locate the following 1. a highly polarized single bond2. A highly…
Q: Our friend carbon. Recall that all organic molecules, by definition, contain carbon. Carbon is a…
A:
Q: which of the following would have free rotation around the two carbon atoms? i. acetylene, HCCH…
A: We are given three different molecules we have to predict whether the free rotations is possible…
Q: Complete these structural formulas by adding enough hydrogens to complete the tetravalence of each…
A: As valency of carbon is four, carbon atoms with single bond will be attached to three hydrogen atoms…
Q: Name: Condensed Molecular Structural Formula Formula Sketch Ethers Dimethyl ether CH,-O-CH, C,H,O…
A: Ethers are the class of compounds having general formula ROR where R is an alkyl group. The sketch…
Q: 1. Which of the following compounds can exist as pairs of cis-trans isomers? Draw each cis-trans…
A: Since you have posted a question with multiple subparts, we will solve only first three sub parts…
Q: Use the structures shown below to count the number of degrees of unsaturation for each molecule. он…
A: Degrees of unsaturation :- The total number of rings and π bonds present in an organic molecule is…
Q: Classify the following pair of molecules as constitutional is omers, cis/trans isomers, different…
A: constitutional isomers: compounds having same molecular formula but difference in connectivity.
Q: Which of the following CAN exist as cis-trans isomers? I. CH3CH2CH=CHCH3 II. CICH=CHCI III.…
A: An isomers which have same molecular formula but different arrangements of atoms .
Q: 5. Identify all functional groups in the structure of fusidic acid (circle each group and name it…
A:
Q: Which one of the following compounds could have exactly one ring and two C3DO double bonds? O…
A: Ans a
Q: For which compounds can a second resonance structure be drawn? Draw an additional resonance…
A: Resonance structures are the Lewis structures having the same placement of atoms but a different…
Q: 1. Calculate the number of elements of unsaturation implied by the molecular formula: a. CaH12 b.…
A: Molecular formula = CxHyXzNa (DBE = x- (y+z-a)/2 + 1).
Q: Which of the following compounds exists as cis-trans isomers? Draw the structures of those that do.…
A: Double bonded carbon having both remaining vacancy is satisfied by two different groups ,then it…
Q: Describe the shape of the acetone molecule: :0: | | || C2 Сз - Н C1: H C2: H - C1 C3:
A: This is acetone molecule
Q: Predict the geometry around each indicated atom. a. CH;CH2CH2CH3 e. CH3-C-oH C. b. (CHa)2N d. BF f.…
A: Since you are posted with multiple sub-parts. As per the rule, I am answering the first three…
Q: e) How many asymmetric carbon atoms are present in the following compound? . NHCH3 A) 0 B) 1| C) 2…
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: 1.Determine the number of lone pairs (if any) on each oxygen, nitrogen, and charged carbon atoms in…
A: Lone pair of electrons on an atom are the electrons that are unshared pair of electrons present on…
Q: How many of the following molecules exhibit cis, trans isomerism? H,C CH,CH, CH, CH,CH, CH,CH, H,C…
A:
Q: Between which two carbons would a double bond form according to Zaitsev's Rule? 3 2 4 Br 2-4 1-3 3-4…
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: Is cis-trans isomerism possible for either of the double bonds of geraniol, a naturally occurring…
A: Cis-trans isomerism is a kind a sterioisomerism in which the atoms have different arrangement in 3D…
Q: On each chair structure identify the relationship of the substituents as cis or trans? II | = trans;…
A:
Q: Metvixia is a cream used to treat precancerous skin growths. Identify the number of double bonds in…
A: double bond are the pi bonds that is located in between two atoms.
Q: Classifying a carbon atom by the number of carbons to which it is bonded can also be done in more…
A: sp3 hybridized carbon atoms are those atoms that have arranged themselves in the tetrahedral…
Q: There are four cis-trans isomers of menthol including the one shown Which of the following…
A: Cis-trans isomers are those compounds which have same structural formula but different configuration…
Q: For which compounds can a second resonance structure be drawn?Draw an additional resonance structure…
A:
Q: Identify a to f CH3 CHCH,OH CH3 снсно PBR3 HCN H30* C CH3 CHCH MgBr
A:
Q: 2. Indicate the direction of the polarity for each of the bonds indicated except for all carbon to…
A:
Q: Which of the following statements is/are wrong? 1. Shape of sp2 hybridized orbital is tetrahedral.…
A: The sp2 hybridized orbital is formed by the mixing of one s-orbital and two p-orbitals to give three…
Q: Capsaicin is an active component of chili peppers, which are plan belonging to the genus Capsicum.…
A: The geometry around an atom in the molecule depends upon its hybridisation. For example, for sp3…
Q: 5. The CCO bond angle in acetone (CH3COCH3) is degrees. 6. In boron trifluoride (BF3) the boron atom…
A: 5. The CCO bond angle in acetone is 120 degrees. 6. In boron trifluoride (BF3) the boron atom is…
Q: The carbon skeleton for 2-methylpropane is shown below. How many hydrogens are present? c-d-c 4 9 10…
A: Since we know that valency of carbon = 4
Q: which hadrocarbon contains both sp3 and sp2 hybridized atom 1. cyclopropane 2. propane 3.…
A: The hydrocarbon which contains both sp3 and sp2 hybridized atom has to be given,
Q: Benzene is especially stable due to a. the electrons of the double bonds are delocalized b. its…
A: Benzene is an aromatic compound. The molecular formula of benzene is C6H6. The structure of the…
Q: The three-dimensional structure of organic molecules is very important in many areas including in…
A: Three dimensional structure of a molecule represents the structure in which the elements are out of…
Q: Determine the number of hydrogen atoms.in a compound which has 1 ring and 5 double bonds.…
A:
Q: 2) Determine the IHD of each molecule: a) C6H1205C12 b) C4H1ÓN6B14
A:
Q: What two atomic orbitals or hybrid atomic orbitals overlap to form the C-C o bond in acetylene? O C…
A:
Q: What are the absolute configurations of the two central carbon atoms in this compound? C6H5 CH3 SCH3…
A: Note! We can answer only first question (as you have not specified any) as per bartleby guidelines.…
Q: 1. For each of the following molecules, draw a clearly labeled diagram showing the hydrogen bonding…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What type of orbital overlap is responsible for the n bond between carbon and oxygen in the molecule…
A: The given molecule is The electronic configuration for Carbon is [He]2s22p2. Thus, there are 4…
Q: Theobromine (Greek theobroma – “food of the gods") is a constituent of cocoa. How many lone pairs…
A: Structures of theobromine which is a constituent of cocoa and melamine which is a plasticizer are…
Q: In the following molecule, how many MeO groups in the following molecule are in axial position?
A: The objective of the question is to understand and identify how many -OMe groups in substituted…
Q: Which of the following statements can be used to prove that carbon is tetrahedral? a. CH3Br does not…
A: Constitutional Isomers: The compounds that have the same molecular formula but different in their…
Q: a. Propane, CH,CH,CH; b. Heptane, CH;(CH,);CH; c. Propanol, CH;CH,CH,OH d. Heptanol, CH;(CH,),OH 2.…
A: Note: according to our guidelines we are supposed to answer only one question
Q: 1. Give IUPAC names for the following compounds: (a) H3C CH3 (b) CH3 H2C=CHCHCCH3 CH3CH2CH=ĊCH2CH3…
A:
Step by step
Solved in 2 steps with 1 images

- A. Draw the meantime r of ephedrine. You can use line structures or a hybrid line structure/condense drawing like the one shown. Be sure it is clear which bonds are wedge and which are dash. B. Draw a diastereomer of ephedrine.How is each compound (B–D) related to A? Choose from enantiomers, diastereomers, identical molecules, constitutional isomers, or not isomers of each other.Organic chemistry 5.6.7.
- Make sure all requirements are filled drawing the isomer. from a to fbased on the following, choose: a) Pigment of B has more conjugated double bonds than pigment A b) Pigment B has an HOMO-LUMO transition greater than pigment A c) Pigment B emits photons with lower energy than Pigment A d) Pigment B can be seen by naked eye.Answer the following questions about erlotinib and terbinafine. Erlotinib,sold under the trade name Tarceva, was introduced in 2004 for thetreatment of lung cancer. Terbinafine is an antifungal medication used totreat ringworm and fungal nail infections. Rank the labeled bonds in terbinafine in order of increasing bondstrength.
- Zolpidem (trade name Ambien) promotes the rapid onset of sleep, making it a widely prescribed drug for treating insomnia. a.In what type of orbital does the lone pair on each N atom in the heterocycle reside? b. Explain why the bicyclic ring system that contains both N atoms is aromatic. c.Draw all reasonable resonance structures for the bicyclic ring system.Classify each compound as identical to A or its enantiomer.Drawn are four isomeric dimethylcyclopropanes. a.How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D? b.Label each compound as chiral or achiral. c.Which compounds alone would be optically active? d.Which compounds have a plane of symmetry? e.How do the boiling points of the compounds in each pair compare: A and B; B and C; C and D? f.Which of the compounds are meso compounds? g.Would an equal mixture of compounds C and D be optically active? What about an equal mixture of B and C?
- Label each of the following as cis, trans or neither. Below each structure that is cis draw a transconfigurational stereoisomer or vice versa.How many rings and π bonds are contained in compounds A–C? Draw one possible structure for each compound. a. Compound A has molecular formula C5H18 and is hydrogenated to a compound having molecular formula C510H10. b. Compound B has molecular formula C10H16 and is hydrogenated to a compound having molecular formula C10H18. c. Compound C has molecular formula C8H8 and is hydrogenated to a compound having molecular formula C8H16.The [α] of pure quinine, an antimalarial drug, is −165.Calculate the ee of a solution with the following [α] values: −50, −83,and −120.For each ee, calculate the percent of each enantiomer present.What is [α] for the enantiomer of quinine?If a solution contains 80% quinine and 20% of its enantiomer, what isthe ee of the solution?What is [α] for the solution described in part (d)?

